Vestibular Disorders

On this page
🌀 The Vestibular Universe: Mastering Balance's Complex Orchestra
Your patient can't stand without falling, the room won't stop spinning, and the diagnosis isn't obvious-welcome to vestibular disorders, where the inner ear's delicate balance machinery meets complex neurological pathways. You'll master how the vestibular system maintains equilibrium, learn to distinguish peripheral from central causes through targeted examination techniques, and build systematic approaches to diagnose and treat conditions from benign positional vertigo to life-threatening posterior circulation strokes. This lesson transforms vertigo from a diagnostic puzzle into a structured clinical challenge you can confidently navigate.

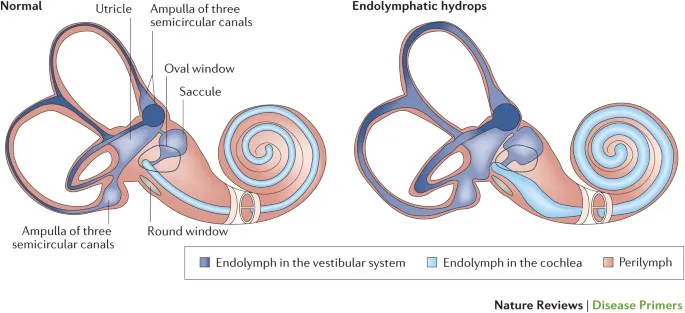
Vestibular disorders represent a complex spectrum of conditions affecting the inner ear's balance organs and their central nervous system connections. These disorders impact 15% of adults annually, with 40% of people over 65 experiencing significant balance problems. The vestibular system processes angular and linear acceleration through five distinct sensory organs: three semicircular canals detecting rotational movement and two otolith organs (utricle and saccule) sensing linear acceleration and gravity.
📌 Remember: SCUBA for vestibular anatomy - Superior, Costerior, Utricle, Baseline (horizontal canal), Accelerometer (saccule). The superior canal detects pitch movements, posterior canal handles roll, horizontal canal processes yaw, utricle senses horizontal linear acceleration, and saccule detects vertical movements.
The vestibular system maintains spatial orientation through three primary mechanisms: the vestibulo-ocular reflex (VOR) stabilizing vision during head movement, vestibulo-spinal reflexes maintaining postural stability, and vestibulo-collic reflexes controlling neck muscle responses. Normal VOR gain approaches 1.0, meaning eye movement velocity precisely matches head movement velocity in the opposite direction.

- Peripheral Vestibular Disorders (85% of cases)
- Benign Paroxysmal Positional Vertigo (BPPV): 17% of all vertigo cases
- Vestibular neuritis: 7% of vertigo presentations
- Ménière's disease: 3-5% of dizzy patients
- Endolymphatic hydrops with fluctuating hearing loss
- Classic triad occurs in only 45% of cases
- Central Vestibular Disorders (15% of cases)
- Vestibular migraine: 10-15% of migraine patients
- Posterior circulation stroke: 3% of acute vertigo
- Multiple sclerosis: 20% of MS patients develop vertigo
| Disorder Type | Prevalence | Duration | Hearing Loss | Nystagmus Pattern | Recovery Time |
|---|---|---|---|---|---|
| BPPV | 17% | <1 minute | None | Rotatory/upbeating | 2-6 weeks |
| Vestibular Neuritis | 7% | Hours-days | None | Horizontal | 6-12 weeks |
| Ménière's Disease | 3-5% | 20min-24hrs | Fluctuating | Horizontal | Variable |
| Vestibular Migraine | 1% population | Minutes-hours | Rare | Variable | Hours-days |
| Central Vertigo | 15% | Variable | Rare | Direction-changing | Variable |
💡 Master This: Vestibular disorders follow predictable patterns based on anatomical involvement. Peripheral lesions produce unidirectional nystagmus that decreases with fixation, while central lesions cause direction-changing nystagmus that persists with fixation. Understanding this fundamental difference guides immediate diagnostic decisions and determines urgency of intervention.
