Hb Synthesis Intro - Blood's Building Blocks
- Hemoglobin (Hb): Primary O₂ transport protein in Red Blood Cells (RBCs).
- Structure: Tetramer; 4 globin chains, each covalently bound to a heme group.
- Heme: Ferrous iron (Fe²⁺) + Protoporphyrin IX.
- Globin: Polypeptide chains (e.g., α, β).
- Major Adult Hemoglobins:
- HbA ($α_2β_2$): >95%
- HbA2 ($α_2δ_2$): 2-3.5%
- HbF ($α_2γ_2$): <2% (fetal Hb)
- Site of Synthesis: Predominantly in erythroid precursors in bone marrow.
⭐ Heme is synthesized in both mitochondria and cytoplasm of erythroid cells, while globin chains are synthesized on ribosomes in the cytoplasm.
Heme Pathway - Crimson Core Creation
Heme, an iron-porphyrin compound, is crucial for oxygen binding in hemoglobin. Its synthesis involves eight enzymatic steps, starting in mitochondria, moving to cytoplasm, and returning to mitochondria for final heme production. Defects lead to porphyrias.
- Rate-Limiting Step (Mitochondria):
- $Glycine + Succinyl CoA \rightarrow \delta-ALA$ by ALA Synthase.
- Requires: Pyridoxal Phosphate (B6).
- Regulation: Heme (↓ feedback), drugs like barbiturates (↑ induction).
- Cytoplasmic Conversions:
- $2 \times \delta-ALA \rightarrow Porphobilinogen (PBG)$ by ALA Dehydratase. ⚠️ Pb sensitive.
- $4 \times PBG \rightarrow Hydroxymethylbilane$ by PBG Deaminase (HMB Synthase). Deficiency: Acute Intermittent Porphyria (AIP) - neurovisceral symptoms, port-wine urine on standing.
- Hydroxymethylbilane $\rightarrow$ Uroporphyrinogen III $\rightarrow$ Coproporphyrinogen III.
- Uroporphyrinogen Decarboxylase deficiency: Porphyria Cutanea Tarda (PCT) - most common porphyria, photosensitivity, skin lesions.
- Final Steps (Mitochondria):
- Coproporphyrinogen III $\rightarrow$ Protoporphyrin IX.
- Protoporphyrin IX + Fe²⁺ $\rightarrow$ Heme by Ferrochelatase. ⚠️ Pb sensitive.
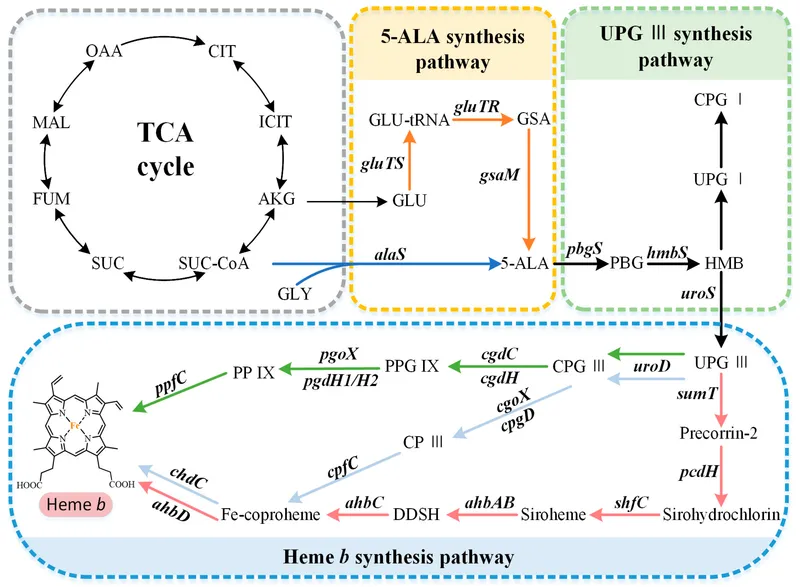
⭐ Lead poisoning inhibits both ALA Dehydratase and Ferrochelatase, causing accumulation of $\delta$-ALA (neurotoxic) and Protoporphyrin IX, respectively. This results in microcytic, hypochromic anemia with basophilic stippling.
