Endocrine Pathology

On this page
🗺️ The Endocrine Architecture - Hormonal Command Centers
The endocrine system operates as a distributed network of specialized glands, each producing specific hormones that regulate metabolism, growth, reproduction, and homeostasis. Understanding gland-specific anatomy, embryology, and vascular supply predicts both normal function and characteristic pathology patterns.
Anatomical Organization and Clinical Correlates
The major endocrine glands occupy strategic anatomical positions that reflect their embryological origins and functional relationships:
-
Hypothalamic-Pituitary Axis
- Hypothalamus: Median eminence connects to anterior pituitary via portal system
- Pituitary gland: 10-13 mm height (normal), 6 mm in men, 8 mm in women, 10 mm in pregnancy
- Blood supply: Superior and inferior hypophyseal arteries from internal carotid
- Portal system delivers releasing hormones at 10-100× higher concentrations than systemic
- Posterior pituitary receives direct neuronal input from supraoptic and paraventricular nuclei
- Clinical significance: Pituitary tumors >10 mm classified as macroadenomas with higher compression risk
-
Thyroid Gland Architecture
- Weight: 15-20 g in adults, increases to 25-30 g in pregnancy
- Follicular organization: 20-40 follicles per lobule, lined by cuboidal epithelium
- Parafollicular C-cells: Scattered between follicles, produce calcitonin, represent <0.1% of thyroid mass
- C-cell hyperplasia threshold: >50 cells per low-power field
- Distribution: Upper two-thirds of lateral lobes (embryological ultimobranchial body origin)
-
Adrenal Gland Stratification
- Combined weight: 8-10 g (each gland 4-5 g)
- Cortex comprises 90% of gland mass, organized in three functional zones
- Medulla represents 10%, derived from neural crest chromaffin cells
- Cortical-to-medullary blood flow delivers 100× higher cortisol to medulla
- This high cortisol environment induces phenylethanolamine N-methyltransferase (PNMT) for epinephrine synthesis
📌 Remember: GFR for adrenal cortex zones (outside to inside) - Glomerulosa makes aldosterone (salt), Fasciculata makes cortisol (sugar), Reticularis makes androgens (sex). Blood flows centripetally, delivering highest cortisol concentrations to medulla for epinephrine synthesis.
Embryological Origins and Pathology Patterns
Understanding embryological development predicts tumor histology and syndromic associations:
| Structure | Embryological Origin | Key Developmental Timepoint | Characteristic Tumors | Clinical Significance |
|---|---|---|---|---|
| Anterior pituitary | Rathke's pouch (oral ectoderm) | Week 3-4 | Adenomas (lactotroph 40%, somatotroph 20%) | Craniopharyngiomas arise from remnants |
| Posterior pituitary | Neural ectoderm (diencephalon) | Week 5-6 | Rare: granular cell tumors | Diabetes insipidus with stalk damage |
| Thyroid follicular cells | Endoderm (foregut) | Week 3-4 migration | Papillary (80%), follicular (15%), anaplastic (2%) | Thyroglossal duct cysts along migration path |
| Parafollicular C-cells | Neural crest (ultimobranchial) | Week 5-6 | Medullary thyroid carcinoma (4% of thyroid cancers) | MEN 2 syndromes involve C-cells |
| Adrenal cortex | Mesoderm (coelomic epithelium) | Week 4-5 | Adenomas, carcinomas | Fetal cortex involutes postnatally |
| Adrenal medulla | Neural crest (sympathetic ganglia) | Week 6-7 migration | Pheochromocytoma, neuroblastoma | Chromaffin cells = modified neurons |
⭐ Clinical Pearl: Thyroid C-cells originate from neural crest, explaining why medullary thyroid carcinoma produces calcitonin and associates with other neural crest tumors (pheochromocytoma) in MEN 2 syndromes. Serum calcitonin >100 pg/mL has >95% sensitivity for medullary carcinoma.
