Xenobiotics - Body's Alien Busters
Xenobiotics are foreign chemical substances not naturally produced or expected within an organism (e.g., drugs, pesticides, pollutants). The body metabolizes them to facilitate excretion, primarily by increasing water solubility.
- Goal: Convert lipophilic compounds $\rightarrow$ hydrophilic compounds for easier excretion.
- Primary Site: Liver (smooth endoplasmic reticulum & cytosol). Other sites include GIT, lungs, kidneys, skin.
- Two Phases:
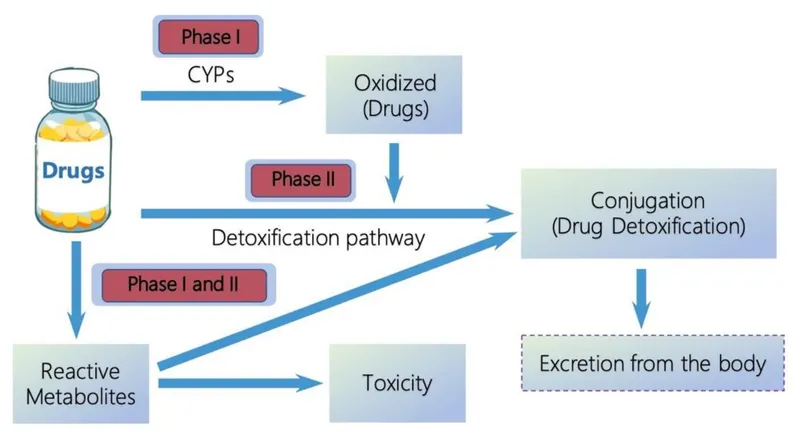
- Phase I (Functionalization):
- Reactions: Oxidation, reduction, hydrolysis.
- Key Enzymes: Cytochrome P450 (CYP450) superfamily, flavin-containing monooxygenases, epoxide hydrolases.
- Outcome: Introduces or exposes polar functional groups (e.g., $-\text{OH}$, $-\text{NH}_2$, $-\text{SH}$). Metabolites can be inactive, active (prodrug activation), or more toxic.
- Phase II (Conjugation):
- Reactions: Glucuronidation (most common), sulfation, glutathione conjugation, acetylation, methylation, amino acid conjugation.
- Outcome: Covalently adds endogenous polar molecules to the functional group from Phase I, significantly $\uparrow$ water solubility and molecular size, facilitating excretion (usually detoxification).
- Phase I (Functionalization):
⭐ Grapefruit juice inhibits intestinal CYP3A4, a key Phase I enzyme, leading to $\uparrow$ bioavailability and potential toxicity of many drugs like statins (e.g., simvastatin, atorvastatin) and calcium channel blockers (e.g., nifedipine).
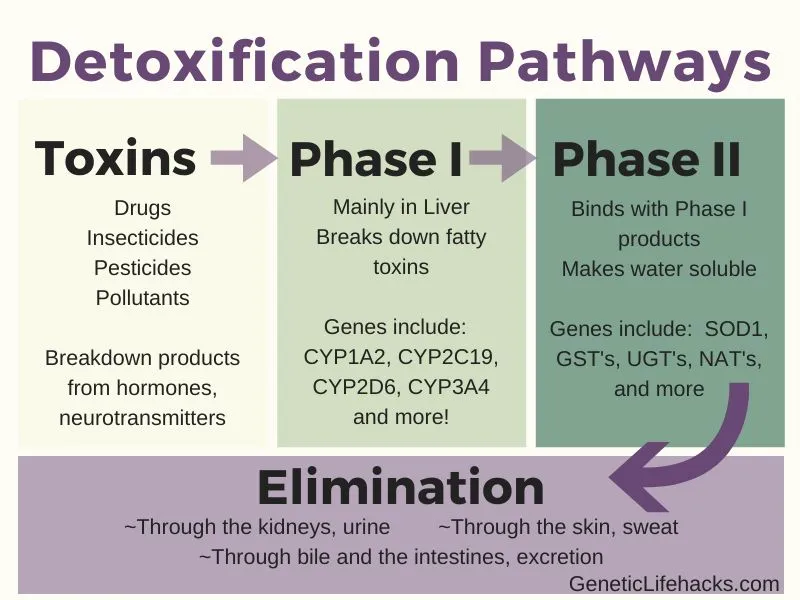
Phase II Rxns - Tag, Bag, & Drag!
