Principles of Electrophoresis - Charge & Go!
- Separates charged molecules by differential migration in an electric field ($E$).
- Migration velocity ($v$): $v = \frac{Eq}{f}$
- $E$: Electric field strength
- $q$: Net molecular charge
- $f$: Frictional coefficient (depends on size, shape, medium viscosity)
- Electrophoretic mobility ($\mu$): $\mu = \frac{v}{E} = \frac{q}{f}$. Intrinsic property.
- Key factors influencing speed & direction:
- Net charge ($q$): ↑ magnitude $\rightarrow$ ↑ speed. Sign determines direction.
- Size/Shape (via $f$): ↓ size/more compact $\rightarrow$ ↑ speed.
- Electric field ($E$): ↑ $E$ $\rightarrow$ ↑ speed.
- Support medium: Viscosity, pore size.
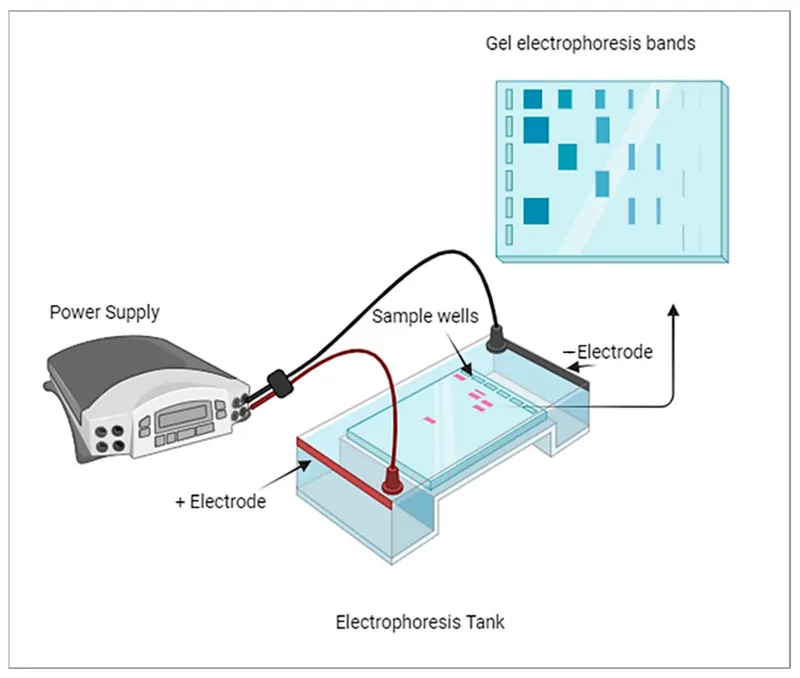 , buffer, support medium with sample wells, and direction of ion movement)
, buffer, support medium with sample wells, and direction of ion movement)
⭐ Anions $\rightarrow$ Anode (+); Cations $\rightarrow$ Cathode (-). 📌 PANIC: Positive Anode, Negative Is Cathode (electrode polarity).
Gel Electrophoresis Types - Matrix Masters
- Agarose Gel Electrophoresis (AGE)
- Agarose matrix (natural); large, adjustable pores (0.5-2% concentration).
- Separates nucleic acids (DNA > 50 bp, RNA) primarily by size.
- Uses: DNA fingerprinting, PCR product analysis, restriction mapping.
- Stains: Ethidium Bromide (EtBr), SYBR Safe.
- 📌 "AGE for large DNA, like sifting sand."
- Polyacrylamide Gel Electrophoresis (PAGE)
- Polyacrylamide matrix (synthetic); small, uniform, adjustable pores (controlled by %T & %C).
- Separates proteins and small DNA/RNA fragments (< 500 bp).
- Types:
- Native PAGE: Separates by charge, size, and shape. Proteins remain active.
- SDS-PAGE: Sodium Dodecyl Sulfate (SDS) denatures proteins, imparts uniform negative (-) charge. Separates by molecular weight. Reducing agents (e.g., β-mercaptoethanol) break disulfide (S-S) bonds.
- Stains: Coomassie Brilliant Blue, Silver stain (for proteins).
⭐ SDS-PAGE is the most common method for determining the molecular weight of proteins.
 oka
oka
Advanced & Blotting Techniques - Focus & Find
-
Advanced Electrophoresis:
- Isoelectric Focusing (IEF): Separates proteins by isoelectric point (pI) using a pH gradient. Proteins migrate until net charge is zero.
- 2D Gel Electrophoresis: Combines IEF (1st dimension: pI) with SDS-PAGE (2nd dimension: mass). High resolution for complex mixtures (e.g., proteomics).
- Capillary Electrophoresis (CE): Automated, high-throughput, high-resolution separation in narrow fused-silica capillaries.
-
Blotting Techniques: Transfer of separated biomolecules (DNA, RNA, or proteins) from a gel to a solid membrane for specific detection.

* **Southern Blot:** Detects specific DNA sequences. Probe: Labeled DNA/RNA.
* **Northern Blot:** Detects specific RNA sequences (gene expression). Probe: Labeled DNA/RNA.
* **Western Blot:** Detects specific proteins. Probe: Labeled antibody.
> ⭐ Western Blot is the confirmatory test for HIV, detecting antibodies to viral proteins (e.g., p24, gp41, gp120/160).
* **Southwestern Blot:** Identifies DNA-binding proteins. Probe: Labeled DNA oligonucleotides.
* **Far-Western Blot:** Detects protein-protein interactions. Probe: Labeled non-antibody protein.
* **Eastern Blot:** Analyzes post-translational modifications (PTMs) of proteins.
* 📌 Mnemonic: **SNoW DRoP** (Southern=DNA, Northern=RNA, Western=Protein).
Clinical Applications - Health Detectives

- Serum Protein Electrophoresis (SPEP):
- Key for monoclonal gammopathies (Multiple Myeloma: M-spike; MGUS).
- Patterns reveal: inflammation (↑ acute phase reactants), nephrotic syndrome (↓ albumin, ↑ α2), cirrhosis (β-γ bridge).
- Hemoglobin Electrophoresis:
- Diagnoses hemoglobinopathies: Sickle cell (HbS), β-thalassemia (↑HbA2/HbF).
- Lipoprotein Electrophoresis:
- Classifies dyslipidemias (e.g., Fredrickson types).
- Isoenzyme Analysis:
- CK-MB for MI; LDH patterns for organ-specific damage.
- Nucleic Acid Electrophoresis:
- Genetic diagnosis (e.g., mutations), viral detection.
⭐ The M-spike on SPEP, a sharp peak usually in the γ-globulin region, is a hallmark of Multiple Myeloma.
High‑Yield Points - ⚡ Biggest Takeaways
- Electrophoresis separates molecules by charge, size, and shape in an electric field.
- SDS-PAGE separates proteins by molecular weight; SDS imparts uniform negative charge.
- Agarose gels are used for DNA/RNA separation; polyacrylamide for proteins.
- Isoelectric focusing (IEF) separates proteins based on their isoelectric point (pI).
- 2D electrophoresis combines IEF and SDS-PAGE for superior protein resolution.
- Key blotting techniques: Southern (DNA), Northern (RNA), Western (Protein) (SNoW DRoP).
- Diagnoses hemoglobinopathies, paraproteinemias, and genetic disorders.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

