Postoperative Care

On this page
🎯 The PACU Command Center: Operational Excellence Framework
The Post-Anesthesia Care Unit functions as the critical interface between operating room control and ward autonomy. Understanding PACU operations requires mastering admission protocols, monitoring standards, staffing requirements, and discharge systems that determine patient safety during the vulnerable emergence period.
PACU admission triggers a standardized assessment cascade within the first 5 minutes. The anesthesiologist provides structured handoff covering 7 critical domains: surgical procedure and duration, anesthetic technique and agents used, estimated blood loss and fluid replacement, intraoperative complications, current vital signs and ventilatory status, pain management plan, and special monitoring requirements. This handoff reduces communication errors by 65% compared to unstructured sign-outs.
PACU Staffing and Physical Standards
📌 Remember: PACU 1:1:2 - One nurse per patient in Phase I recovery, one nurse per two patients in Phase II, maintaining 1.5× bed capacity relative to operating rooms served
Modern PACU design separates Phase I (immediate recovery) from Phase II (preparation for discharge) zones. Phase I requires 120 square feet per bed with direct visualization of all patients from the central nursing station. Each bed space must include:
- Monitoring equipment: Continuous ECG, pulse oximetry, non-invasive blood pressure (NIBP) cycling every 5 minutes initially
- Capnography for patients with artificial airways or receiving supplemental oxygen >4 L/min
- Temperature monitoring capability with warming devices
- Invasive pressure monitoring connections for arterial lines and central venous catheters
- Emergency access: Oxygen delivery system providing 0-15 L/min, suction apparatus with 120 mmHg negative pressure capacity
- Difficult airway cart within 30 seconds access time
- Defibrillator and emergency medications visible and reachable
- Documentation systems: Real-time vital signs trending, medication administration records, pain scores every 15 minutes
⭐ Clinical Pearl: PACU nurse-to-patient ratios directly impact complication detection-1:1 staffing catches respiratory depression 85% faster than 1:2 ratios, with 40% reduction in critical interventions required
The Modified Aldrete Scoring System
The Aldrete score quantifies recovery readiness across 5 physiological domains, each scored 0-2 points for a maximum of 10 points. Discharge from Phase I requires a score ≥9 with no individual category scoring zero.
| Domain | 2 Points | 1 Point | 0 Points | Clinical Significance |
|---|---|---|---|---|
| Activity | Moves 4 extremities voluntarily | Moves 2 extremities | No movement | Neuromuscular recovery, residual blockade detection |
| Respiration | Deep breathing, effective cough | Dyspnea, shallow breathing | Apneic | Respiratory drive restoration, airway patency |
| Circulation | BP ±20% of baseline | BP ±20-50% of baseline | BP ±50% of baseline | Hemodynamic stability, fluid status |
| Consciousness | Fully awake, oriented | Arousable to voice | Unresponsive | Emergence completion, cognitive recovery |
| Oxygen Saturation | SpO2 >92% on room air | Supplemental O2 needed for SpO2 >90% | SpO2 <90% despite O2 | Pulmonary gas exchange adequacy |
💡 Master This: The Aldrete score identifies 95% of patients safe for Phase II transfer but misses subtle cognitive deficits-always combine objective scoring with clinical gestalt assessment of patient interaction quality and pain control adequacy
Fast-Track Recovery Integration
Fast-track protocols bypass traditional Phase I recovery for selected patients, reducing PACU time by 45-60 minutes and increasing operating room utilization by 15-20%. Eligibility requires meeting strict criteria at operating room emergence:
- Hemodynamic stability: Heart rate 60-100 bpm, blood pressure within 20% of baseline, no active bleeding
- Respiratory adequacy: Spontaneous ventilation with tidal volume >5 mL/kg, SpO2 >95% on minimal supplemental oxygen
- Neurological recovery: Awake, oriented, following commands, no residual neuromuscular blockade (train-of-four ratio >0.9)
- Pain control: Numeric rating scale <4/10 with established multimodal analgesia
- Absence of complications: No nausea/vomiting, normothermic (>36°C), stable glucose if diabetic
