Electrolyte Essentials - Spark of Life
- Electrolytes: Charged ions vital for nerve/muscle function, fluid balance, pH.
- Major Cations:
- $Na^{+}$: 135-145 mEq/L (ECF osmolality, nerve impulses)
- $K^{+}$: 3.5-5.0 mEq/L (ICF osmolality, cardiac/muscle function)
- $Ca^{2+}$: 8.5-10.5 mg/dL (bones, muscle contraction, clotting)
- $Mg^{2+}$: 1.5-2.5 mEq/L (enzyme cofactor, neuromuscular function)
- Major Anions:
- $Cl^{-}$: 98-106 mEq/L (ECF osmolality, acid-base)
- $HCO_3^{-}$: 22-28 mEq/L (acid-base buffer)
- $PO_4^{3-}$: 2.5-4.5 mg/dL (ICF anion, bone, ATP)
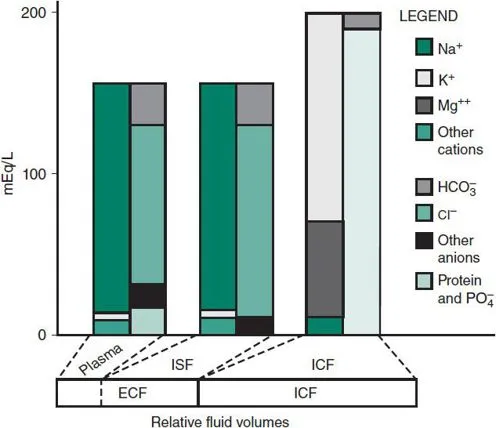 vs extracellular fluid (ECF))
vs extracellular fluid (ECF))
⭐ K+ is the major intracellular cation; Na+ is the major extracellular cation.
Sodium & Water Balance - Fluid Symphony
- Sodium (Na+): Major ECF cation; normal 135-145 mEq/L. Drives plasma osmolality & ECF volume.
- Water Balance: Governed by ADH (from posterior pituitary) & thirst mechanism.
- Regulation:
- RAAS (Aldosterone): ↑Na+ & H₂O reabsorption (distal nephron).
- ADH (Vasopressin): ↑H₂O reabsorption (collecting ducts via aquaporin-2). 📌 'Adds Da H₂O'.
- Natriuretic Peptides (ANP, BNP): Promote natriuresis & diuresis; counter RAAS.
- Plasma Osmolality: $2 \times [Na^+]{pl} + \frac{[Glucose]{mg/dL}}{18} + \frac{[BUN]_{mg/dL}}{2.8}$. Normal: 275-295 mOsm/kg H₂O.
⭐ Rapid correction of chronic hyponatremia (e.g., <120 mEq/L for >48h) risks Osmotic Demyelination Syndrome (ODS).
Potassium Power - Cardiac Conductor
-
Major intracellular cation; Normal: 3.5-5.0 mEq/L.
-
Crucial for neuromuscular excitability & cardiac function (resting membrane potential).
-
Regulation: Kidneys (aldosterone), transcellular shifts (insulin, pH).
-
Hypokalemia (<3.5 mEq/L):
- Causes: ↓ intake, ↑ loss (diuretics, GI), shifts (alkalosis, insulin).
- ECG: Flat/inverted T, U waves, ST depression. 📌 Hypo-K: LOW & SLOW.
- Symptoms: Weakness, cramps, ileus, arrhythmias.
-
Hyperkalemia (>5.0 mEq/L):
- Causes: ↓ excretion (renal failure), shifts (acidosis, cell lysis), ↑ intake.