Vascular Supply and Surgical Landmarks
Endocrine glands receive disproportionately high blood flow relative to their size, reflecting intense metabolic activity:
-
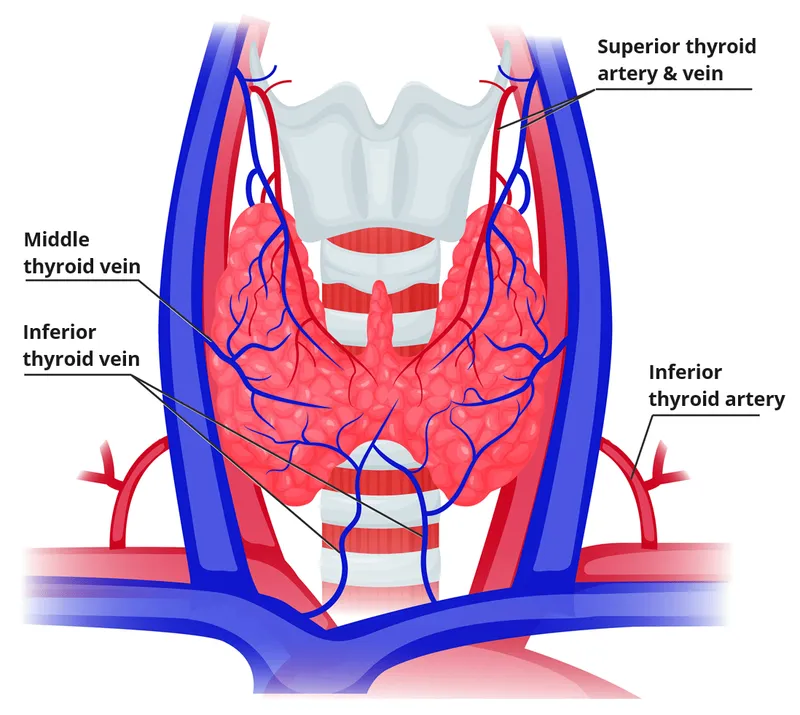
Thyroid Blood Flow
- 4-6 mL/min/g tissue (highest perfusion per gram in body)
- Superior thyroid artery: First branch of external carotid, supplies upper pole
- Inferior thyroid artery: Branch of thyrocervical trunk, supplies lower pole and parathyroids
- Recurrent laryngeal nerve crosses inferior thyroid artery in >80% of cases
- Nerve injury occurs in 1-2% of thyroidectomies, causing hoarseness
- Thyroidea ima artery: Present in 3-10%, arises from brachiocephalic trunk
-
Parathyroid Vascular Anatomy
- Superior parathyroids: 80% dorsal to recurrent laryngeal nerve, supplied by inferior thyroid artery
- Inferior parathyroids: Variable position (50% near lower pole, 25% in thymus, 10% mediastinal)
- Each gland weight: 30-40 mg, combined 120-160 mg
- Adenoma typically >500 mg (often 1-5 g)
- Hyperplastic glands: 60-80 mg each
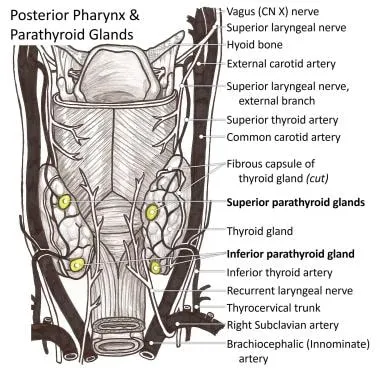
💡 Master This: The inferior thyroid artery supplies both inferior parathyroids and most superior parathyroids in >80% of cases. During thyroidectomy, preserving this arterial branch protects parathyroid viability. Transient hypocalcemia occurs in 20-30% of total thyroidectomies, permanent hypoparathyroidism in 1-3%.
Functional Reserve and Compensation Patterns
Endocrine glands demonstrate remarkable functional reserve, with clinical manifestations appearing only after substantial tissue loss:
-
Adrenal Cortical Reserve
- Clinical insufficiency requires >90% destruction of both glands
- Unilateral adrenalectomy: Contralateral gland hypertrophies to maintain function
- Autoimmune adrenalitis: Progressive destruction over months to years
- Subclinical phase: Elevated ACTH with normal cortisol
- Clinical crisis: Cortisol <3 μg/dL with stress
-
Pancreatic Islet Reserve
- Type 1 diabetes manifests after 80-90% beta-cell loss
- "Honeymoon period": Residual 10-20% beta-cells provide temporary function
- C-peptide levels: <0.6 ng/mL indicates severe beta-cell loss
- Fasting C-peptide <0.2 ng/mL confirms absolute insulin deficiency
-
Thyroid Functional Capacity
- Hypothyroidism after thyroidectomy if <20% remnant tissue
- Radioiodine ablation: Hypothyroidism develops in >90% within 1 year
- Thyroid reserve assessed by TSH response to TRH stimulation
- Normal: TSH rises >5-25 mIU/L above baseline
- Primary hypothyroidism: Exaggerated response (>30 mIU/L)
📌 Remember: 90-90-90 Rule for endocrine reserve - 90% adrenal destruction for insufficiency, 90% beta-cell loss for diabetes, 90% thyroid ablation for hypothyroidism. This extensive reserve explains why early endocrine disease is often subclinical.
Understanding these anatomical relationships, embryological patterns, and functional reserves provides the foundation for recognizing how pathological processes manifest clinically. Connect these structural principles through to understand how anatomical constraints influence tumor behavior and hormonal dysfunction patterns.