⭐ Clinical Pearl: Fast-track candidates represent 30-40% of ambulatory surgery patients but require 50% more intraoperative attention to anesthetic depth, multimodal analgesia, and PONV prophylaxis-the operating room work enables PACU efficiency
PACU Monitoring Intensity and Duration
Standard Phase I monitoring continues until Aldrete score ≥9 is sustained for 30 minutes, typically requiring 60-90 minutes for general anesthesia, 45-60 minutes for regional techniques, and 30-45 minutes for monitored anesthesia care. High-risk patients require extended monitoring:
- Cardiac surgery: Minimum 2-4 hours with continuous arterial pressure, central venous pressure, cardiac output monitoring
- Chest tube output assessment every 15 minutes, cumulative drainage <200 mL/hour
- 12-lead ECG for arrhythmia detection, troponin if ischemia suspected
- Thoracic surgery: Continuous pulse oximetry and capnography until chest tube drainage stable
- Incentive spirometry every 1 hour, forced vital capacity >10 mL/kg before discharge
- Neurological procedures: Hourly neurological assessments using standardized scales
- Immediate CT imaging for any deterioration in consciousness or new focal deficits
💡 Master This: PACU length of stay directly correlates with intraoperative anesthetic management-every 30 minutes of intraoperative volatile anesthetic beyond 2 hours adds 10-15 minutes to emergence time, while total intravenous anesthesia with propofol-remifentanil reduces PACU stay by 20-25%
Complication Surveillance Protocols
Systematic complication screening occurs every 15 minutes during Phase I recovery, targeting the 5 most frequent PACU emergencies that account for 80% of critical interventions:
- Respiratory depression: Respiratory rate <8 breaths/minute, SpO2 <90%, somnolence score ≥3
- Immediate naloxone 0.04-0.08 mg IV titrated to effect, avoiding complete opioid reversal
- Flumazenil 0.2 mg IV every 60 seconds (maximum 1 mg) for benzodiazepine-related depression
- Hypotension: Systolic BP <90 mmHg or >30% below baseline for >5 minutes
- Fluid bolus 250-500 mL crystalloid over 10-15 minutes, vasopressor initiation if unresponsive
- Echocardiography for persistent hypotension to assess cardiac function and volume status
- Hypertension: Systolic BP >180 mmHg or >30% above baseline
- Pain assessment and treatment first-inadequate analgesia causes 60% of PACU hypertension
- Labetalol 5-20 mg IV or hydralazine 5-10 mg IV for persistent elevation
- Emergence delirium: Agitation, disorientation, combativeness within 30 minutes of emergence
- Exclude hypoxia, hypoglycemia, bladder distention, pain before pharmacological intervention
- Propofol 10-20 mg IV or dexmedetomidine 0.5-1 mcg/kg over 10 minutes for severe agitation
Understanding PACU operational standards establishes the foundation for recognizing and managing specific postoperative complications. Connect this structural knowledge through to master the clinical syndromes that challenge recovery room providers.
🎯 The PACU Command Center: Operational Excellence Framework
🫁 Respiratory Vigilance: The Oxygen Delivery Challenge
Respiratory complications represent 30-40% of PACU critical events and account for 50% of anesthesia-related mortality in the immediate postoperative period. Mastering respiratory assessment and intervention separates routine recovery from life-threatening crisis management.
Postoperative respiratory physiology undergoes predictable deterioration from baseline. Functional residual capacity (FRC) decreases by 15-20% after general anesthesia and 50-70% after upper abdominal or thoracic surgery, persisting for 7-10 days. This FRC reduction causes small airway closure, ventilation-perfusion mismatch, and increased shunt fraction-the primary mechanism of postoperative hypoxemia affecting 30-50% of patients.
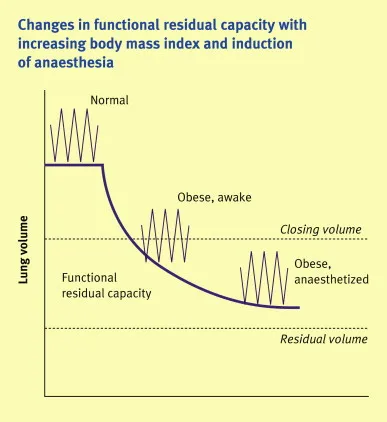
Hypoxemia Recognition and Grading
📌 Remember: HYPOXEMIA 5-4-3-2-1 - Room air SpO2 <95%, PaO2 <80 mmHg mild; <60 mmHg moderate; <50 mmHg severe; <40 mmHg critical requiring immediate intubation
Pulse oximetry provides continuous oxygenation monitoring but requires understanding its limitations. SpO2 accuracy deteriorates at saturations <80%, with ±4% variance. Methemoglobinemia, carboxyhemoglobinemia, severe anemia (<5 g/dL), and peripheral vasoconstriction cause false readings. Arterial blood gas analysis remains the gold standard when SpO2 values conflict with clinical assessment.
