Electrolyte Essentials - Ion Power Players
Crucial ions maintaining physiological balance. Normal serum values and key roles:
| Electrolyte | Normal Serum Range | Key Function(s) |
|---|---|---|
| Sodium ($Na^+$) | 135-145 mEq/L | Main ECF cation; fluid balance, nerve impulse transmission, muscle contraction |
| Potassium ($K^+$) | 3.5-5.0 mEq/L | Main ICF cation; cardiac function, nerve impulses, muscle activity |
| Chloride ($Cl^-$) | 96-106 mEq/L | Main ECF anion; fluid balance, acid-base balance, gastric $HCl$ production |
| Bicarbonate ($HCO_3^-$) | 22-28 mEq/L | Key blood buffer; regulates acid-base balance (pH) |
| Calcium ($Ca^{2+}$) Total | 8.5-10.5 mg/dL | Bone/teeth formation, muscle contraction, blood clotting, nerve transmission |
| Magnesium ($Mg^{2+}$) | 1.5-2.5 mEq/L | Enzyme cofactor (ATP metabolism), neuromuscular function, cardiac stability |
| Phosphate ($PO_4^{3-}$) | 2.5-4.5 mg/dL | Bone/teeth structure, ATP component, DNA/RNA synthesis, acid-base buffer |
Sodium & Potassium Imbalances - Salty & Shocking Tales
Sodium (Na+) Imbalances:
- Hyponatremia (Na+ < 135 mmol/L; Severe < 125 mmol/L)
- Causes: SIADH, diuretics, GI loss.
- Symptoms: Confusion, seizures.
- Hypernatremia (Na+ > 145 mmol/L)
- Causes: Dehydration, Diabetes Insipidus, ↑Na intake.
- Symptoms: Thirst, restlessness, coma.
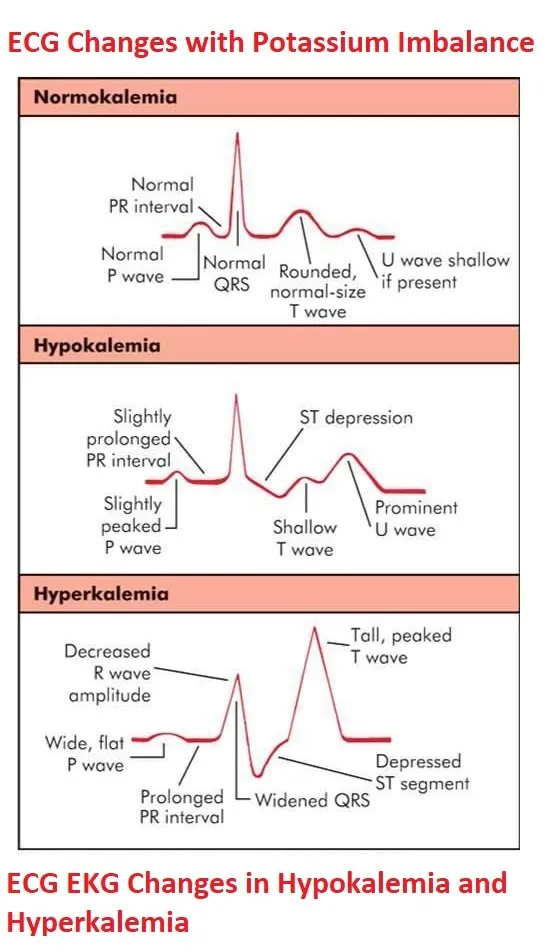
Potassium (K+) Imbalances:
- Hypokalemia (K+ < 3.5 mmol/L)
- Causes: Diuretics, GI loss, insulin.
- Symptoms: Weakness, cramps, U waves.
- ECG: Flat/inverted T, U waves.
- Hyperkalemia (K+ > 5.5 mmol/L)
- Causes: Renal failure, ACE-I, acidosis.
- Symptoms: Weakness, paralysis, arrhythmias.