🗺️ The Endocrine Architecture — Hormonal Command Centers
📚 Hormone Synthesis Machinery - From Gene to Secretion
Hormone production involves complex biosynthetic pathways with multiple enzymatic steps, each representing a potential point of pathological disruption. Mastering these mechanisms reveals why specific enzyme deficiencies produce characteristic clinical syndromes and why certain tumors secrete particular hormone combinations.
Steroid Hormone Biosynthesis Cascade
All steroid hormones derive from cholesterol through sequential enzymatic modifications. The specific enzymes expressed in each zone determine the final hormone product:
- Adrenal Steroidogenesis Pathway
- Cholesterol → Pregnenolone (via CYP11A1/20,22-desmolase in mitochondria)
- Rate-limiting step: Cholesterol transport into mitochondria via StAR protein
- ACTH stimulation increases StAR expression 5-10 fold within 30 minutes
- Congenital lipoid adrenal hyperplasia: StAR mutation causes complete steroid deficiency
- Zona Glomerulosa (aldosterone synthesis)
- Pregnenolone → Progesterone (3β-HSD) → 11-Deoxycorticosterone (CYP21A2) → Corticosterone (CYP11B1) → Aldosterone (CYP11B2/aldosterone synthase)
- CYP11B2 expressed only in zona glomerulosa (angiotensin II and K+ regulation)
- Aldosterone production: 100-150 μg/day, increases 2-3 fold with sodium restriction
- Zona Fasciculata (cortisol synthesis)
- Pregnenolone → 17-Hydroxypregnenolone (CYP17A1) → 17-Hydroxyprogesterone (3β-HSD) → 11-Deoxycortisol (CYP21A2) → Cortisol (CYP11B1)
- Cortisol production: 15-25 mg/day, circadian rhythm with peak at 6-8 AM
- Cortisol-binding globulin (CBG) binds 90% of circulating cortisol, free fraction 5-10% is biologically active
- Zona Reticularis (androgen synthesis)
- 17-Hydroxypregnenolone → DHEA (CYP17A1 with 17,20-lyase activity) → Androstenedione → Testosterone (peripheral conversion)
- DHEA-sulfate (DHEA-S): 2-15 mg/day, exclusively adrenal origin (no ovarian/testicular production)
- Adrenarche: DHEA-S rises 10-20 fold between ages 6-15 years
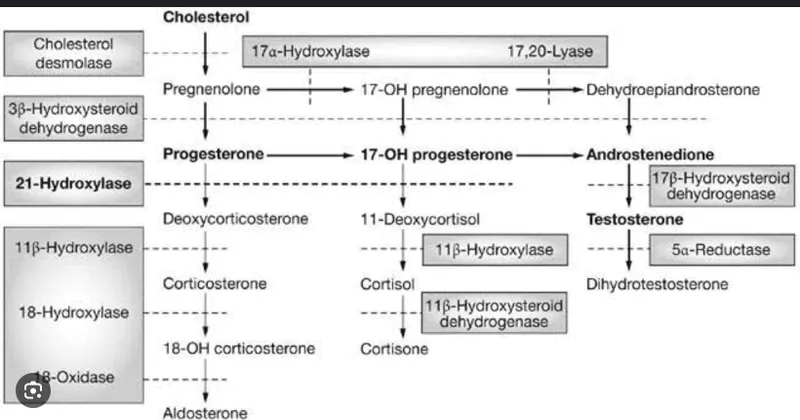
📌 Remember: "The deeper you go, the sweeter it gets" - Zona glomerulosa (superficial) makes salty aldosterone, zona fasciculata (middle) makes sweet cortisol (gluconeogenesis), zona reticularis (deep) makes sex steroids (androgens). Each zone expresses specific enzymes determining its product.
