Principles of Centrifugation - Spin Basics
- Core Principle: Separates substances based on density, size, and shape using centrifugal force.
- Centrifugal Force: Generated by spinning; expressed as multiples of gravity (g) or Relative Centrifugal Force (RCF).
- RCF Calculation: $RCF = 1.118 \times 10^{-5} \times r \times (RPM)^2$, where 'r' is rotor radius (cm).
- Sedimentation: Particles move based on their properties; denser/larger particles sediment faster.
⭐ Sedimentation Coefficient: Measured in Svedberg units (S); $1S = 10^{-13}$ seconds. Indicates sedimentation velocity per unit of centrifugal field.
Types of Centrifuges & Rotors - Machine Menagerie
Centrifuges:
- Low-Speed (Benchtop): < 10,000 rpm; < 6,000 $g$. Pellets cells, nuclei.
- High-Speed: Up to 25,000 rpm; up to 60,000 $g$. Refrigerated. Pellets organelles, microbes.
- Ultracentrifuge: Up to 80,000 rpm (prep) or 150,000 rpm (analytical); > 600,000 $g$. Refrigerated, vacuum. Isolates viruses, ribosomes, macromolecules.
⭐ Analytical ultracentrifuges determine molecular weight & purity.
Rotors:
- Fixed-Angle: Tubes at 14°-40°. Efficient pelleting. For differential centrifugation.
- Pellet compact, on side/bottom.
- Swinging-Bucket (Horizontal): Tubes swing to 90°. For density gradients (rate-zonal, isopycnic).
- Bands well-resolved; pellet at bottom.
- Vertical: Tubes vertical. Shortest path. For isopycnic DNA separation (e.g., CsCl).
Centrifugation Techniques - Separation Strategies
Separates components by size, shape, density using centrifugal force. Sedimentation rate ($v$) depends on particle properties (e.g., radius $r$, particle density $\rho_p$) and medium properties (e.g., medium density $\rho_m$, viscosity $\eta$).
-
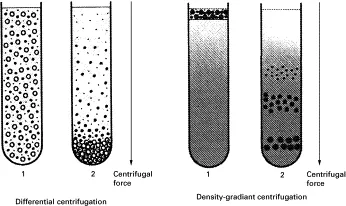
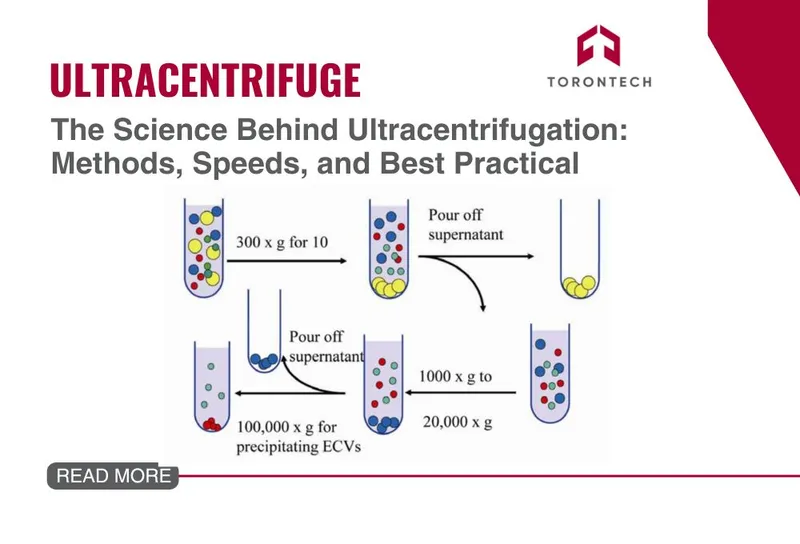
Differential Centrifugation (DC)
- Basis: Different sedimentation rates (primarily size & density).
- Method: Sequential pelleting at increasing g-forces (↑ g-force).
- Yields: Pellets of decreasing size/density (e.g., Nuclei → Mitochondria/Lysosomes → Microsomes → Ribosomes).
⭐ Mnemonic for pelleting order (largest to smallest): "Never Make Little Mice Run" (Nuclei, Mitochondria/Lysosomes, Microsomes, Ribosomes) 📌
-
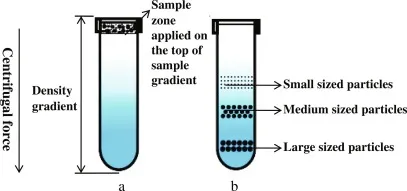
Density Gradient Centrifugation (DGC)
- Uses a density gradient medium (e.g., Sucrose, CsCl, Percoll).
- Rate-Zonal (Size/Shape based):
- Sample layered on pre-formed shallow gradient.
- Separates particles by differences in sedimentation rate through the gradient.