- Goal: Conjugate xenobiotics/Phase I metabolites with endogenous molecules, ↑ water solubility for excretion.
- Mechanism: Adds polar groups; products usually inactive, readily excreted (urine/bile).
- Major Pathways:
- Glucuronidation: Most common. Enzyme: UGT. Donor: UDPGA. Site: ER.
- Sulfation: Enzyme: SULT. Donor: PAPS. Site: Cytosol.
- Acetylation: Enzyme: NAT. Donor: Acetyl CoA. Site: Cytosol.
- 📌 Polymorphism: Slow acetylators (e.g., isoniazid toxicity) vs. Fast.
- Glutathione Conjugation: Enzyme: GST. Donor: GSH. Site: Cytosol/Mito.
- Amino Acid Conjugation: e.g., Glycine, Taurine. Cofactors: ATP, CoA. Site: Mito/Cytosol.
- Methylation: Enzyme: Methyltransferase. Donor: SAM. Site: Cytosol.
⭐ Paracetamol at therapeutic doses is mainly metabolized by glucuronidation & sulfation. Overdose saturates these, leading to NAPQI formation (Phase I) & glutathione depletion, causing hepatotoxicity.
Metabolism Modulators - Xeno‑Factors Impact
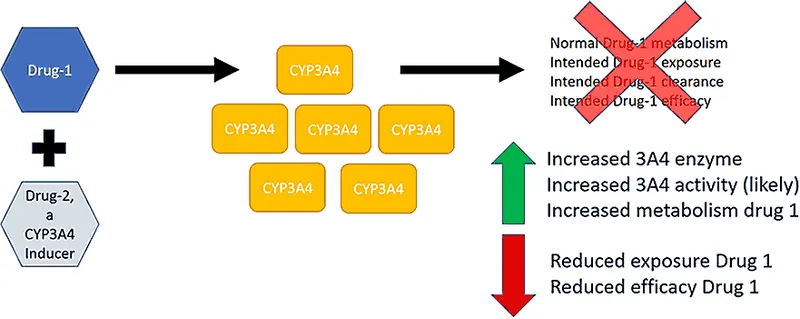
Xenobiotics modulate drug metabolism, primarily of Cytochrome P450 (CYP) enzymes, via induction or inhibition, impacting therapeutic outcomes.
-
Enzyme Induction: ↑ CYP enzyme synthesis by specific xenobiotics.
- Consequences: ↓ Drug efficacy (e.g., Warfarin + Rifampicin), ↑ prodrug activation, ↑ formation of toxic metabolites (e.g., Paracetamol hepatotoxicity with chronic alcohol use).
- Key Inducers: 📌 CRAP GPS: Carbamazepine, Rifampicin, Alcohol (chronic), Phenytoin, Griseofulvin, Phenobarbital, St. John's Wort/Smoking.
-
Enzyme Inhibition: ↓ CYP enzyme activity by specific xenobiotics.
- Consequences: ↑ Drug levels & potential toxicity (e.g., Statins + Macrolides), ↓ prodrug activation (e.g., Clopidogrel + Omeprazole).
- Key Inhibitors: 📌 SICKFACES.COM Group: Sodium valproate, Isoniazid, Cimetidine, Ketoconazole, Fluconazole, Alcohol (acute), Chloramphenicol, Erythromycin, Sulfonamides, Ciprofloxacin, Omeprazole, Metronidazole, Grapefruit juice.
⭐ Grapefruit juice is a classic inhibitor of intestinal CYP3A4, significantly increasing bioavailability and risk of toxicity for numerous common drugs like statins, calcium channel blockers, and certain immunosuppressants.
High‑Yield Points - ⚡ Biggest Takeaways
- Xenobiotic metabolism: Phase I (functionalization) & Phase II (conjugation), mainly in the liver.
- Phase I: Primarily oxidation by Cytochrome P450 (CYP450) enzymes in ER, adds/unmasks polar groups.
- Phase II: Conjugation (e.g., glucuronidation, sulfation, glutathione) greatly ↑ water solubility for excretion.
- Glucuronidation (by UGTs) is crucial for many drugs and bilirubin.
- CYP450 induction/inhibition causes significant drug-drug interactions.
- Genetic polymorphisms (e.g., CYP2D6, NAT2) lead to varied drug responses and toxicity.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