Congenital Adrenal Hyperplasia Enzyme Defects
Enzyme deficiencies in steroidogenesis produce predictable clinical syndromes based on accumulated precursors and deficient products:
| Enzyme Deficiency | Frequency | Cortisol | Aldosterone | Androgens | Accumulated Precursor | Clinical Features | 17-OHP Level |
|---|---|---|---|---|---|---|---|
| 21-Hydroxylase (CYP21A2) | 90-95% of CAH | ↓↓ | ↓ (classic) or normal (non-classic) | ↑↑↑ | 17-Hydroxyprogesterone | Virilization, salt-wasting in 75% of classic | >10,000 ng/dL (classic), 1,500-10,000 (non-classic) |
| 11β-Hydroxylase (CYP11B1) | 5-8% of CAH | ↓↓ | ↓ but DOC ↑↑ | ↑↑↑ | 11-Deoxycortisol, DOC | Virilization plus hypertension (DOC effect) | Normal, ↑ 11-deoxycortisol |
| 17α-Hydroxylase (CYP17A1) | <1% of CAH | ↓↓ | ↑ (via DOC pathway) | ↓↓↓ | Progesterone, DOC | Hypertension, sexual infantilism, hypokalemia | <3 ng/dL (very low) |
| 3β-HSD deficiency | <1% of CAH | ↓↓ | ↓↓ | Mild ↑ (DHEA) | DHEA, 17-Hydroxypregnenolone | Mild virilization (females), incomplete masculinization (males), salt-wasting | Elevated with high DHEA |
| StAR protein deficiency | Rare | ↓↓↓ | ↓↓↓ | ↓↓↓ | Cholesterol (lipoid accumulation) | Severe salt-wasting, complete steroid deficiency, lipoid adrenals | Very low all steroids |
⭐ Clinical Pearl: 21-Hydroxylase deficiency causes 90-95% of CAH cases. Newborn screening detects 17-OHP elevation (>10,000 ng/dL diagnostic for classic form). Salt-wasting crisis typically presents at 1-3 weeks of life with hyponatremia (<130 mEq/L), hyperkalemia (>6.5 mEq/L), and hypoglycemia. Non-classic form presents later with premature pubarche or hirsutism, with 17-OHP 1,500-10,000 ng/dL.
Thyroid Hormone Synthesis and Regulation
Thyroid hormone production involves iodine organification, thyroglobulin synthesis, and proteolytic release-a multi-step process vulnerable to autoimmune attack and genetic defects:
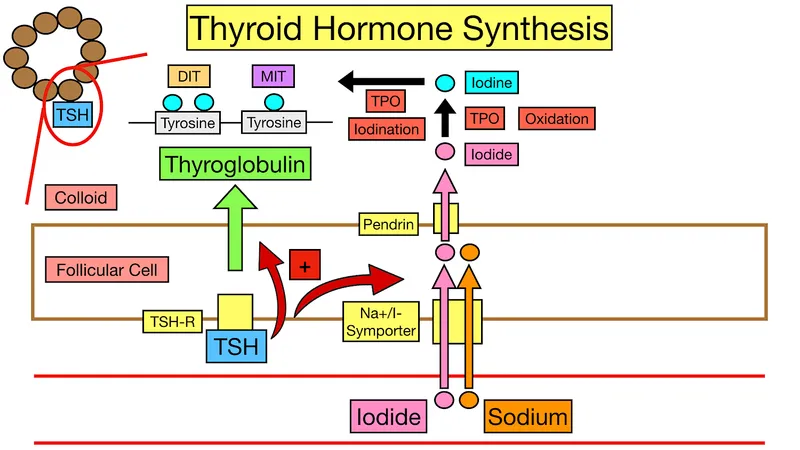
- Thyroid Hormone Biosynthesis Steps
- Step 1: Iodide uptake via sodium-iodide symporter (NIS) at basolateral membrane
- Active transport against concentration gradient (20-40:1 thyroid:plasma ratio)
- Daily iodine requirement: 150 μg, thyroid stores 8-10 mg (60-day supply)
- NIS expression regulated by TSH, suppressed by excess iodine (Wolff-Chaikoff effect)
- Step 2: Thyroglobulin (Tg) synthesis in follicular cells
- Large glycoprotein (660 kDa, dimer of 330 kDa subunits)
- Contains 134 tyrosine residues, only 25-30 become iodinated
- Secreted into follicular lumen, stored as colloid
- Step 3: Iodide oxidation and organification at apical membrane
- Thyroid peroxidase (TPO) oxidizes I⁻ to reactive iodine species using H₂O₂
- Iodination of tyrosine residues: MIT (monoiodotyrosine) and DIT (diiodotyrosine)
- Coupling reaction: MIT + DIT → T₃, DIT + DIT → T₄
- Step 4: Proteolytic release of T₄ and T₃
- Colloid endocytosis and lysosomal digestion of thyroglobulin
- T₄:T₃ production ratio 10:1 (T₄ 80-90 μg/day, T₃ 30-40 μg/day)
- Peripheral deiodination converts 80% of circulating T₃ from T₄
- Step 1: Iodide uptake via sodium-iodide symporter (NIS) at basolateral membrane
- Peripheral Thyroid Hormone Metabolism
- Type 1 deiodinase (D1): Liver and kidney, converts T₄ → T₃ (active)
- Type 2 deiodinase (D2): Brain and pituitary, local T₃ production for feedback
- Type 3 deiodinase (D3): Placenta and fetal tissues, converts T₄ → reverse T₃ (inactive)
- Reverse T₃ (rT₃): Biologically inactive, elevated in non-thyroidal illness syndrome
- Normal rT₃: 10-25 ng/dL, rises 2-3 fold in severe illness
- Thyroid hormone binding proteins
- Thyroxine-binding globulin (TBG): Binds 75% of T₄ and T₃
- Transthyretin (TTR): Binds 15-20% of T₄, minimal T₃ binding
- Albumin: Binds 5-10% of thyroid hormones
- Free hormone fraction: T₄ 0.03%, T₃ 0.3% (biologically active)
💡 Master This: The thyroid produces predominantly T₄ (10:1 ratio T₄:T₃), but T₃ is 3-4 times more potent at nuclear receptors. Peripheral conversion via Type 1 deiodinase generates 80% of circulating T₃. In non-thyroidal illness, D1 activity decreases and D3 increases, reducing active T₃ and elevating inactive reverse T₃-an adaptive response reducing metabolic rate during severe illness.