The alveolar gas equation predicts expected PaO2 and identifies the mechanism of hypoxemia:
$$PAO_2 = FiO_2 × (P_{atm} - P_{H_2O}) - \frac{PaCO_2}{RQ}$$
At sea level breathing room air: PAO2 = 0.21 × (760 - 47) - 40/0.8 = 100 mmHg
The A-a gradient (PAO2 - PaO2) normally increases with age: 2.5 + 0.21 × age in mmHg. Widened A-a gradient (>20 mmHg in young adults) indicates ventilation-perfusion mismatch, shunt, or diffusion impairment-the hallmarks of postoperative atelectasis and pneumonia.
Hypoxemia Treatment Algorithm
⭐ Clinical Pearl: Supplemental oxygen at 2-4 L/min via nasal cannula increases FiO2 to approximately 28-36%, sufficient for 85% of postoperative hypoxemia cases-higher flows rarely benefit patients with adequate respiratory drive and airway patency
Airway Obstruction: The Silent Killer
Upper airway obstruction causes 15-20% of PACU respiratory emergencies, with peak incidence 10-30 minutes post-extubation when residual anesthetic effects persist. Obstruction severity ranges from subtle increased work of breathing to complete apnea with rapid desaturation.
Clinical recognition requires vigilant observation:
- Mild obstruction: Snoring, use of accessory muscles, paradoxical chest-abdomen movement
- SpO2 maintained >92% with supplemental oxygen
- Respiratory rate 20-25 breaths/minute with prolonged inspiratory phase
- Moderate obstruction: Suprasternal and intercostal retractions, tracheal tug, agitation
- SpO2 85-92% despite high-flow oxygen
- Respiratory rate >25 breaths/minute or decreasing with fatigue
- Severe obstruction: Minimal air movement, silent chest, cyanosis, bradycardia
- SpO2 <85% and falling rapidly
- Altered consciousness progressing to unresponsiveness
💡 Master This: The "rocking boat" sign-paradoxical inward chest movement during inspiration with outward abdominal movement-indicates severe upper airway obstruction requiring immediate intervention; this pattern reflects negative intrathoracic pressure generation against a closed glottis
| Intervention | Indication | Technique | Success Rate | Time to Effect |
|---|---|---|---|---|
| Jaw thrust | First-line for all obstruction | Lift mandible anteriorly at angles | 70% for tongue-based | Immediate |
| Oral airway | Persistent obstruction, sedated | Size 80-100 mm, insert upside-down then rotate | 85% combined with jaw thrust | <30 seconds |
| Nasal airway | Oral airway not tolerated | Lubricate, insert perpendicular to face | 75% for nasopharyngeal collapse | <30 seconds |
| CPAP mask | Partial obstruction, cooperative | 5-10 cm H2O pressure | 90% for soft tissue obstruction | 2-5 minutes |
| Laryngeal mask | Failed simple maneuvers | Size based on weight, inflate to seal | 95% rescue ventilation | 1-2 minutes |
Residual Neuromuscular Blockade
Incomplete reversal of neuromuscular blocking agents causes 40-50% of postoperative respiratory complications, with residual blockade present in 30-60% of patients receiving intermediate-acting relaxants despite apparent clinical recovery. Train-of-four (TOF) ratio <0.9 impairs pharyngeal coordination, reduces hypoxic ventilatory response by 50%, and increases aspiration risk 5-fold.
