ETC Overview - Electron Hustle
- Location: Inner Mitochondrial Membrane (IMM); cristae ↑ surface area.
- Function: Series of redox reactions. Electrons ($e^-$) from NADH/FADH₂ pass down chain. Energy released pumps $H^+$ from matrix to intermembrane space (IMS), creating proton gradient.
- Electron Carriers:
- Mobile: Ubiquinone (CoQ), Cytochrome c.
- Fixed: Flavoproteins (FMN, FAD), Iron-Sulfur (Fe-S) proteins, Cytochromes.
- Electron Flow & $H^+$ Pumping: (Protons pumped per $e^-$ pair)
- NADH → Complex I (4$H^+$) → CoQ → Complex III (4$H^+$) → Cyt c → Complex IV (2$H^+$) → $O_2$
- FADH₂ → Complex II → CoQ → Complex III (4$H^+$) → Cyt c → Complex IV (2$H^+$) → $O_2$
- Complex II (Succinate Dehydrogenase) does NOT pump protons.
- Final $e^-$ Acceptor: $O_2$ reduced to $H_2O$.
⭐ Cyanide (CN⁻) & CO inhibit Complex IV (Cytochrome c oxidase), blocking $O_2$ use.
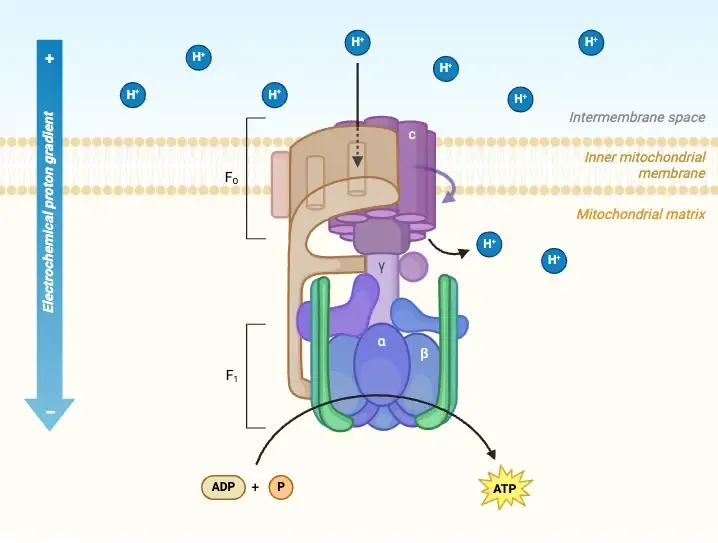
ATP Synthesis - Proton Powerhouse
- ATP Synthase (Complex V): Inner Mitochondrial Membrane (IMM) enzyme; utilizes proton-motive force (PMF) for ATP synthesis.
- F₀ subunit: IMM proton channel (H⁺ flow: Intermembrane Space (IMS) → Matrix).
- F₁ subunit: Matrix-side; contains catalytic sites for $ADP + P_i \rightarrow ATP$.
- Chemiosmotic Mechanism (Boyer's Binding Change Model):
- H⁺ flow via F₀ rotates its c-ring & the γ-subunit of F₁.
- Rotation drives sequential conformational changes (Loose-Tight-Open) in F₁ β-subunits, leading to ATP synthesis & release.
- Stoichiometry & Yield:
- Requires ~3-4 H⁺ (typically 4 H⁺) translocated per 1 ATP synthesized.
- P/O Ratios: NADH ≈ 2.5 ATP; FADH₂ ≈ 1.5 ATP.
- Inhibitor:
- Oligomycin: Binds to F₀ subunit, blocks the H⁺ channel, thereby halting ATP synthesis.
⭐ The F₁ component of ATP synthase contains the catalytic sites, and its β-subunits cycle through three conformations: Open (O), Loose (L), and Tight (T) to produce ATP.
Regulation & Yield - Energy Accountants
- Regulation (Respiratory Control):
- Acceptor Control: Primary. ↑ADP stimulates; ↑ATP inhibits. ATP/ADP ratio key.
