ETC Overview - Electron Expressway
- The "Electron Expressway": Final common pathway for oxidizing fuel molecules (e.g., glucose, fatty acids).
- Location: Inner Mitochondrial Membrane (IMM); cristae increase surface area for efficiency.
- Purpose: Harvests energy from $NADH$ and $FADH_2$ by transferring their electrons sequentially to $O_2$.
- Process: Electrons flow through multi-protein complexes (I-IV) and mobile carriers (Coenzyme Q, Cytochrome c).
- Energy released pumps protons (H⁺), creating an electrochemical gradient (proton-motive force) that drives ATP synthesis.

⭐ Cyanide and Carbon Monoxide (CO) are potent inhibitors of Complex IV (Cytochrome c oxidase), halting cellular respiration and ATP production. This is a frequently tested toxicological point!
ETC Components - The Electron Team

- Complex I (NADH Dehydrogenase): $NADH \rightarrow NAD^+$ (FMN, Fe-S). Pumps 4H⁺; e⁻ to CoQ.
- Inhibitors: Rotenone, Amytal, Piericidin A (📌 RAP-1).
- Complex II (Succinate Dehydrogenase): $FADH_2 \rightarrow FAD$ (FAD, Fe-S). e⁻ to CoQ; No H⁺ pump.
- Inhibitor: Malonate. ⭐ > Complex II is unique: part of TCA cycle & ETC; does NOT pump H⁺.
- Coenzyme Q (Ubiquinone): Mobile lipid carrier; e⁻ from C-I & C-II to C-III.
- Complex III (Cytochrome bc₁): $QH_2 \rightarrow Q$ (Hemes, Fe-S). Pumps 4H⁺; e⁻ to Cyt c; Q-cycle.
- Inhibitor: Antimycin A (📌 Anti-3).
- Cytochrome c: Mobile protein carrier (intermembrane space).
- Complex IV (Cytochrome c Oxidase): Uses Cyt c e⁻ (Heme a, a₃, CuA, CuB). Pumps 2H⁺; $\frac{1}{2}O_2 + 2H^{+} \rightarrow H_2O$.
- Inhibitors: CN⁻, CO, N₃⁻ (Azide) (📌 C.O.N. IV).
Mechanism & PMF - Proton Power Play
- NADH & FADH₂ donate e⁻; flow via CoQ, Cyt c to O₂ (final acceptor).
- NADH path: Cmplx I → CoQ → Cmplx III → Cyt c → Cmplx IV → O₂
- FADH₂ path: Cmplx II → CoQ → Cmplx III → Cyt c → Cmplx IV → O₂
- Proton Pumping: H⁺ actively moved from matrix to IMS by Complexes I, III, IV.
- Complex I: 4H⁺
- Complex III: 4H⁺
- Complex IV: 2H⁺ (Note: Complex II pumps 0H⁺)
- PMF: Electrochemical H⁺ gradient (ΔpH chemical, ΔΨ electrical) across inner membrane.
- Drives ATP synthesis via ATP Synthase (Complex V).
⭐ Peter Mitchell's Chemiosmotic theory: PMF powers ATP synthesis.
 oka
oka
Oxidative Phosphorylation - ATP Factory Firing
- ATP Synthase (Complex V): F₀ ($H^+$ channel) & F₁ (catalytic site, makes ATP).
- Chemiosmosis: $H^+$ pumped to intermembrane space (IMS) creates proton-motive force (PMF).
- PMF drives $H^+$ via F₀, rotating F₁ ($ ext{γ}$ subunit), making ATP from ADP + $P_i$.
- Oligomycin: Inhibits F₀, blocks $H^+$ flow, stops ATP synthesis.
- Uncouplers (DNP, thermogenin): Disrupt PMF; energy lost as heat. 📌 UCPs = UnCouple Protons.
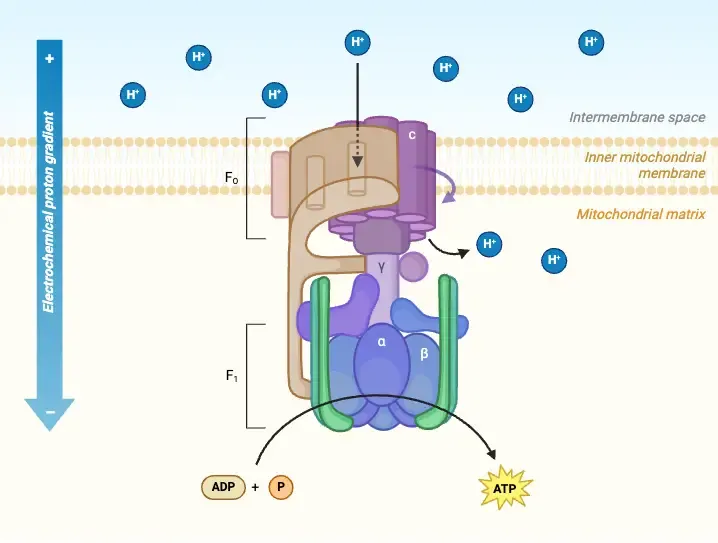
⭐ ATP synthase's F₁ uses a "binding change mechanism" (Open, Loose, Tight states) for ATP synthesis.
Inhibitors & Uncouplers - Chain Blockers Breakers
- ETC Inhibitors (Blockers): Halt electron (e⁻) flow, ↓ATP synthesis, ↓O₂ consumption.
- C-I: Rotenone, Amytal, Piericidin A. 📌 R.A.P. at One.
- C-III: Antimycin A.
- C-IV: Cyanide (CN⁻), CO, H₂S, Azide (N₃⁻).
- ATP Synthase (Complex V): Oligomycin.
- Uncouplers (Breakers): Disrupt proton (H⁺) gradient; ↓ATP synthesis, BUT ↑O₂ consumption, ↑Heat production.
- E.g., 2,4-Dinitrophenol (DNP), Aspirin (high doses), Thermogenin (UCP1).
⭐ Cyanide inhibits Complex IV (Cytochrome c oxidase), causing histotoxic hypoxia; characterized by almond odor on breath (not always present).
High‑Yield Points - ⚡ Biggest Takeaways
- ETC in inner mitochondrial membrane: major ATP production via oxidative phosphorylation.
- Electrons from NADH/FADH2 flow via complexes I-IV to Oxygen (final acceptor).
- Proton pumping creates electrochemical gradient (proton-motive force).
- ATP synthase (Complex V) uses this gradient for ATP synthesis (chemiosmosis).
- Inhibitors (e.g., Cyanide, Rotenone) block ETC; Uncouplers (e.g., DNP) disrupt gradient, make heat.
- P:O Ratios: ~2.5 ATP/NADH, ~1.5 ATP/FADH2.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

