ATP: Structure & Function - The Body's Battery
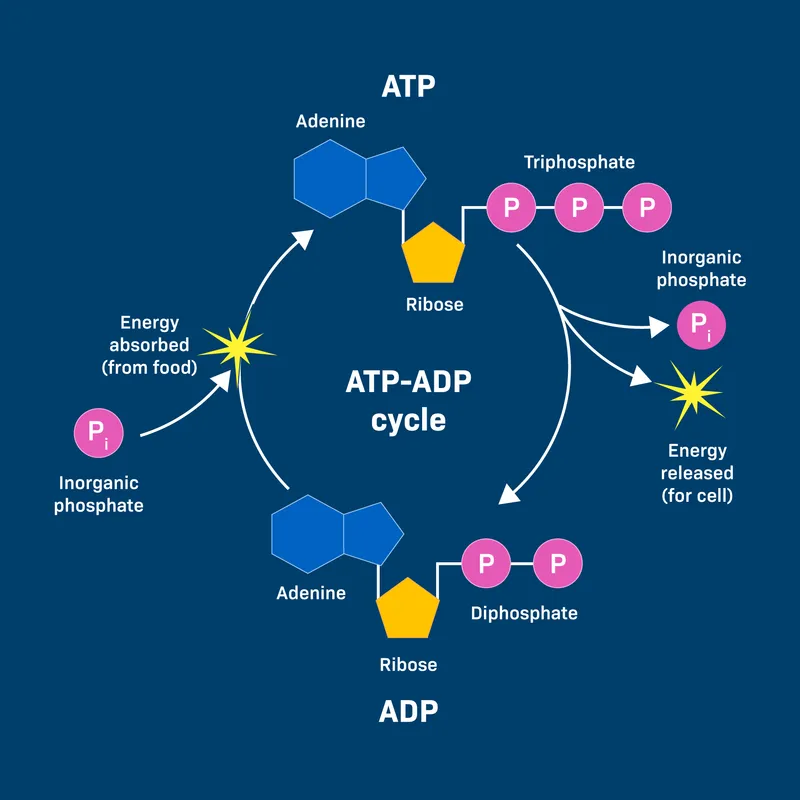
- Structure: Composed of Adenine (nitrogenous base), Ribose (pentose sugar), and a Triphosphate group ($\alpha, \beta, \gamma$ phosphates).
- Two high-energy phosphoanhydride bonds link the phosphate groups.
- Often called the "energy currency" of the cell.

- Function: Primary molecule for storing and transferring energy in cells.
- Energy Release: Hydrolysis of terminal phosphate(s).
- ATP + $H_2O \rightarrow$ ADP + $P_i$ + Energy ($\Delta G \approx \textbf{-30.5}$ kJ/mol or $\textbf{-7.3}$ kcal/mol).
- ATP + $H_2O \rightarrow$ AMP + $PP_i$ + Energy (larger release).
- Powers cellular work: Biosynthesis, active transport, muscle contraction, nerve transmission.
- Energy Release: Hydrolysis of terminal phosphate(s).
⭐ $Mg^{2+}$ is essential for ATP biological function; ATP usually exists as MgATP complex, which is the true substrate for most kinases and ATPases.
ATP Synthesis Mechanisms - Energy Factories
ATP is primarily generated via two mechanisms: Substrate-Level Phosphorylation (SLP) and Oxidative Phosphorylation (OP).
-
Substrate-Level Phosphorylation (SLP)
- Direct transfer of $PO_4^{3-}$ from a high-energy substrate to ADP $\rightarrow$ ATP.
- Occurs in cytoplasm (Glycolysis) & mitochondrial matrix (Krebs cycle).
- Examples: Phosphoenolpyruvate $\rightarrow$ Pyruvate; Succinyl CoA $\rightarrow$ Succinate.
- Enzymes: Kinases.
- Anaerobic or aerobic.
-
Oxidative Phosphorylation (OP)
- Major ATP source (~90%). Indirect, via chemiosmosis.
- Location: Inner Mitochondrial Membrane (IMM).
- Process:
- Electrons from NADH/FADH₂ pass through Electron Transport Chain (ETC).
- $O_2$ is the final electron acceptor, forming $H_2O$.
- Energy released pumps $H^+$ from matrix to intermembrane space, creating a proton gradient.
- $H^+$ flow back via ATP synthase ($F_0F_1$ ATPase) drives ATP synthesis.
⭐ P/O Ratios: NADH yields ~2.5 ATP; FADH₂ yields ~1.5 ATP.

ATP Hydrolysis & Utilization - Spending Spree
- Energy Release:
- Hydrolysis of terminal phosphoanhydride bonds:
- $ATP + H_2O \rightarrow ADP + P_i$; $ \Delta G^{\circ'} $ ≈ -30.5 kJ/mol (-7.3 kcal/mol)
- $ATP + H_2O \rightarrow AMP + PP_i$; $ \Delta G^{\circ'} $ ≈ -45.6 kJ/mol ($PP_i$ rapidly hydrolyzed to $2P_i$, pulling reaction forward)
- "High-energy" bonds ($~P$) release significant free energy upon hydrolysis.
- Hydrolysis of terminal phosphoanhydride bonds:
- Cellular Work Examples:
- Mechanical: Muscle contraction.
- Active transport: Na+/K+ pump, $Ca^{2+}$ pumps.
- Synthesis: Anabolic reactions (protein synthesis).
- Signaling: Kinase phosphorylation. 📌 Mnemonic: ATP for MASsive work (Mechanical, Active transport, Synthesis).
- Reaction Coupling: Exergonic ATP hydrolysis drives endergonic processes.
- $Mg^{2+}$ Role: ATP is $MgATP^{2-}$; $Mg^{2+}$ stabilizes phosphates, aids enzyme use.
⭐ Cellular ATP hydrolysis releases -50 to -65 kJ/mol (actual free energy change). This energy release is substantially larger than the standard free energy change value, due to prevailing reactant and product concentrations in cells.
ATP Regulation & Clinical Links - Energy Economics
- Feedback Control:
- High $ATP/ADP$ ratio, Citrate: Inhibit PFK-1 (glycolysis).
- High ATP: Inhibits Isocitrate Dehydrogenase (TCA).
- AMP: Activates PFK-1, Glycogen Phosphorylase.
- Hormonal: Insulin (anabolic), Glucagon/Epinephrine (catabolic).
- Clinical Links:
- Hypoxia: ↓ATP → $Na^{+}/K^{+}$ pump failure → cell swelling.
- Mitochondrial Myopathies (MELAS): Impaired ATP synthesis.
- Uncouplers (DNP, Aspirin OD): ↓ATP, ↑heat.
⭐ Cyanide inhibits Complex IV (Cytochrome c oxidase), halting ATP synthesis via oxidative phosphorylation.
High‑Yield Points - ⚡ Biggest Takeaways
- ATP is the universal energy currency of the cell, vital for all life processes.
- Hydrolysis of ATP to ADP + Pi releases significant free energy, approximately -7.3 kcal/mol.
- ATP structure includes adenine, ribose, and three phosphate groups, with two high-energy bonds.
- Substrate-level phosphorylation and oxidative phosphorylation are the two main mechanisms for ATP synthesis.
- Creatine phosphate in muscle/brain rapidly regenerates ATP during high demand anoxia or intense activity.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