- Particles form distinct zones based on size/shape.
- E.g., Separation of ribosomal subunits (40S, 60S).
- Isopycnic (Density based):
- Particles migrate to a point where their buoyant density equals the gradient density.
- Equilibrium method; separation independent of time once equilibrium is reached.
- E.g., Separation of DNA isoforms (plasmid vs chromosomal), organelles.
💡 Isopycnic: Particles stop when their density matches the gradient's density.
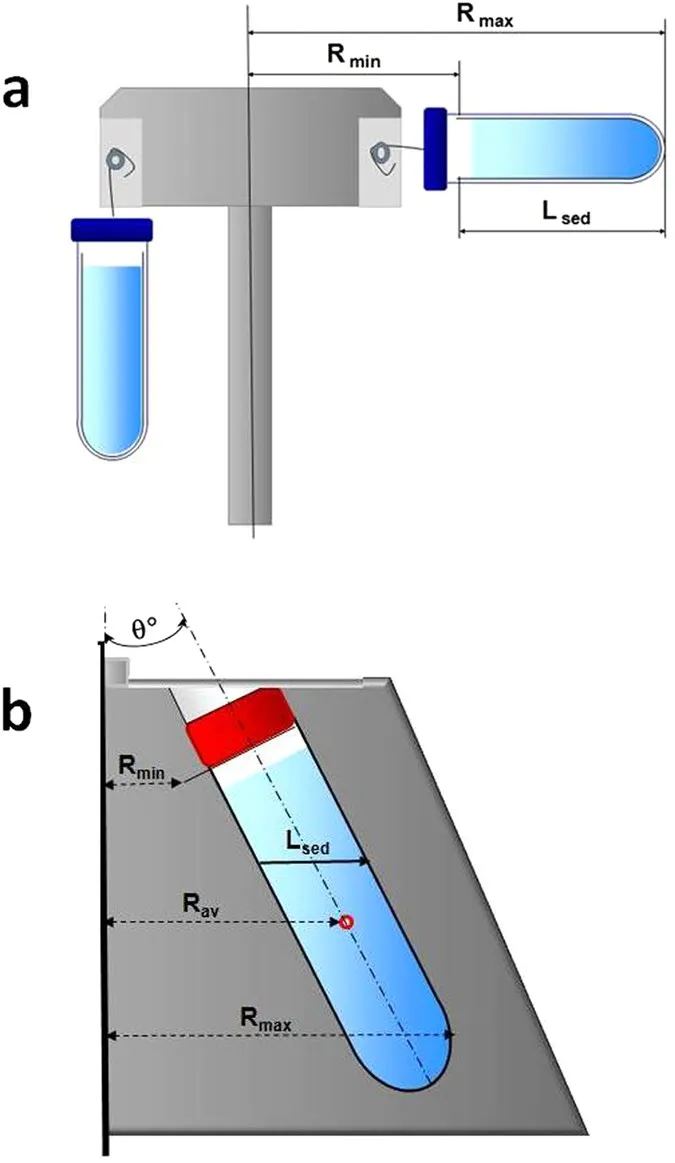
Ultracentrifugation & Applications - Super Spin & Uses
- Achieves very high speeds (>20,000 rpm) & g-forces (>100,000 x g).
- Operates under vacuum & refrigeration to manage heat & air friction.
- Types & Primary Uses:
- Analytical (AUC):
- Determines molecular weight, purity, shape of macromolecules.
- Measures sedimentation coefficient ($S$). Optical detection systems monitor particle movement.
- Preparative:
- Isolates/purifies subcellular organelles (e.g., ribosomes, mitochondria), viruses, macromolecules.
- Used in differential & density gradient (rate-zonal, isopycnic) methods.
- Analytical (AUC):
- Key Applications:
- Lipoprotein fractionation (e.g., HDL, LDL, VLDL).
- Virus isolation & characterization.
- Studying protein-DNA interactions & macromolecular assembly.
- Svedberg unit ($S$): Unit of sedimentation rate; $1 S = 10^{-13}$ seconds.
⭐ Ultracentrifugation is essential for the separation and characterization of plasma lipoproteins (chylomicrons, VLDL, LDL, HDL) based on their differing densities, which is vital in dyslipidemia studies.
High‑Yield Points - ⚡ Biggest Takeaways
- Differential centrifugation separates by size and density through increasing g-forces.
- Rate-zonal centrifugation separates by sedimentation rate (S value), reflecting mass and shape.
- Isopycnic centrifugation separates particles based solely on their buoyant density.
- Svedberg unit (S) measures sedimentation rate, not directly molecular weight.
- Ultracentrifugation employs very high g-forces for separating macromolecules and organelles.
- Analytical ultracentrifugation determines molecular weight, shape, and purity of macromolecules.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more