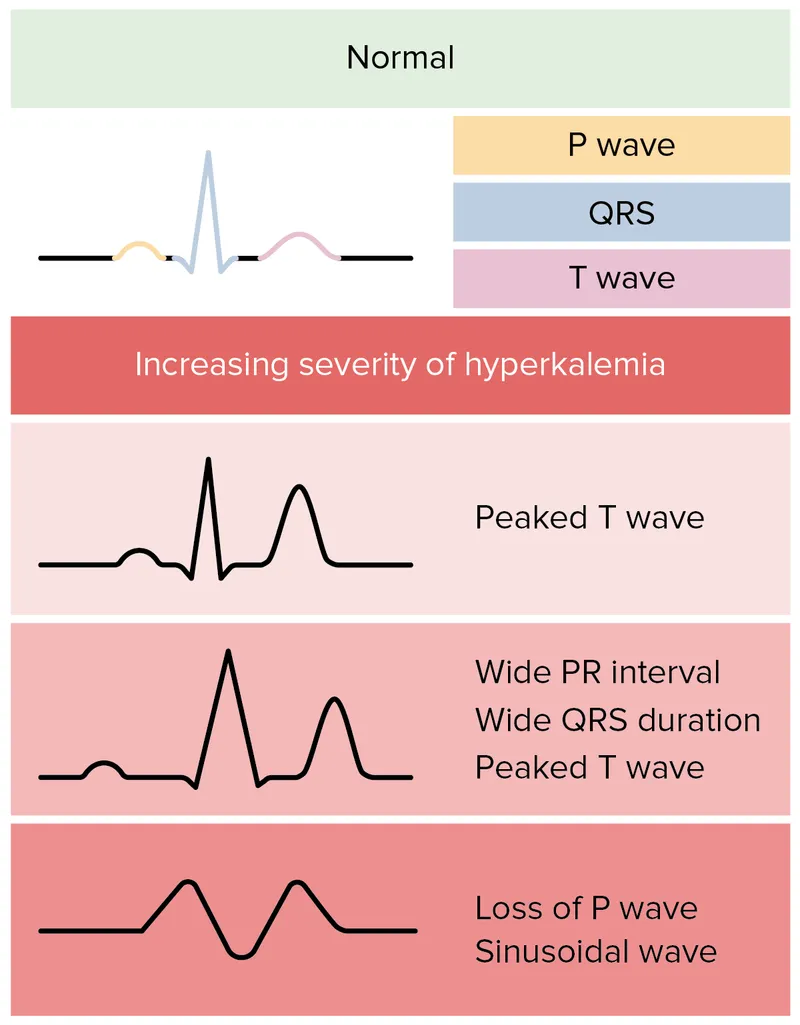
- ECG: Peaked T, wide QRS, sine wave. 📌 MURDER.
- Symptoms: Weakness, paralysis, arrhythmias.
⭐ ECG changes are often the earliest and most life-threatening manifestations of potassium imbalance.
Calcium, Phosphate & Magnesium - Mineral Trinity
- Calcium ($Ca^{2+}$): Total 8.5-10.5 mg/dL; Ionized 4.5-5.6 mg/dL.
- Roles: Bone, muscle, nerve, clotting.
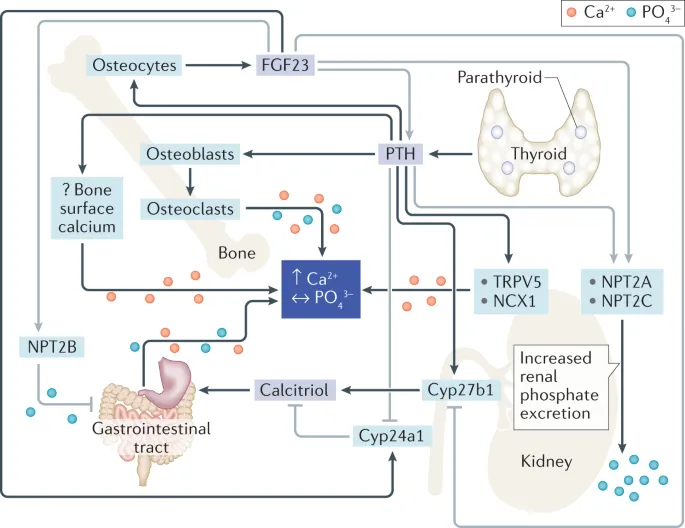
- Control: PTH (↑$Ca^{2+}$), Vit D (↑absorption), Calcitonin (↓$Ca^{2+}$).
- ↓$Ca^{2+}$: Tetany (Chvostek/Trousseau), long QT.
- ↑$Ca^{2+}$: "Bones, stones, groans, thrones, psych overtones"; short QT.
- Phosphate ($PO_4^{3-}$): Serum 2.5-4.5 mg/dL.
- Roles: Bone, ATP, DNA/RNA, buffer.
- Control: PTH (↓renal reabsorption), Vit D (↑absorption).
- Magnesium ($Mg^{2+}$): Serum 1.7-2.2 mg/dL.
- Roles: Enzyme cofactor (ATPases), K⁺/Ca²⁺ channels, neuromuscular.
- ↓$Mg^{2+}$: Tetany, Torsades; causes refractory ↓K⁺, ↓$Ca^{2+}$.
- ↑$Mg^{2+}$: ↓DTRs, respiratory depression.
⭐ Hypomagnesemia causes refractory hypokalemia (renal K⁺ wasting) & hypocalcemia (impaired PTH).
High‑Yield Points - ⚡ Biggest Takeaways
- Na+: Main ECF cation; dictates volume & osmolality. Regulated by RAAS, ADH, ANP.
- K+: Main ICF cation; critical for cardiac/nerve function. Insulin, alkalosis shift K+ into cells.
- Ca2+: Regulated by PTH (↑), Vit D (↑ absorption), Calcitonin (↓). Ionized Ca2+ is active.
- Mg2+: Essential cofactor; vital for neuromuscular excitability, cardiac rhythm. Parallels K+, Ca2+.
- PO43-: Key for ATP, bone. Reciprocal to Ca2+. Regulated by PTH, Vit D, FGF-23.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

