Amide LAs: Introduction - Identity & Basics
- Characterized by an amide link; generally more stable in solution than esters.
- 📌 Amides: Typically have two "i"s in their name (e.g., Lidocaine, Bupivacaine, Ropivacaine, Mepivacaine).

- Allergic reactions are rare compared to ester LAs.
⭐ Amides (e.g., Lidocaine, Mepivacaine) generally have two 'i's in their name and are primarily metabolized in the liver by microsomal enzymes, distinguishing them from ester LAs (one 'i', e.g., Procaine) which are hydrolyzed by plasma pseudocholinesterases.
Amide LAs: Mechanism & Journey - MOA & Pharmacokinetics
-
MOA: Reversible Na⁺ channel blockade (intracellular).
- Unionized (lipid-soluble) form crosses nerve membrane.
- Ionized (cationic) form is active blocker.
- Use-dependent: ↑ affinity for open/inactivated channels.

-
Pharmacokinetics (ADME):
- Absorption: Site vascularity, dose. Epinephrine ↓ absorption, ↑ duration.
- Distribution: Protein binding (α1-acid glycoprotein) affects duration.
- Metabolism: Hepatic (CYP450). 📌 Amides = Liver. Slower than esters.
- Excretion: Renal (metabolites).
-
Key Equation: Henderson-Hasselbalch: $pH = pKa + log([B]/[BH⁺])$. Lower pKa = faster onset. Acidic tissue ↓ efficacy.
⭐ The unionized (lipid-soluble) form of a local anesthetic penetrates the nerve sheath and membrane, while the ionized (cationic) form is responsible for blocking the intracellular side of the voltage-gated sodium channel.
Amide LAs: The Clinical Roster - Agents & Applications
| Agent | Onset | Duration | Potency | Max Dose (Plain) | Max Dose (w/ Epi) | Key Features / Uses |
|---|---|---|---|---|---|---|
| Lidocaine | Rapid | Med (1-2h) | Med | 4.5 mg/kg (300mg) | 7 mg/kg (500mg) | Versatile; antiarrhythmic; CNS tox (metabolites) |
| Bupivacaine | Slow | Long (2-8h) | High | 2 mg/kg (175mg) | 2.5 mg/kg (225mg) | Labor analgesia; 📌 High cardiotoxicity; differential block |
| Ropivacaine | Mod. | Long (2-8h) | High | 3 mg/kg (200mg) | - | ↓ Cardiotox vs Bupi; motor-sparing; epidural, regional blocks |
| Levobupivacaine | Slow | Long (2-8h) | High | 2 mg/kg (150mg) | - | S-enantiomer Bupi; ↓ cardiotox & CNS tox vs Bupi; similar uses |
| Prilocaine | Mod. | Med (1-3h) | Med | 6 mg/kg (400mg) | 8 mg/kg (600mg) | Methemoglobinemia risk (>8mg/kg); EMLA (topical) |
| Mepivacaine | Rapid | Med (1.5-3h) | Med | 4.5 mg/kg (400mg) | 7 mg/kg (500mg) | Less vasodilation; avoid obstetrics (fetal accum.); infiltration, blocks |
Amide LAs: Adverse Effects - Toxicity & Management
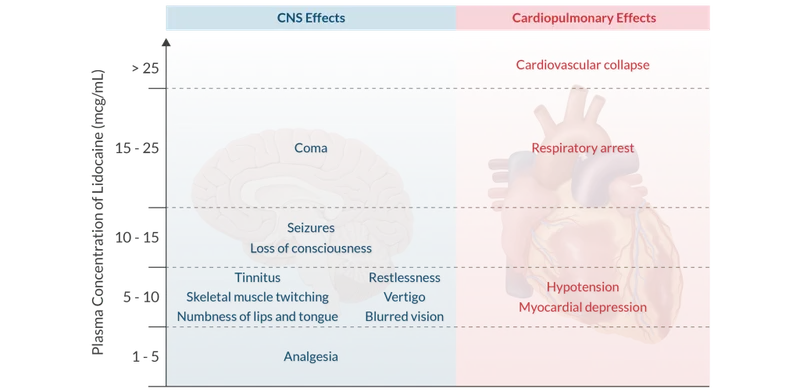
- CNS Toxicity: Early signs (low dose): circumoral numbness, tinnitus, lightheadedness, metallic taste. Progression: muscle twitching, visual disturbances, slurred speech, seizures, unconsciousness, respiratory arrest.
- CVS Toxicity: (Higher dose): Initial hypertension/tachycardia, then ↓contractility, hypotension, bradycardia, arrhythmias (VT/VF), asystole. Bupivacaine is highly cardiotoxic.
- Methemoglobinemia: Prilocaine, benzocaine. Cyanosis unresponsive to O2. Tx: Methylene blue 1-2 mg/kg IV.
- Allergic Reactions: Rare for amides (no PABA metabolite). Usually due to preservatives (e.g., methylparaben).
⭐ Severe Local Anesthetic Systemic Toxicity (LAST) Management: Stop LA. Airway (O2). Seizure control (benzodiazepines). 20% Lipid Emulsion (Intralipid): Bolus 1.5 mL/kg (1 min), then 0.25 mL/kg/min infusion. Repeat bolus x2 if needed. Max 10-12 mL/kg in 30 min. Modified ACLS for cardiac arrest.
High‑Yield Points - ⚡ Biggest Takeaways
- Amide LAs undergo hepatic metabolism (CYP450 enzymes), unlike esters.
- Amides typically have a longer duration of action than ester local anesthetics.
- True allergic reactions to amides are extremely rare.
- Systemic toxicity manifests as CNS excitation (seizures) then depression, and cardiovascular collapse.
- Bupivacaine exhibits significant cardiotoxicity; Ropivacaine and Levobupivacaine are safer alternatives.
- Lidocaine is also a Class IB antiarrhythmic.
- Prilocaine use carries a risk of methemoglobinemia (treat with methylene blue).
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