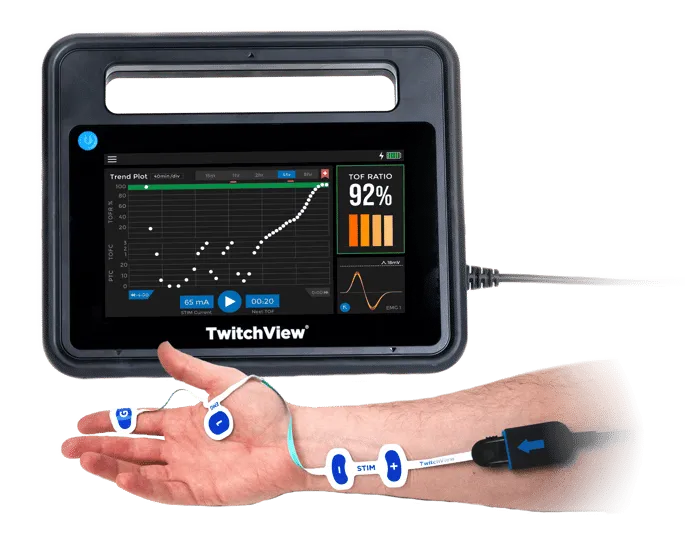
Quantitative neuromuscular monitoring provides objective assessment:
- TOF ratio 0.4-0.6: Adequate hand grip strength, sustained head lift <3 seconds, diplopia, dysphagia
- TOF ratio 0.6-0.7: Sustained head lift 3-5 seconds, normal vital capacity, impaired pharyngeal function
- TOF ratio 0.7-0.9: Sustained head lift >5 seconds, reduced FEV1 by 20%, subtle coordination deficits
- TOF ratio >0.9: Full recovery of respiratory muscle strength and airway protective reflexes
⭐ Clinical Pearl: Clinical tests (sustained head lift, hand grip) detect TOF ratios <0.4 but miss clinically significant residual blockade at 0.4-0.9-quantitative monitoring identifies 3-fold more patients requiring additional reversal than clinical assessment alone
Sugammadex revolutionized neuromuscular blockade reversal, providing dose-dependent antagonism within 2-3 minutes:
- Routine reversal (TOF count 2-4): 2 mg/kg achieves TOF >0.9 in 95% of patients
- Deep blockade (TOF count 1-2): 4 mg/kg required for adequate reversal
- Immediate reversal (3 minutes post-rocuronium): 16 mg/kg enables rapid sequence reversal
Neostigmine remains cost-effective for routine reversal but requires 10-15 minutes for peak effect and fails when TOF count <2. Maximum dose 5 mg (or 0.07 mg/kg) with glycopyrrolate 0.01 mg/kg or atropine 0.02 mg/kg to prevent muscarinic side effects.
Postoperative Pulmonary Complications Risk Stratification
The ARISCAT score predicts postoperative respiratory failure risk using 7 preoperative and intraoperative factors, validated across >2,000 patients with area under curve 0.89:
- Age: <50 years (0 points), 51-80 years (3 points), >80 years (16 points)
- Preoperative SpO2: ≥96% (0 points), 91-95% (8 points), ≤90% (24 points)
- Respiratory infection in last month: No (0 points), Yes (17 points)
- Preoperative anemia: Hb ≥10 g/dL (0 points), Hb <10 g/dL (11 points)
- Surgical incision: Peripheral (0 points), upper abdominal (15 points), intrathoracic (24 points)
- Duration of surgery: <2 hours (0 points), 2-3 hours (16 points), >3 hours (23 points)
- Emergency procedure: No (0 points), Yes (8 points)
Risk categories guide PACU management intensity:
- Low risk (<26 points, 1.6% complication rate): Standard monitoring, early mobilization
- Intermediate risk (26-44 points, 13.3% complication rate): Extended PACU stay, incentive spirometry, aggressive pain control
- High risk (≥45 points, 42.1% complication rate): Consider ICU admission, non-invasive ventilation, chest physiotherapy
💡 Master This: High-risk patients benefit from protective ventilation strategies continued into the PACU-tidal volume 6-8 mL/kg ideal body weight, PEEP 5-8 cm H2O, recruitment maneuvers every 30 minutes-reducing postoperative pulmonary complications by 30-40% compared to standard care
Respiratory mastery enables hemodynamic optimization. Connect these airway and ventilation principles through to build comprehensive pulmonary management expertise, then advance to cardiovascular stability assessment.
🫁 Respiratory Vigilance: The Oxygen Delivery Challenge
💉 Hemodynamic Mastery: The Circulation Control Matrix
Cardiovascular instability affects 20-30% of PACU patients, ranging from benign blood pressure fluctuations to life-threatening shock syndromes requiring immediate intervention. Systematic assessment distinguishes physiological emergence responses from pathological deterioration.
Normal postoperative hemodynamics reflect competing influences: sympathetic activation from pain and emergence increases heart rate by 15-30% and blood pressure by 20-40% above baseline, while residual anesthetic vasodilation, hypovolemia, and myocardial depression exert opposing effects. Blood pressure within ±20% of preoperative baseline represents acceptable stability, with heart rate 60-100 bpm and adequate tissue perfusion markers.
