Allosteric Regulation Basics - Switch On, Switch Off
- Allosteric Enzyme: An enzyme with an allosteric site, distinct from its active site, allowing activity modulation.
- Allosteric Site: 📌 'Allo' = 'other'. Specific regulatory site on the enzyme, separate from active site, where effectors bind.
- Allosteric Effectors (Modulators): Small molecules binding to allosteric sites, altering enzyme activity (↑ positive, ↓ negative).
- Positive Effectors (Activators): ↑ enzyme activity.
- Negative Effectors (Inhibitors): ↓ enzyme activity.
- Key Characteristics:
- Reversible, non-covalent effector binding.
- Induces conformational change, transmitted to active site, altering substrate affinity/catalytic rate.

⭐ Allosteric enzymes are often oligomeric (multisubunit) proteins, enabling cooperativity.
Allosteric Mechanisms & Models - Shape Shifters at Work
Allosteric enzymes: effectors bind to allosteric sites (not active site), inducing conformational changes altering activity.
- Conformational States:
- T (Tense) state: ↓ affinity, favored by inhibitors.
- R (Relaxed) state: ↑ affinity, favored by activators.
- Cooperativity: Ligand binding affects further binding.
- Homotropic: Substrate as effector (e.g., O₂ for Hb).
- Heterotropic: Different effector (e.g., ATP/CTP for ATCase).

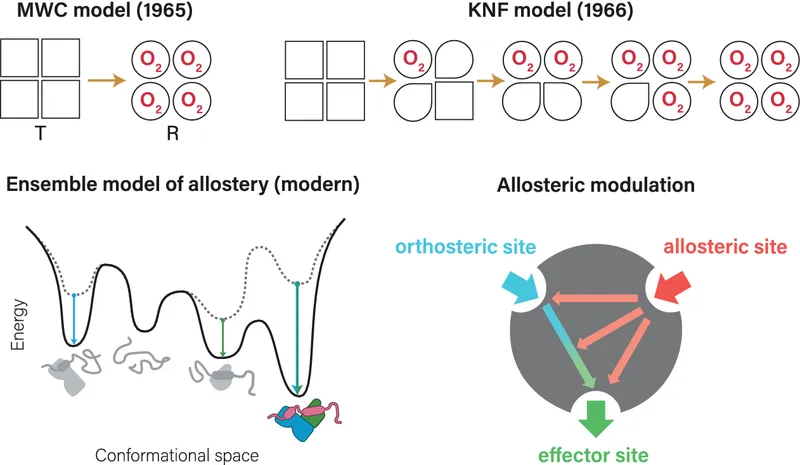
- Models of Allostery:
Feature MWC (Concerted) KNF (Sequential) Transition All subunits change simultaneously Sequential change, induced fit Pre-existing Eqm. Yes ($T \rightleftharpoons R$ equilibrium) Ligand induces fit; sequential change Symmetry Preserved (all-T or all-R) Intermediates (mixed T/R) possible Negative Coop. Not easily explained Can explain
⭐ The MWC model assumes all subunits change conformation simultaneously, while the KNF model allows for sequential changes.
Allosteric Enzyme Kinetics - Curve Ball Kinetics
- Curve: Sigmoidal (S-shaped) v vs. [S] plot, distinct from Michaelis-Menten's hyperbolic curve. Indicates cooperativity.
- K0.5: [S] for 1/2 $V_{max}$; reflects enzyme affinity in allosteric enzymes.
- Allosteric Activators:
- Shift curve left (↓K0.5, ↑affinity).
- Favor R (relaxed, high-affinity) state.
- Types: K-type (↓K0.5), V-type (↑$V_{max}$).
- Allosteric Inhibitors:
- Shift curve right (↑K0.5, ↓affinity).
- Favor T (taut, low-affinity) state.
- Types: K-type (↑K0.5), V-type (↓$V_{max}$).
- Hill Equation: Describes cooperativity: $v = V_{max} [S]^{n_H} / (K_{0.5}^{n_H} + [S]^{n_H})$.
- Hill Coefficient ($n_H$): Measures degree of cooperativity.
- $n_H > \textbf{1}$: Positive cooperativity.
- $n_H < \textbf{1}$: Negative cooperativity.
- $n_H = \textbf{1}$: No cooperativity (Michaelis-Menten like).
⭐ Positive cooperativity: one substrate binding increases the enzyme's affinity for subsequent substrate molecules.
Key Examples & Clinical Impact - Allostery in Action
Key allosteric molecules:
-
Phosphofructokinase-1 (PFK-1): Pivotal enzyme in glycolysis.
- Activators: AMP, Fructose-2,6-bisphosphate (signal low energy).
- Inhibitors: ATP, Citrate (signal high energy).
- 📌 PFK-1: 'ATP inhibits, AMP activates Progress of glycolysis'.
⭐ High ATP levels allosterically inhibit PFK-1, signaling that the cell has sufficient energy.
-
Aspartate Transcarbamoylase (ATCase): Early step in pyrimidine biosynthesis.
- Activator: ATP.
- Inhibitor: CTP (end-product feedback inhibition).
-
Hemoglobin (Hb): Classic allosteric protein (not an enzyme), crucial for $O_2$ transport.
- Homotropic positive effector: $O_2$.
- Heterotropic negative effectors: $H^+$, $CO_2$, 2,3-Bisphosphoglycerate (2,3-BPG).
- Clinical Relevance: Allosteric drugs act as precise modulators.
- Examples: Cinacalcet (CaSR in parathyroid disorders), Maraviroc (CCR5 in HIV).
High‑Yield Points - ⚡ Biggest Takeaways
- Allosteric enzymes possess regulatory sites distinct from their active sites.
- Binding of allosteric modulators (activators or inhibitors) causes conformational changes.
- They typically display sigmoidal kinetics (cooperativity), not Michaelis-Menten.
- Homotropic effectors are substrates; heterotropic effectors are non-substrate molecules.
- PFK-1 (glycolysis) is a classic example, regulated by ATP (inhibitor) and AMP (activator).
- This regulation provides fine-tuned control over metabolic pathways.
- K-type modulators alter Km (substrate affinity); V-type modulators alter Vmax (maximal velocity).
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