The vestibular system's remarkable 100-fold dynamic range allows detection of head movements from 0.1°/second to 1000°/second, enabling function from gentle walking to rapid athletic movements. This sophisticated sensory apparatus connects through the 8th cranial nerve to brainstem nuclei, processing information within 16 milliseconds to generate compensatory responses.
🌀 The Vestibular Universe: Mastering Balance's Complex Orchestra
⚙️ Vestibular Pathophysiology: The Balance Breakdown Blueprint
Vestibular dysfunction occurs through four primary pathophysiological mechanisms: mechanical displacement (BPPV), inflammatory damage (vestibular neuritis), fluid pressure changes (Ménière's disease), and central processing disorders (vestibular migraine). Each mechanism produces distinct temporal patterns and recovery trajectories based on the underlying cellular damage and repair processes.
Mechanical Displacement Pathophysiology dominates BPPV presentations, where calcium carbonate otoconia (specific gravity 2.94) become dislodged from the utricle and migrate into semicircular canals. These dense particles create inappropriate endolymph flow during head position changes, generating false rotational signals. The posterior semicircular canal is affected in 85% of BPPV cases due to its dependent anatomical position.
📌 Remember: DAMP for otoconia displacement - Dense particles, Anatomical dependency, Mechanical stimulation, Positional triggers. Otoconia are 3x denser than endolymph, creating gravitational effects that normal cupular deflection cannot produce.
Inflammatory Pathophysiology characterizes vestibular neuritis, where viral infection (commonly HSV-1) damages the superior vestibular nerve. This creates acute unilateral vestibular hypofunction with complete loss of horizontal canal function on the affected side. The inflammatory process peaks within 24-48 hours and gradually resolves over 6-12 weeks through central compensation mechanisms.
- Acute Phase Compensation (0-72 hours)
- Static symptoms: severe vertigo, nausea, vomiting
- VOR gain drops to 0.1-0.3 on affected side
- Spontaneous nystagmus 10-15°/second toward healthy ear
- Intensity decreases 50% within 24 hours
- Completely suppressed by visual fixation after 48 hours
- Subacute Compensation (3 days-6 weeks)
- Dynamic symptoms: oscillopsia during head movement
- Central adaptation begins within 72 hours
- Brainstem plasticity restores 60-80% of function
- Commissural inhibition rebalancing
- Cerebellar adaptation mechanisms
- Chronic Compensation (6 weeks-6 months)
- Residual symptoms: motion sensitivity, imbalance
- 20-30% develop chronic dizziness
- Vestibular rehabilitation accelerates recovery by 40%
Pressure-Related Pathophysiology defines Ménière's disease through endolymphatic hydrops - abnormal accumulation of endolymph causing increased intralabyrinthine pressure. This pressure elevation reaches 2-3x normal levels, distorting the Reissner's membrane and creating fluctuating symptoms. The hydrops affects both auditory and vestibular functions simultaneously.
| Pathophysiology Type | Primary Mechanism | Time Course | Recovery Pattern | Compensation Rate |
|---|---|---|---|---|
| Mechanical (BPPV) | Otoconia displacement | Seconds-minutes | Complete with treatment | 95% in 1-2 sessions |
| Inflammatory (VN) | Viral nerve damage | Hours-days | Gradual compensation | 70-80% in 6-12 weeks |
| Pressure (Ménière's) | Endolymphatic hydrops | 20min-24hrs | Fluctuating episodes | Variable, progressive |
| Central (VM) | Cortical hypersensitivity | Minutes-hours | Episode resolution | 60-80% with prophylaxis |
| Vascular (Stroke) | Ischemic damage | Sudden onset | Limited recovery | 30-50% improvement |
Central Processing Pathophysiology underlies vestibular migraine, where cortical spreading depression and trigemino-vascular activation create vestibular hypersensitivity. This mechanism affects vestibular nuclei and thalamo-cortical pathways, producing motion sensitivity without peripheral vestibular damage. The process involves CGRP release and neurogenic inflammation similar to classic migraine pathophysiology.
💡 Master This: Vestibular compensation follows predictable neuroplasticity principles. Acute static symptoms resolve through brainstem rebalancing within days, while dynamic symptoms require cerebellar adaptation over weeks to months. Understanding this timeline guides treatment expectations and rehabilitation strategies, with early mobilization accelerating compensation by 40-60% compared to prolonged bed rest.