Globin & Assembly - Protein Partnering
- Globin Chain Synthesis:
- Site: Cytoplasmic ribosomes in erythroid precursors (normoblasts, reticulocytes).
- Genetic Control:
- α-globin gene cluster: Chromosome 16 (HBA1, HBA2 genes).
- β-globin gene cluster: Chromosome 11 (HBB, HBD, HBG1, HBG2, HBE1 genes).
- 📌 Mnemonic: Alpha-16 (Sweet 16), Beta-11 (One-One, like legs).
- Key Globin Chains & Hemoglobins (Hb):
- Adult: HbA (α₂β₂, >95%), HbA₂ (α₂δ₂, 2-3.5%), HbF (α₂γ₂, <2%).
- Fetal: HbF (α₂γ₂) - higher $O_2$ affinity, predominant until ~6 months post-birth.
- Assembly Process:
- Individual globin polypeptide chains are synthesized.
- α-chains combine with non-α chains (β, γ, or δ) to form stable heterodimers (e.g., αβ).
- Two heterodimers associate to form the final tetrameric hemoglobin (e.g., α₂β₂).
- Heme Incorporation: Each globin chain enfolds one heme molecule within a hydrophobic pocket, protecting the $Fe^{2+}$ ion.
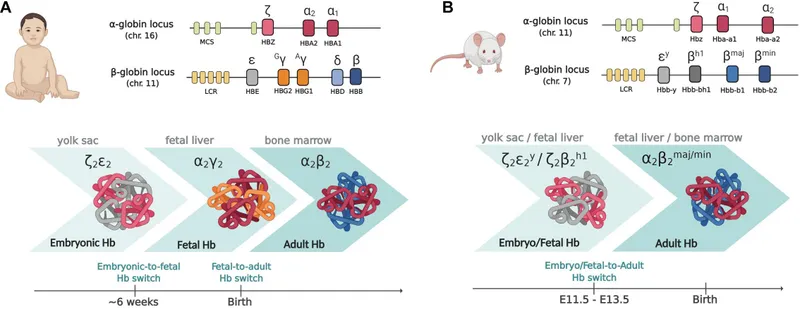
⭐ Balanced synthesis of α and non-α globin chains (ratio ~1:1) is crucial; an imbalance leads to thalassemias due to precipitation of excess unmatched chains.
Porphyrias - Synthesis Gone Wrong
- Genetic/acquired enzyme defects in heme synthesis; specific porphyrin precursors accumulate.
- Acute Intermittent Porphyria (AIP):
- Defect: HMB synthase (PBG deaminase).
- ↑ ALA, PBG in urine.
- Symptoms: Acute neurovisceral attacks. 📌 5 P's: Painful abdomen, Polyneuropathy, Psychological disturbances, Pink urine, Precipitated by drugs. No photosensitivity.
- Porphyria Cutanea Tarda (PCT):
- Defect: Uroporphyrinogen decarboxylase.
- ↑ Uroporphyrins.
- Symptoms: Photosensitivity, chronic blistering skin lesions. ⭐ > PCT is the most common porphyria; urine appears tea-colored or pink under Wood's lamp.
- Lead Poisoning: Inhibits ALA dehydratase & Ferrochelatase. ↑ALA, ↑protoporphyrin (zinc protoporphyrin).
High‑Yield Points - ⚡ Biggest Takeaways
- Heme synthesis occurs in mitochondria and cytoplasm of erythroid precursors.
- ALA synthase is the rate-limiting enzyme, requiring pyridoxal phosphate (B6).
- Key substrates are succinyl CoA and glycine; first step in mitochondria.
- Lead poisoning inhibits ALA dehydratase and ferrochelatase, causing microcytic anemia.
- Heme provides feedback inhibition to ALA synthase, regulating its own production.
- Globin chains are synthesized on ribosomes in the cytoplasm.
- Ferrochelatase adds Fe²⁺ to protoporphyrin IX, forming heme in mitochondria.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