Peptide Hormone Processing and Secretion
Peptide hormones undergo post-translational modifications including proteolytic cleavage, glycosylation, and regulated exocytosis:
- Insulin Biosynthesis and Secretion
- Preproinsulin (110 amino acids) → Proinsulin (86 amino acids) → Insulin (51 amino acids) + C-peptide (31 amino acids)
- Proinsulin cleaved by prohormone convertases PC1/3 and PC2
- Equimolar secretion of insulin and C-peptide
- C-peptide half-life 30 minutes vs insulin 5 minutes (renal clearance)
- Fasting C-peptide 0.8-3.1 ng/mL, insulin 5-15 μU/mL
- Glucose-stimulated insulin secretion (GSIS)
- Glucose enters beta-cells via GLUT2 transporter (Km 15-20 mM, acts as glucose sensor)
- Glucokinase phosphorylates glucose (rate-limiting step, Km 8-10 mM)
- ATP/ADP ratio increases, closing K⁺-ATP channels → depolarization
- Voltage-gated Ca²⁺ channels open, Ca²⁺ influx triggers exocytosis
- Biphasic insulin secretion
- First phase: Rapid release of stored granules within 5-10 minutes
- Second phase: Sustained release from 10-120 minutes
- First-phase loss is earliest defect in type 2 diabetes
📌 Remember: "C-peptide is the Clue" - C-peptide levels distinguish endogenous insulin production from exogenous insulin administration. Insulinoma: Elevated C-peptide (>0.6 ng/mL) with hypoglycemia. Factitious hypoglycemia: Low C-peptide (<0.2 ng/mL) with elevated insulin. C-peptide suppression test: Failure to suppress <0.6 ng/mL after insulin administration confirms autonomous insulin secretion.
These biosynthetic pathways demonstrate how molecular defects translate into clinical syndromes. Connect these mechanisms through and to understand how enzyme deficiencies and autoimmune destruction produce characteristic hormone excess and deficiency states.
📚 Hormone Synthesis Machinery — From Gene to Secretion
🔬 Pattern Recognition - Hormone Excess vs Deficiency Signatures
Endocrine disorders present with characteristic clinical patterns determined by which hormones are elevated or deficient. Recognizing these signatures enables rapid syndrome identification and guides targeted laboratory evaluation.
Hormone Excess Syndromes: Clinical Recognition Frameworks
Hormone hypersecretion produces distinct clinical phenotypes based on the specific hormone's physiological actions:
- Cushing's Syndrome Recognition Pattern
- Central obesity: Truncal fat accumulation with preserved limb muscle mass
- Buffalo hump (dorsocervical fat pad) in 50-60%
- Supraclavicular fat pads in 40-50%
- Catabolic features: Protein breakdown manifestations
- Proximal muscle weakness (difficulty rising from chair) in 60-70%
- Skin thinning with easy bruising in 60-80%
- Wide purple striae (>1 cm width) in 50-70%, most specific feature
- Metabolic derangements
- Hyperglycemia in 60-80% (cortisol antagonizes insulin)
- Hypertension in 80-90% (mineralocorticoid effects + increased vascular reactivity)
- Hypokalemia in 10-20% (mainly with ectopic ACTH)
- Quantitative thresholds
- 24-hour urinary free cortisol >3× upper limit (>300 μg/24h)
- Late-night salivary cortisol >145 ng/dL (loss of circadian rhythm)
- Dexamethasone suppression: Failure to suppress cortisol <1.8 μg/dL after 1 mg overnight
- Central obesity: Truncal fat accumulation with preserved limb muscle mass
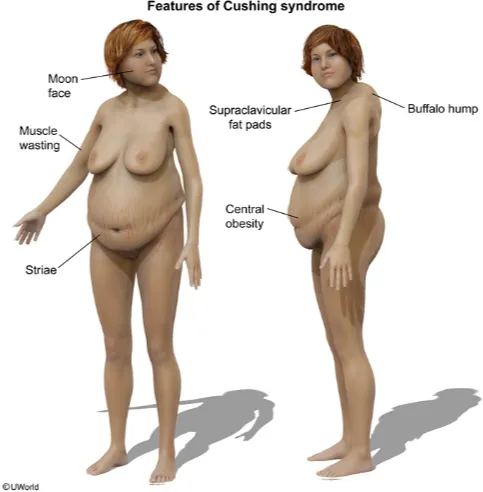
⭐ Clinical Pearl: Wide purple striae >1 cm are the most specific clinical feature of Cushing's syndrome (sensitivity 60%, specificity >95%). Distinguish from common obesity striae (pink, narrow, <1 cm). Proximal myopathy demonstrated by inability to rise from squatting position without arm support appears in 60-70% and reflects severe protein catabolism.