The vestibular system's bilateral symmetry creates vulnerability to unilateral damage, as normal function depends on balanced tonic firing rates of 90 spikes/second from each labyrinth. Any asymmetry exceeding 20% produces vertigo symptoms, explaining why even partial vestibular loss creates significant clinical presentations.
⚙️ Vestibular Pathophysiology: The Balance Breakdown Blueprint
🎯 Clinical Recognition Mastery: The Diagnostic Detective Framework
Vestibular pattern recognition centers on temporal characteristics, trigger identification, and associated symptoms that create diagnostic fingerprints. The key principle: "When you see X pattern, think Y disorder" - but always confirm with objective testing since 40% of patients provide misleading symptom descriptions.
BPPV Recognition Patterns follow the "Position-Trigger-Duration" triad:
- Position triggers: Rolling in bed (85%), looking up (70%), bending forward (**60%)
- Duration signature: <60 seconds per episode, immediate onset with position change
- Symptom pattern: Rotational vertigo with nausea but no hearing loss
- Recovery characteristic: Complete resolution between episodes
📌 Remember: BPPV-STOP for recognition - Brief episodes, Positional triggers, Vertigo rotational, Sudden onset, Transient duration, Otologic normal, Pattern reproducible. Episodes last 15-45 seconds typically, never exceeding 2 minutes.
Vestibular Neuritis Recognition Patterns demonstrate the "Acute-Unilateral-Prolonged" signature:
-
Onset pattern: Sudden severe vertigo reaching peak intensity within 6 hours
-
Duration signature: Continuous symptoms for 24-72 hours, then gradual improvement
-
Movement pattern: Horizontal nystagmus beating away from affected ear
-
Hearing preservation: Normal audiometry distinguishes from labyrinthitis
-
Acute Phase Recognition (0-72 hours)
- Severe vertigo preventing ambulation
- Spontaneous nystagmus visible without fixation removal
- Positive head impulse test with catch-up saccades
- Abnormal VOR gain <0.6 on affected side
- Corrective saccades indicate peripheral lesion
- Nausea and vomiting in 95% of cases
-
Subacute Phase Recognition (3 days-6 weeks)
- Motion-provoked dizziness replaces constant vertigo
- Oscillopsia during rapid head movements
- Unsteadiness worse in dark or uneven surfaces
- Romberg test positive with eyes closed
- Dynamic visual acuity drops >3 lines
-
Chronic Phase Recognition (>6 weeks)
- Persistent motion sensitivity in 25% of patients
- Exercise intolerance with head movement
- Functional disability requiring rehabilitation
Ménière's Disease Recognition Patterns follow the "Fluctuating-Episodic-Progressive" model:
- Episode pattern: Spontaneous attacks lasting 20 minutes to 24 hours
- Hearing pattern: Low-frequency hearing loss that fluctuates with attacks
- Fullness pattern: Aural pressure preceding or accompanying vertigo
- Progression pattern: Increasing hearing loss over years with decreasing vertigo
| Recognition Pattern | BPPV | Vestibular Neuritis | Ménière's Disease | Vestibular Migraine | Central Vertigo |
|---|---|---|---|---|---|
| Onset Speed | Immediate | Hours | Minutes | Variable | Sudden/gradual |
| Episode Duration | <1 minute | Days | 20min-24hrs | Minutes-hours | Variable |
| Trigger Pattern | Positional | None | Spontaneous | Stress/food | Variable |
| Hearing Loss | None | None | Fluctuating | Rare | Rare |
| Nystagmus Type | Rotatory | Horizontal | Horizontal | Variable | Direction-changing |
- Trigger pattern: Stress (70%), hormonal changes (60%), dietary factors (40%)
- Duration variability: Minutes to hours, unlike other vestibular disorders
- Associated symptoms: Headache (80%), photophobia (60%), phonophobia (50%)
- Response pattern: Improvement with migraine medications confirms diagnosis
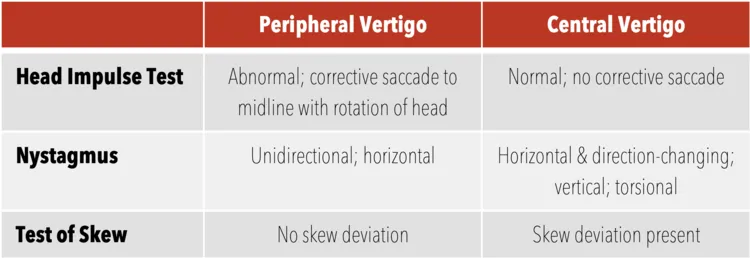
⭐ Clinical Pearl: The "HINTS Plus" examination (Head Impulse, Nystagmus, Test of Skew, Plus hearing test) achieves 99.5% sensitivity for stroke detection in acute vertigo. Any central feature (normal head impulse, direction-changing nystagmus, skew deviation, or sudden hearing loss) mandates immediate neuroimaging.