- Substrate Availability: O2, NADH, FADH2.
- IF1 (Inhibitory Factor 1): Hypoxia-active, prevents ATP hydrolysis by F1Fo-ATPase.
- ATP Yield (P/O Ratios):
- NADH (mito./M-A shuttle): 2.5 ATP.
- NADH (cyto. via G3P shuttle): 1.5 ATP.
- FADH2 (mitochondrial): 1.5 ATP.
- Total ATP per Glucose (Aerobic Respiration):
- 32 ATP (M-A shuttle for cyto. NADH; e.g., heart, liver, kidney).
- 30 ATP (G3P shuttle for cyto. NADH; e.g., sk. muscle, brain).
⭐ Uncoupling proteins (e.g., UCP1/Thermogenin in brown adipose tissue) dissipate the proton gradient as heat, reducing ATP yield but crucial for non-shivering thermogenesis.
Inhibitors & Uncouplers - Chain Saboteurs
ETC Inhibitors: Block $e^-$ flow; ↓ATP, ↓O₂ use.
- Complex I: Rotenone, Piericidin A, Amytal, MPP+.
- 📌 Really Potent Agents Mess-up Complex I.
- Complex II: Malonate (competitive).
- Complex III: Antimycin A, Dimercaprol.
- Complex IV: Cyanide (CN⁻), CO, H₂S, Azide.
- 📌 Can Complex IV Halt Suddenly? Absolutely!
- ATP Synthase (Complex V): Oligomycin.
Uncouplers: Disrupt $H^+$ gradient. ETC & O₂ use ↑, ATP ↓. Energy $ ightarrow$ heat.
- 2,4-Dinitrophenol (DNP)
- Aspirin (high doses)
- Thermogenin (UCP1, brown fat)
- Pentachlorophenol
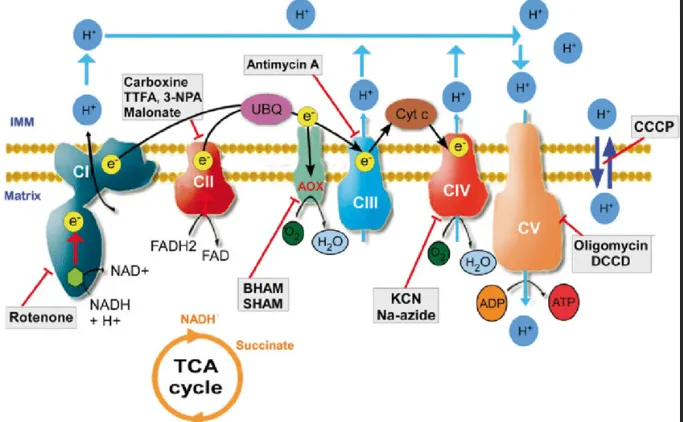
⭐ Cyanide (CN⁻) inhibits Complex IV, causing histotoxic hypoxia (cells can't use O₂ despite normal or high arterial oxygen levels).
High‑Yield Points - ⚡ Biggest Takeaways
- Location: Inner mitochondrial membrane; O₂ is the final electron acceptor.
- ETC complexes (I-IV) transfer electrons, creating a proton gradient across this membrane.
- ATP synthase (Complex V) harnesses this gradient for ATP synthesis (chemiosmosis).
- Inhibitors block ETC at specific sites: Rotenone/Amytal (Complex I), Antimycin A (Complex III), Cyanide/CO/Azide (Complex IV).
- Uncouplers (e.g., DNP, aspirin, thermogenin) dissipate proton gradient, ↓ATP synthesis, ↑heat production.
- P/O ratio (ATP per O atom reduced): NADH ≈ 2.5 ATP; FADH₂ ≈ 1.5 ATP.
- The chemiosmotic theory by Peter Mitchell explains the coupling of electron transport to ATP synthesis via the proton gradient.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more