-
Acromegaly Recognition Pattern
- Acral enlargement: Progressive growth of hands, feet, jaw
- Ring and shoe size increase over 2-3 years
- Frontal bossing, prognathism (jaw protrusion) in >90%
- Macroglossia (tongue enlargement) in 40-50%
- Soft tissue overgrowth
- Coarse facial features with enlarged nose, lips
- Skin tags in 40-50% (correlate with colonic polyps)
- Carpal tunnel syndrome in 50-60% (median nerve compression)
- Systemic complications
- Diabetes mellitus in 40-50% (GH antagonizes insulin)
- Hypertension in 40-50% (increased sodium retention)
- Cardiomyopathy in 30-40% (biventricular hypertrophy, diastolic dysfunction)
- Colonic polyps in 30-50%, colon cancer risk increased 2-3 fold
- Quantitative diagnosis
- IGF-1 elevated >2.5× upper limit for age and sex
- GH fails to suppress <0.4 ng/mL during oral glucose tolerance test
- Random GH >10 ng/mL highly suggestive
- Acral enlargement: Progressive growth of hands, feet, jaw
-
Pheochromocytoma Recognition Pattern
- Classic triad (only 25-30% have all three)
- Episodic headaches (80-90% of symptomatic patients)
- Profuse sweating (60-70%)
- Palpitations with tachycardia (60-70%)
- Paroxysmal hypertension: Episodes lasting 15-60 minutes
- Sustained hypertension in 50-60%, paroxysmal in 30-40%
- Blood pressure spikes >200/120 mmHg during episodes
- Orthostatic hypotension in 30-40% (volume depletion from pressure natriuresis)
- Metabolic effects
- Hyperglycemia during episodes (catecholamine-induced insulin resistance)
- Weight loss despite normal appetite (increased metabolic rate)
- Diagnostic thresholds
- Plasma metanephrines >4× upper limit (normetanephrine + metanephrine)
- 24-hour urinary metanephrines >2× upper limit
- Specificity >95% when >3× elevated
- Classic triad (only 25-30% have all three)
📌 Remember: "5 P's of Pheo" - Pressure (hypertension), Palpitations, Perspiration (sweating), Pallor, Pounding headache. But remember only 25-30% have the classic triad. The "rule of 10s": 10% bilateral, 10% extra-adrenal (paraganglioma), 10% malignant, 10% familial, 10% in children. Modern series show familial cases actually 30-40% with genetic testing.
Hormone Deficiency Syndromes: Clinical Recognition Frameworks
Hormone deficiency produces characteristic patterns reflecting loss of specific physiological actions:
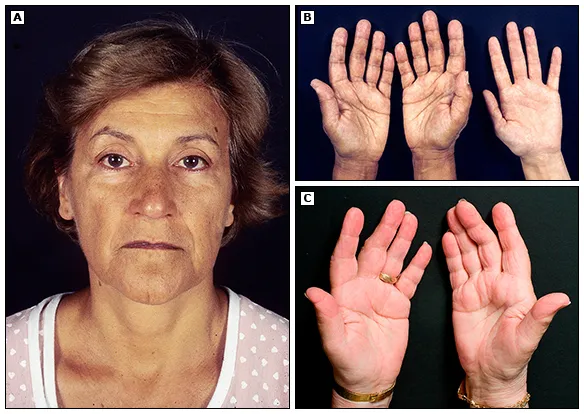
- Primary Adrenal Insufficiency (Addison's Disease) Pattern
- Hyperpigmentation: ACTH elevation stimulates melanocortin receptors
- Generalized darkening, most prominent in sun-exposed areas, skin creases, buccal mucosa
- Develops over 3-6 months in chronic insufficiency
- Absent in secondary adrenal insufficiency (low ACTH)
- Hypotension and salt craving
- Orthostatic hypotension in 80-90% (loss of aldosterone and cortisol)
- Systolic BP typically <110 mmHg, drops >20 mmHg on standing
- Salt craving reported in 15-20% (aldosterone deficiency)
- Laboratory hallmarks
- Hyponatremia (<135 mEq/L) in 80-90% (cortisol and aldosterone deficiency)
- Hyperkalemia (>5.5 mEq/L) in 60-70% (aldosterone deficiency)
- Hypoglycemia in 10-20% (cortisol deficiency impairs gluconeogenesis)
- Eosinophilia (>400/μL) in 15-20% (cortisol normally suppresses eosinophils)
- Diagnostic confirmation
- Morning cortisol <3 μg/dL diagnostic, >15 μg/dL excludes