Hypotension: The Perfusion Crisis
Postoperative hypotension (systolic BP <90 mmHg or >30% below baseline for >5 minutes) demands rapid etiology identification and targeted treatment. The 5 primary mechanisms account for 95% of PACU hypotension cases:
📌 Remember: HYPOTENSION 5 H's - Hypovolemia (40%), Heart failure (20%), Hypoxia (15%), Hypothermia (10%), Histamine/anaphylaxis (5%)-percentages reflect relative frequency in general PACU population
Hypovolemia causes 40% of postoperative hypotension, resulting from inadequate intraoperative fluid replacement, ongoing bleeding, or third-space losses. Clinical signs include:
- Hemodynamic patterns: Narrow pulse pressure (<25 mmHg), tachycardia (>100 bpm), orthostatic changes
- Physical examination: Dry mucous membranes, decreased skin turgor, flat neck veins, cool extremities
- Advanced monitoring: Central venous pressure <5 mmHg, stroke volume variation >13%, passive leg raise test positive (>10% cardiac output increase)
Fluid resuscitation follows goal-directed principles:
- Initial bolus: Crystalloid 500 mL over 10-15 minutes (or 250 mL colloid)
- Response assessment: Repeat blood pressure and perfusion markers after each bolus
- Endpoint: Mean arterial pressure >65 mmHg, heart rate <100 bpm, urine output >0.5 mL/kg/hour
- Caution: Limit crystalloid to 30-40 mL/kg in first 24 hours to avoid fluid overload complications
⭐ Clinical Pearl: Fluid bolus responsiveness decreases with successive administrations-50% of patients respond to first bolus, 30% to second, 15% to third-persistent hypotension after 1,500 mL crystalloid indicates need for vasopressor or inotropic support rather than continued volume loading
Cardiac dysfunction accounts for 20% of hypotension cases, encompassing myocardial ischemia, arrhythmias, heart failure exacerbation, and valvular abnormalities. Echocardiography provides rapid bedside assessment:
- Left ventricular systolic function: Ejection fraction <40% indicates need for inotropic support
- Right ventricular function: Dilation and dysfunction suggest pulmonary embolism or acute cor pulmonale
- Valvular pathology: Severe stenosis or regurgitation may decompensate under anesthetic stress
- Pericardial effusion: Tamponade physiology requires immediate drainage
| Vasopressor/Inotrope | Mechanism | Dose Range | Primary Indication | Onset Time | Major Side Effect |
|---|---|---|---|---|---|
| Phenylephrine | Pure α1-agonist | 50-200 mcg IV bolus; 0.5-3 mcg/kg/min infusion | Vasodilation with normal cardiac function | 1-2 minutes | Reflex bradycardia, decreased CO |
| Ephedrine | Mixed α/β-agonist | 5-10 mg IV bolus | Hypotension with bradycardia | 2-3 minutes | Tachycardia, tachyphylaxis |
| Norepinephrine | α1 + β1-agonist | 0.05-0.5 mcg/kg/min | Septic shock, severe vasodilation | 1-2 minutes | Arrhythmias, tissue ischemia |
| Epinephrine | α + β-agonist | 5-20 mcg IV bolus; 0.05-0.5 mcg/kg/min | Anaphylaxis, cardiac arrest, severe shock | <1 minute | Tachycardia, myocardial ischemia |
| Dobutamine | β1-agonist | 2.5-20 mcg/kg/min | Low cardiac output with adequate BP | 5-10 minutes | Tachycardia, hypotension |
Hypertension: The Pressure Paradox
Postoperative hypertension (systolic BP >180 mmHg or >30% above baseline) occurs in 15-20% of PACU patients, with peak incidence 15-45 minutes post-emergence. Untreated severe hypertension increases risk of myocardial ischemia, cerebral hemorrhage, surgical site bleeding, and wound dehiscence.