Central Vertigo Recognition Patterns demonstrate "Red Flag" characteristics:
- Nystagmus pattern: Direction-changing or purely vertical
- Neurological signs: Diplopia, dysarthria, ataxia, weakness
- HINTS findings: Normal head impulse in acute vertigo
- Hearing pattern: Sudden bilateral hearing loss suggests AICA stroke
💡 Master This: Effective vestibular diagnosis requires systematic pattern analysis rather than symptom checklists. Temporal patterns (onset, duration, triggers) provide 80% of diagnostic information, while physical examination confirms the suspected diagnosis. The "Rule of 3s" guides urgency: 3 minutes for BPPV episodes, 3 days for vestibular neuritis peak, 3 weeks for compensation, and 3 months for chronic symptoms requiring intervention.
Recognition mastery develops through understanding that vestibular symptoms represent final common pathways from diverse pathophysiological mechanisms. The diagnostic fingerprint emerges from pattern combinations, not individual symptoms, requiring systematic evaluation of temporal characteristics, triggers, and associated features.
🎯 Clinical Recognition Mastery: The Diagnostic Detective Framework
🔬 Differential Diagnosis Architecture: The Systematic Discrimination Matrix
Tier 1: Temporal Pattern Discrimination uses quantitative timing thresholds:
- Seconds-duration (5-60 seconds): BPPV, orthostatic hypotension
- Minutes-duration (2-20 minutes): Transient ischemic attack, panic attacks
- Hours-duration (1-24 hours): Ménière's disease, vestibular migraine
- Days-duration (24 hours-weeks): Vestibular neuritis, labyrinthitis
- Persistent (continuous >4 weeks): Central lesions, bilateral vestibulopathy
Tier 2: Objective Testing Discrimination provides quantitative differentiation:
| Test Parameter | BPPV | Vestibular Neuritis | Ménière's Disease | Vestibular Migraine | Central Vertigo |
|---|---|---|---|---|---|
| Head Impulse Test | Normal | Abnormal (gain <0.6) | Variable | Normal | Normal |
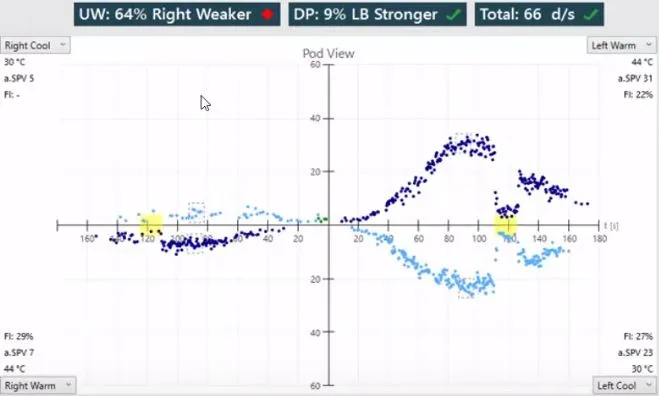
| Caloric Response | Normal | Reduced >25% | Variable | Normal | Variable |
| VEMP Thresholds | Normal | Normal | Elevated/absent | Normal | Variable |
| Audiometry | Normal | Normal | Low-freq loss | Normal | Variable |
| VNG Findings | Positional nystagmus | Unilateral weakness | Variable | Normal | Central patterns |
- Latency: 1-3 seconds after position change (never immediate)
- Duration: 15-60 seconds maximum (never >2 minutes)
- Intensity: Crescendo-decrescendo pattern with peak at 5-10 seconds
- Fatigability: Decreases with repetition (pathognomonic for BPPV)
- Direction: Rotatory-upbeating for posterior canal (85% of cases)
📌 Remember: BPPV-QUANT for quantitative discrimination - Quick onset (1-3 sec), Upbeating rotatory, Amplitude crescendo-decrescendo, Nystagmus <60 seconds, Tires with repetition. These five quantitative features distinguish BPPV from central positional vertigo with >95% accuracy.