- ACTH stimulation test: Cortisol fails to rise >18-20 μg/dL at 30 or 60 minutes
- Elevated ACTH >100 pg/mL (normal 10-60 pg/mL) confirms primary insufficiency
- Hyperpigmentation: ACTH elevation stimulates melanocortin receptors
- Hypothyroidism Recognition Pattern
- Metabolic slowing: Reduced basal metabolic rate manifestations
- Fatigue and lethargy in >90%
- Cold intolerance in 60-80%
- Weight gain (2-5 kg) despite reduced appetite in 60-70%
- Constipation in 40-60% (reduced GI motility)
- Myxedema features: Glycosaminoglycan accumulation
- Non-pitting edema of face, periorbital region in 50-70%
- Coarse, dry skin in 60-80%
- Hair loss, brittle hair in 40-60%
- Delayed relaxation phase of deep tendon reflexes in 30-40%
- Cardiovascular effects
- Bradycardia (<60 bpm) in 30-40%
- Diastolic hypertension in 20-30% (increased peripheral resistance)
- Pericardial effusion in 30-50% of severe cases (rarely symptomatic)
- Laboratory thresholds
- TSH >10 mIU/L with low free T₄ = overt hypothyroidism
- TSH 4.5-10 mIU/L with normal free T₄ = subclinical hypothyroidism
- Free T₄ <0.8 ng/dL (normal 0.8-1.8 ng/dL)
- Elevated LDL cholesterol in 60-80% (reduced LDL receptor expression)
- Metabolic slowing: Reduced basal metabolic rate manifestations
💡 Master This: Distinguish primary from secondary adrenal insufficiency by ACTH and pigmentation. Primary (Addison's): High ACTH (>100 pg/mL), hyperpigmentation present, hyperkalemia common (aldosterone deficiency). Secondary (pituitary): Low ACTH (<10 pg/mL), no hyperpigmentation, normal potassium (intact aldosterone), often with other pituitary hormone deficiencies. Primary causes 80-90% autoimmune in developed countries, tuberculosis in endemic areas.
- Hypoparathyroidism Recognition Pattern
- Neuromuscular irritability: Hypocalcemia effects
- Paresthesias (perioral, fingertips) in >90%
- Muscle cramps, tetany in 60-80%
- Chvostek's sign: Facial twitching with facial nerve tap (present in 70-80%, but also 10-15% of normals)
- Trousseau's sign: Carpopedal spasm after 3 minutes of BP cuff inflation (more specific, 90% sensitivity)
- CNS manifestations
- Seizures in 20-30% of severe hypocalcemia
- Altered mental status, confusion in severe cases
- Basal ganglia calcification on CT in chronic cases (30-40%)
- Cardiovascular effects
- Prolonged QT interval (corrected QT >460 ms in women, >440 ms in men)
- Risk of torsades de pointes if QTc >500 ms
- Laboratory diagnosis
- Serum calcium <8.5 mg/dL (ionized Ca <4.6 mg/dL)
- PTH inappropriately low or normal (should be elevated with hypocalcemia)
- Phosphate >4.5 mg/dL (PTH deficiency reduces renal excretion)
- 25-hydroxyvitamin D normal (distinguishes from vitamin D deficiency)
- Neuromuscular irritability: Hypocalcemia effects
These recognition patterns enable rapid syndrome identification. Connect these clinical frameworks through to understand how to confirm suspected diagnoses with targeted testing and distinguish primary from secondary disorders.
🔬 Pattern Recognition — Hormone Excess vs Deficiency Signatures
🏥 Differential Diagnosis - Distinguishing Similar Presentations
Endocrine syndromes often present with overlapping features requiring systematic differentiation based on quantitative laboratory parameters and specific clinical discriminators.