Pain represents the most common cause, accounting for 60% of hypertensive episodes. Systematic assessment prioritizes:
- Pain evaluation: Numeric rating scale, behavioral indicators in non-communicative patients
- Adequate analgesia resolves hypertension in 70-80% of cases within 15-30 minutes
- Opioid titration: Fentanyl 25-50 mcg IV every 5-10 minutes or morphine 2-4 mg IV every 10-15 minutes
- Bladder distention: Palpable suprapubic mass, agitation, inability to void
- Bladder volumes >400-500 mL trigger hypertensive response
- Catheterization provides immediate relief and diagnostic confirmation
- Emergence delirium: Agitation, confusion, combativeness masking underlying discomfort
- Exclude hypoxia, hypoglycemia, neurological events before attributing to delirium
💡 Master This: Hypertension with bradycardia suggests Cushing reflex from increased intracranial pressure-immediate neurological assessment and imaging required; hypertension with tachycardia indicates pain, anxiety, or sympathetic surge from inadequate anesthetic depth during emergence
Pharmacological management targets mean arterial pressure reduction by 20-25% over 30-60 minutes, avoiding precipitous drops that compromise cerebral or coronary perfusion:
- Labetalol: Combined α/β-blockade, dose 5-20 mg IV every 10 minutes (maximum 300 mg)
- Reduces BP by 10-15% within 5-10 minutes, duration 3-6 hours
- Contraindicated in asthma, heart block, severe heart failure
- Hydralazine: Direct arteriolar vasodilator, dose 5-10 mg IV every 20 minutes
- Onset 10-20 minutes, duration 2-4 hours, unpredictable response
- Reflex tachycardia limits use in coronary artery disease
- Nicardipine: Calcium channel blocker, infusion 5-15 mg/hour
- Precise titration, cerebral vasodilation benefits neurological patients
- Preferred for hypertensive emergencies requiring controlled reduction
Arrhythmia Recognition and Management
Cardiac arrhythmias complicate 10-15% of PACU admissions, ranging from benign sinus tachycardia to life-threatening ventricular fibrillation. Continuous ECG monitoring detects rhythm disturbances, but clinical correlation determines intervention urgency.
Sinus tachycardia (heart rate >100 bpm with normal P-wave morphology) represents a physiological response in 80% of cases:
- Common triggers: Pain (40%), hypovolemia (25%), hypoxia (15%), fever (10%), anxiety (10%)
- Treatment: Address underlying cause rather than rate control
- Concerning features: Heart rate >130 bpm, chest pain, hypotension, altered consciousness
Atrial fibrillation occurs in 5-10% of postoperative patients, with higher incidence (30-40%) after cardiac or thoracic surgery. New-onset AF within 72 hours post-surgery typically resolves spontaneously but increases stroke risk 5-fold and mortality 2-fold:
- Hemodynamically stable: Rate control with metoprolol 2.5-5 mg IV every 5 minutes (target <110 bpm)
- Diltiazem 0.25 mg/kg IV over 2 minutes if β-blockers contraindicated
- Consider anticoagulation if AF persists >48 hours and CHA2DS2-VASc score ≥2
- Hemodynamically unstable: Synchronized cardioversion starting at 120-200 J biphasic
- Amiodarone 150 mg IV over 10 minutes if cardioversion delayed or unsuccessful
⭐ Clinical Pearl: Postoperative AF often reflects underlying electrolyte disturbances-60% of patients have hypokalemia (<3.5 mEq/L) or hypomagnesemia (<1.8 mg/dL)-aggressive repletion (potassium >4.5 mEq/L, magnesium >2 mg/dL) reduces recurrence by 40-50%
Ventricular arrhythmias demand immediate assessment and treatment. Premature ventricular contractions (PVCs) occur commonly but frequent PVCs (>6/minute), multifocal PVCs, R-on-T phenomenon, or runs of ventricular tachycardia indicate myocardial irritability requiring intervention:
- Exclude reversible causes: Hypoxia, hypercarbia, electrolyte abnormalities, myocardial ischemia
- Pharmacological suppression: Amiodarone 150 mg IV over 10 minutes, then 1 mg/min for 6 hours
- Lidocaine 1-1.5 mg/kg IV bolus alternative if amiodarone unavailable
- Sustained ventricular tachycardia: Immediate synchronized cardioversion if pulseless or unstable
- Amiodarone or lidocaine for stable VT with pulse and adequate perfusion
Hemodynamic expertise enables recognition of fluid and electrolyte disturbances. Connect cardiovascular principles through to master volume optimization, then progress to temperature regulation challenges.
