Opioid Basics - Receptors & Roadways
- Opioids: Substances binding opioid receptors; mimic endogenous peptides (e.g., endorphins, enkephalins, dynorphins).
- Receptors (All are GPCRs - Gi/Go coupled):
- μ (Mu - MOR): Primary target for analgesia (supraspinal & spinal). Also causes euphoria, respiratory depression, miosis, ↓GI motility, dependence.
- κ (Kappa - KOR): Spinal analgesia, sedation, dysphoria, miosis.
- δ (Delta - DOR): Analgesia (spinal & supraspinal), antidepressant effects, may be proconvulsant.
- NOP (Nociceptin/Orphanin FQ - ORL-1): Complex role; may modulate pain, anxiety, or opioid effects.
- Mechanism of Action:
- Presynaptic: ↓Ca²⁺ influx → ↓release of excitatory neurotransmitters (e.g., Substance P, glutamate).
- Postsynaptic: ↑K⁺ efflux → hyperpolarization → ↓neuronal excitability.
- Pain Pathway Modulation:
- Inhibit ascending pain transmission pathways (e.g., spinothalamic tract).
- Activate descending inhibitory pathways (e.g., PAG → RVM → dorsal horn).
⭐ Activation of μ-opioid receptors in the brainstem (medulla) is primarily responsible for the life-threatening side effect of respiratory depression associated with opioids like morphine and fentanyl.
Opioid Journey - 'Kinetics Express'
- Absorption:
- Oral: Variable bioavailability (F); Morphine F ~30% (high first-pass).
- IV/IM/SC: Reliable. Transdermal (Fentanyl): Slow onset, prolonged effect. Transmucosal (Fentanyl, Buprenorphine): Rapid.
- Distribution:
- Lipid solubility dictates speed & duration:
- High (e.g., Fentanyl): Rapid CNS entry & redistribution; quick on/off.
- Low (e.g., Morphine): Slower CNS entry, longer action.
- Lipid solubility dictates speed & duration:
- Metabolism:
- Hepatic: Predominant. Key enzymes:
- CYP2D6: Codeine → Morphine; Tramadol activation.
- CYP3A4: Fentanyl, Methadone, Oxycodone.
- Glucuronidation: Morphine → M6G (active analgesic), M3G (neuroexcitatory).
- Plasma Esterases: Remifentanil (ultra-short acting).
- Hepatic: Predominant. Key enzymes:
- Excretion:
- Renal: Primary route. Dose adjust for active metabolites (e.g., M6G, Normeperidine) in renal failure.

⭐ Codeine is a prodrug requiring CYP2D6 metabolism to morphine; its analgesic efficacy is highly variable due to genetic polymorphisms (poor, intermediate, extensive, or ultra-rapid metabolizers).
The Opioid Arsenal - Uses & Agents
| Agent | Potency (vs M) | Uses | Notes |
|---|---|---|---|
| Morphine | 1 | Severe pain, MI, Anesth. | Active metabolites (M6G, M3G); Histamine release; Renal caution |
| Fentanyl | 50-100 | Severe pain, Anesth., Epidural | Rapid, short; Chest rigidity; Patch |
| Remifentanil | 100-200 | Intraop anesth. (TIVA) | Ultra-short (esterase); Context-insensitive t½ |
| Pethidine | 0.1 | Pain, Post-op shiver | Norpethidine (seizures); Serotonin synd. risk; Atropine-like |
| Tramadol | 0.1-0.2 | Mod pain, Neuropathic | Weak µ, SNRI; ↓Seizure threshold; Serotonin synd. risk |
| Tapentadol | 0.4 | Mod-Sev pain (acute/chronic) | MOR-NRI; Less GI AEs |
| Buprenorphine | 25-50 | Mod-Sev pain, Opioid dep. | Partial µ-agonist, κ-antagonist; Ceiling effect (resp); SL |
| Naloxone | Antagonist | Opioid overdose | Reversal; Short t½ (repeat dose) |
Opioid Pitfalls - Dangers & Detox
- Acute Dangers:
- ⚠️ Respiratory depression (lethal); CNS depression.
- GI: Nausea, vomiting, severe constipation (no tolerance).
- Pupils: Miosis (pethidine: mydriasis).
- Chronic Issues:
- Tolerance (not miosis, constipation, convulsions).
- Dependence (physical/psychological).
- Opioid-Induced Hyperalgesia (OIH).
- Serotonin Syndrome (tramadol, pethidine, methadone + MAOI/SSRI).
- Seizures (pethidine metabolite, tramadol).
- Opioid Overdose: Triad: Coma, Resp. Depression, Miosis.
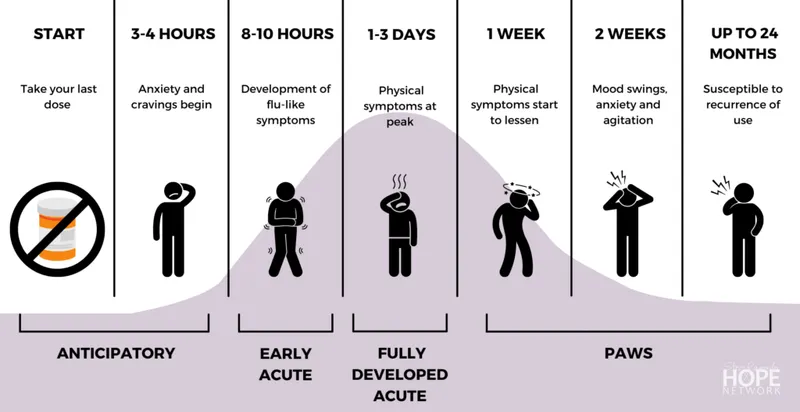
- Withdrawal (📌 FINISH Mnemonic):
- Fever, Insomnia, Nausea, Increased lacrimation, Sweating, Hyperreflexia/Hypertension. Also: yawning, mydriasis, piloerection, GI upset.
- Detox:
- Symptomatic: Clonidine.
- Substitution: Methadone, buprenorphine.
⭐ Naloxone's short half-life (30-90 min) vs. opioids necessitates repeat doses or infusion for sustained reversal, especially with long-acting opioids.
High‑Yield Points - ⚡ Biggest Takeaways
- Opioids act primarily on μ (mu) receptors; others include κ (kappa) and δ (delta).
- Morphine is the prototype; Fentanyl is highly potent (80-100x morphine).
- Naloxone is the specific antagonist for opioid overdose, reversing respiratory depression.
- Key adverse effects: Respiratory depression (most critical), constipation, miosis (pinpoint pupils).
- Pethidine is unique: causes mydriasis, tachycardia; its metabolite norpethidine can cause seizures.
- Buprenorphine, a partial μ-agonist, exhibits a ceiling effect for respiratory depression.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