Cushing's Syndrome vs Pseudo-Cushing's States
Multiple conditions produce cushingoid features without true cortisol excess, requiring careful biochemical distinction:
| Feature | True Cushing's Syndrome | Obesity-Related Pseudo-Cushing's | Alcohol-Related Pseudo-Cushing's | Depression-Related Pseudo-Cushing's | Polycystic Ovary Syndrome |
|---|---|---|---|---|---|
| 24h UFC | >3× ULN (>300 μg/24h) | Normal to 2× ULN | Normal to 2× ULN, normalizes with abstinence | Normal to 2× ULN | Normal |
| Dex suppression | Fails to suppress <1.8 μg/dL | Usually suppresses | May not suppress acutely | May not suppress | Suppresses |
| Midnight cortisol | Elevated >7.5 μg/dL (loss of rhythm) | Normal rhythm preserved | May be elevated | May be elevated | Normal |
| Purple striae | Wide >1 cm, purple | Pink, narrow <5 mm | Absent | Absent | Absent |
| Proximal myopathy | Present 60-70% | Absent or mild | Absent | Absent | Absent |
| Hypokalemia | Present 10-20% (ectopic ACTH) | Absent | Absent | Absent | Absent |
| ACTH level | High (ectopic), normal-high (pituitary), low (adrenal) | Normal | Normal | Normal | Normal |
⭐ Clinical Pearl: The CRH stimulation test distinguishes true Cushing's from pseudo-Cushing's with >90% accuracy. True Cushing's: Cortisol rises >20% above baseline. Pseudo-Cushing's: Blunted response <20% rise. The dexamethasone-CRH test (dexamethasone 0.5 mg every 6 hours × 2 days, then CRH administration) has 95% sensitivity and 100% specificity when cortisol >1.4 μg/dL 15 minutes post-CRH indicates true Cushing's.
Thyrotoxicosis Differential: Distinguishing Causes
Multiple mechanisms produce thyrotoxicosis, requiring radioiodine uptake and clinical correlation for differentiation:
-
High Radioiodine Uptake Causes (increased hormone synthesis)
- Graves' disease (60-80% of thyrotoxicosis)
- Diffuse uptake on scan, uptake >30-40% at 24 hours
- TSH receptor antibodies (TRAb) positive in >95%
- Ophthalmopathy in 30-50%, pretibial myxedema in 1-5%
- Toxic multinodular goiter (15-20% of thyrotoxicosis)
- Patchy uptake with hot and cold nodules
- Older patients (typically >50 years)
- TRAb negative, no ophthalmopathy
- Toxic adenoma (3-5% of thyrotoxicosis)
- Single hot nodule with suppressed surrounding tissue
- Usually >3 cm diameter for autonomous function
- Graves' disease (60-80% of thyrotoxicosis)
-
Low Radioiodine Uptake Causes (thyroid destruction or exogenous hormone)
- Subacute thyroiditis (de Quervain's)
- Uptake <5% at 24 hours
- Painful thyroid in >80%, ESR markedly elevated (>50 mm/h)
- Triphasic course: Thyrotoxic (3-6 weeks) → hypothyroid (2-6 months) → recovery
- Painless thyroiditis (postpartum, silent)
- Uptake <5%, painless thyroid
- Postpartum onset in 5-10% of women (1-6 months postpartum)
- Positive thyroid antibodies in 80%
- Exogenous thyroid hormone (factitious)
- Uptake <2%
- Low thyroglobulin (<5 ng/mL) distinguishes from thyroiditis (elevated Tg)
- History of access to thyroid hormone
- Subacute thyroiditis (de Quervain's)

💡 Master This: Thyroglobulin (Tg) level distinguishes destructive thyroiditis from factitious thyrotoxicosis. Both have low radioiodine uptake. Thyroiditis: Elevated Tg (>40-100 ng/mL) from follicular destruction releasing stored hormone. Factitious: Suppressed Tg (<5 ng/mL) because exogenous hormone suppresses TSH and thyroid function. Normal Tg 3-40 ng/mL.
Hypercalcemia Differential: PTH-Dependent vs PTH-Independent
Serum PTH level immediately categorizes hypercalcemia into two major pathophysiological groups:
- PTH-Dependent Hypercalcemia (PTH elevated or inappropriately normal)
- Primary hyperparathyroidism (>85% of outpatient hypercalcemia)
- Calcium 10.5-12.5 mg/dL (mild elevation)
- PTH elevated or "inappropriately normal" (should be suppressed)
- Phosphate <2.5 mg/dL (PTH increases renal excretion)
- Alkaline phosphatase normal or mildly elevated
- 24-hour urine calcium typically >250 mg (distinguishes from FHH)
- Familial hypocalciuric hypercalcemia (FHH)
- Calcium 10.5-11.5 mg/dL (lifelong, asympto
- Primary hyperparathyroidism (>85% of outpatient hypercalcemia)
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more
Have doubts about this lesson?
Ask Rezzy, your AI Study Mate, to explain anything you didn't understand
Everything you need for NEET-PG prep
Get full Oncourse access with lessons, practice questions, flashcards and AI study tools.
Scan to download app