💉 Hemodynamic Mastery: The Circulation Control Matrix
🌡️ Thermal Control: The Temperature Regulation Imperative
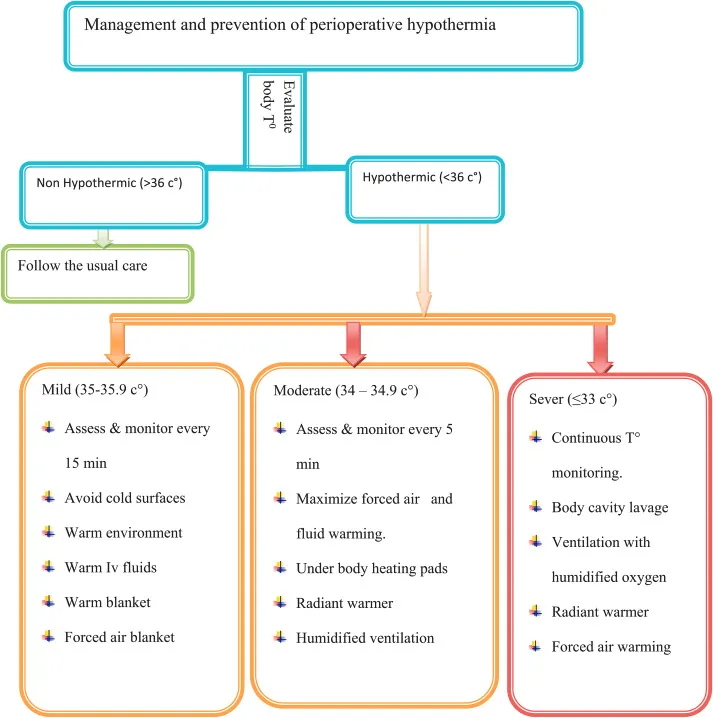
Perioperative hypothermia (core temperature <36°C) affects 50-70% of surgical patients, with peak incidence during the first hour of PACU admission. Understanding thermoregulation physiology and implementing aggressive rewarming protocols prevents the cascade of complications that triple adverse outcomes.
Anesthesia-induced thermoregulation impairment causes 3-5°C core temperature decrease through three mechanisms accounting for 90% of heat loss:
- Redistribution (first 30-60 minutes): Vasodilation transfers heat from core to periphery, causing 0.5-1.5°C drop
- Accounts for 81% of intraoperative heat loss
- Prevented by prewarming 30 minutes before induction
- Radiation (ongoing throughout surgery): Heat transfer to cooler environment, 60% of total heat loss
- Operating room temperature 21-24°C creates 2-4°C gradient
- Reduced by increasing ambient temperature to 23-26°C
- Convection and evaporation: Exposed surgical sites, cold IV fluids, respiratory heat loss
- Each liter of room temperature crystalloid decreases core temperature by 0.25°C
- Warming fluids to 37-40°C prevents iatrogenic cooling
Hypothermia Consequences and Quantification
📌 Remember: HYPOTHERMIA TRIAD - Coagulopathy (platelet dysfunction at <35°C, clotting factor activity reduced 10% per 1°C drop), Cardiac complications (300% increased MI risk), Infection (surgical site infection rate 200-300% higher)
Temperature measurement site selection impacts accuracy and clinical utility:
| Site | Accuracy | Advantages | Disadvantages | Clinical Use |
|---|---|---|---|---|
| Pulmonary artery | Gold standard ±0.1°C | True core temperature, continuous | Requires PA catheter, invasive | Cardiac surgery, ICU |
| Esophageal | ±0.2°C | Reflects core, easy placement | Requires intubation, position-dependent | Intraoperative, intubated patients |
| Nasopharyngeal | ±0.3°C | Core approximation, simple | Contraindicated in skull base fractures | Awake or intubated patients |
| Bladder | ±0.3°C | Continuous core monitoring | Requires catheter, affected by UOP | Extended PACU stay, ICU |
| Tympanic | ±0.5°C | Non-invasive, rapid | User-dependent, cerumen interference | PACU screening |
| Axillary/oral | ±1.0°C | Non-invasive, familiar | Peripheral, influenced by environment | Unreliable for clinical decisions |
Hypothermia severity stratification guides rewarming intensity:
- Mild (35-36°C): Shivering, vasoconstriction, increased oxygen consumption by 200-400%
- Forced-air warming at 43°C, warmed IV fluids, increased ambient temperature
- Rewarming rate 0.5-1°C/hour, typically 1-2 hours to normothermia
- Moderate (32-35°C): Decreased consciousness, arrhythmias, coagulopathy
- Aggressive forced-air warming, warmed humidified oxygen, warmed IV fluids 40-42°C
- Rewarming rate 1-2°C/hour, monitor for afterdrop phenomenon
- Severe (<32°C): Ventricular fibrillation risk, profound coagulopathy, coma
- Active core rewarming: warmed bladder irrigation, peritoneal lavage, cardiopulmonary bypass
- Rewarming rate goal 1-2°C/hour, avoid >2°C/hour to prevent complications
⭐ Clinical Pearl: Afterdrop phenomenon-core temperature continues decreasing 0.5-1°C during first 20-30 minutes of rewarming as cold peripheral blood returns to core-anticipate and continue aggressive warming despite initial temperature decrease
Shivering: The Metabolic Storm
Postoperative shivering occurs in 40-60% of hypothermic patients, increasing metabolic rate by 200-600%, oxygen consumption by 400%, and carbon dioxide production by 300%. This metabolic demand precipitates myocardial ischemia in 15-20% of high-risk patients and delays PACU discharge by 30-45 minutes.