- ECG: 📌 Peaked T → ↑PR → Wide QRS.
| Condition | Level (mmol/L) | Key Causes | Key Symptoms/Signs | ECG (K+ only) |
|---|---|---|---|---|
| Hyponatremia | Na+ < 135 | SIADH, diuretics, GI loss | Confusion, seizures | N/A |
| Hypernatremia | Na+ > 145 | Dehydration, DI, ↑Na intake | Thirst, restlessness, coma | N/A |
| Hypokalemia | K+ < 3.5 | Diuretics, GI loss, insulin | Weakness, cramps, U waves | Flat/inverted T, U waves |
| Hyperkalemia | K+ > 5.5 | Renal failure, ACE-I, acidosis | Weakness, paralysis, arrhythmias | Peaked T, ↑PR, wide QRS |
Acid-Base Fundamentals - pH Tango Titans
- pH: Negative logarithm of $H^+$ activity. Normal: 7.35-7.45.
- Key Buffers:
- Bicarbonate-carbonic acid: Major ECF buffer.
- Phosphate: ICF & renal tubular buffer.
- Proteins (e.g., Hemoglobin): ICF buffer.
- Organ Regulation:
- Lungs: Regulate $PaCO_2$ (Normal: 35-45 mmHg).
- Kidneys: Regulate $HCO_3^-$ (Normal: 22-26 mEq/L) & $H^+$ excretion.
- Henderson-Hasselbalch: $pH = 6.1 + \log_{10}{\frac{[HCO_3^-]}{0.03 \times PaCO_2}}$
- Anion Gap: $AG = [Na^+] - ([Cl^-] + [HCO_3^-])$. Normal: 8-12 mEq/L.
- Helps differentiate metabolic acidosis types.
- 📌 MUDPILES for ↑AG Metabolic Acidosis.
⭐ The bicarbonate buffer system is the most important extracellular fluid buffer.
ABG Analysis & Disorders - Gaswork Sleuthing Steps
- Key Values: pH 7.35-7.45; $PaCO_2$ 35-45 mmHg; $HCO_3^-$ 22-26 mEq/L; Anion Gap (AG) 8-12 mEq/L.
- 📌 ROME: Respiratory Opposite (pH & $PaCO_2$ direction), Metabolic Equal (pH & $HCO_3^-$ direction).
- Metabolic Acidosis $PaCO_2$ Compensation: Expected $PaCO_2 = (1.5 \times [HCO_3^-]) + 8 \pm 2$.
| Disorder | Primary Δ | Compensatory Δ | Causes (Examples) |
|---|---|---|---|
| Met. Acid. | ↓pH, ↓$HCO_3^-$ | ↓$PaCO_2$ | DKA (↑AG), Diarrhea (NAG) |
| Met. Alk. | ↑pH, ↑$HCO_3^-$ | ↑$PaCO_2$ | Vomiting, Diuretics |
| Resp. Acid. | ↓pH, ↑$PaCO_2$ | ↑$HCO_3^-$ | COPD, Opioids (Hypoventilation) |
| Resp. Alk. | ↑pH, ↓$PaCO_2$ | ↓$HCO_3^-$ | Anxiety, PE (Hyperventilation) |
High‑Yield Points - ⚡ Biggest Takeaways
- Anion gap (AG) = Na⁺ - (Cl⁻ + HCO₃⁻); vital for metabolic acidosis diagnosis. Normal: 8-12 mEq/L.
- Winter's formula (PCO₂ = 1.5 × HCO₃⁻ + 8 ± 2): predicts respiratory compensation in metabolic acidosis.
- Delta gap (ΔAG / ΔHCO₃⁻): identifies mixed acid-base disorders.
- ECG: Hyperkalemia → peaked T waves, wide QRS. Hypokalemia → U waves, flat T waves.
- Correct hyponatremia slowly to prevent osmotic demyelination syndrome (ODS).
- SIADH: common cause of euvolemic hyponatremia.
- Henderson-Hasselbalch equation: relates pH, HCO₃⁻, PCO₂ in acid-base balance_._
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