Shivering grading enables targeted pharmacological intervention:
- Grade 0: No shivering
- Grade 1: Piloerection, peripheral vasoconstriction without visible shivering
- Grade 2: Muscular activity in one muscle group (neck, thorax, upper extremities)
- Grade 3: Moderate muscular activity in more than one muscle group
- Grade 4: Violent muscular activity involving entire body
Pharmacological shivering suppression provides rapid symptom control while rewarming continues:
- Meperidine: Most effective anti-shivering agent, dose 12.5-25 mg IV
- Suppresses shivering through κ-opioid receptor agonism and α2-adrenergic effects
- Effective in 80-90% of patients within 5-10 minutes, duration 1-2 hours
- Lower doses (6.25-12.5 mg) reduce respiratory depression risk
- Dexmedetomidine: α2-agonist, loading dose 0.5-1 mcg/kg over 10 minutes
- Reduces shivering threshold, effective in 70-80% of cases
- Causes sedation and bradycardia, useful for agitated hypothermic patients
- Clonidine: Oral or IV 75-150 mcg, slower onset 20-30 minutes
- Prophylactic administration reduces shivering incidence by 50%
💡 Master This: Treat the patient, not the thermometer-aggressive shivering suppression in patients with coronary artery disease or limited cardiac reserve prevents myocardial ischemia even if core temperature remains 35-36°C; prioritize hemodynamic stability over achieving normothermia
Hyperthermia and Fever
Postoperative temperature elevation (>38°C) within the first 24 hours typically represents non-infectious causes, while fever developing >48 hours post-surgery suggests infectious complications. The 5 W's mnemonic guides systematic evaluation:
📌 Remember: 5 W's of Postoperative Fever - Wind (atelectasis, days 1-2), Water (UTI, days 3-5), Wound (surgical site infection, days 5-7), Walking (DVT/PE, days 5-10), Wonder drugs (drug fever, anytime)
Malignant hyperthermia represents a life-threatening hypermetabolic crisis occurring in 1:10,000-1:200,000 anesthetics, triggered by volatile anesthetics or succinylcholine in genetically susceptible individuals (RYR1 or CACNA1S mutations):
- Clinical presentation: Temperature increase 1-2°C every 5 minutes, masseter spasm, hypercarbia (ETCO2 >55 mmHg rising rapidly), tachycardia, arrhythmias
- Metabolic acidosis (pH <7.25), hyperkalemia (>6 mEq/L), elevated creatine kinase (>10,000 U/L)
- Myoglobinuria causing acute kidney injury in 30% of cases
- Immediate management: Discontinue triggering agents, hyperventilate with 100% oxygen at 2-3× minute ventilation
- Dantrolene 2.5 mg/kg IV rapid bolus, repeat every 5 minutes until symptoms resolve (average total dose 10 mg/kg)
- Active cooling: ice packs, cold IV fluids, gastric/bladder lavage with cold saline
- Treat hyperkalemia, acidosis, and arrhythmias; monitor for recrudescence requiring additional dantrolene
Thyroid storm presents with fever >40°C, tachycardia >140 bpm, altered consciousness, and hemodynamic instability in patients with uncontrolled hyperthyroidism stressed by surgery:
- Treatment: Propylthiouracil 500-1,000 mg loading dose (or methimazole 60-80 mg), then 250 mg every 6 hours
- Iodine solution **5-
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more
Have doubts about this lesson?
Ask Rezzy, your AI Study Mate, to explain anything you didn't understand
Everything you need for NEET-PG prep
Get full Oncourse access with lessons, practice questions, flashcards and AI study tools.
Scan to download app