Vestibular Neuritis Discrimination relies on unilateral vestibular hypofunction markers:
- Head Impulse Test: Gain <0.6 with overt catch-up saccades
- Caloric Testing: >25% canal paresis on affected side
- Spontaneous Nystagmus: Horizontal, unidirectional, suppressed by fixation
- Hearing Preservation: Normal audiometry (distinguishes from labyrinthitis)
- Recovery Pattern: Gradual improvement over 6-12 weeks
Ménière's Disease Discrimination requires fluctuating pattern documentation:
-
Hearing Fluctuation: >10 dB variation in low frequencies between attacks
-
Glycerol Test: Temporary hearing improvement in 60-70% of cases
-
Electrocochleography: Elevated SP/AP ratio >0.37 (normal <0.24)
-
Attack Pattern: Spontaneous episodes with complete recovery between
-
Progression: Increasing hearing loss with decreasing vertigo over years
-
Early Stage Ménière's (0-2 years)
- Fluctuating hearing loss 10-40 dB
- Severe vertigo attacks lasting 1-6 hours
- Tinnitus low-pitched, fluctuating
- Aural fullness precedes attacks in 80%
- Normal hearing between episodes
-
Intermediate Stage (2-10 years)
- Progressive hearing loss 40-70 dB
- Variable vertigo intensity and frequency
- Persistent tinnitus with recruitment
- Drop attacks (Tumarkin's) in 15%
- Bilateral involvement in 30-40%
-
Late Stage (>10 years)
- Severe hearing loss >70 dB
- Minimal vertigo ("burned out" labyrinth)
- Constant tinnitus and imbalance
- Bilateral disease in 50%
- Functional disability predominates
Vestibular Migraine Discrimination uses associated symptom patterns:
- Migraine History: Personal (80%) or family history (60%)
- Trigger Correlation: Stress, hormonal, dietary triggers match migraine
- Associated Symptoms: Photophobia (70%), phonophobia (60%), aura (30%)
- Treatment Response: Improvement with migraine prophylaxis confirms diagnosis
- Vestibular Testing: Normal between episodes (distinguishes from peripheral causes)
⭐ Clinical Pearl: Central vertigo discrimination requires high suspicion in patients with normal head impulse tests during acute vertigo. The "INFARCT" criteria identify stroke risk: Impulse normal, Nystagmus direction-changing, Focal neurological signs, Age >65, Risk factors present, Continuous symptoms, Timing sudden onset. ≥3 criteria mandate immediate MRI.
Treatment Response Discrimination provides retrospective confirmation:
- BPPV: >90% improvement with canalith repositioning within 1-3 sessions
- Vestibular Neuritis: Gradual improvement with vestibular rehabilitation over 6-12 weeks
- Ménière's Disease: Variable response to dietary modification and diuretics
- Vestibular Migraine: 60-80% improvement with migraine prophylaxis within 3 months
- Central Vertigo: Limited improvement without specific neurological treatment
💡 Master This: Effective differential diagnosis combines temporal pattern recognition with objective testing confirmation and treatment response validation. The "3-Test Rule" provides >95% diagnostic accuracy: Head impulse test (peripheral vs central), Dix-Hallpike test (BPPV vs other), and audiometry (Ménière's vs other). This systematic approach prevents diagnostic anchoring and ensures appropriate treatment selection.
The discrimination matrix succeeds because vestibular disorders follow predictable pathophysiological patterns that create quantifiable differences in timing, triggers, testing results, and treatment responses. Mastering these quantitative thresholds transforms vestibular diagnosis from subjective interpretation to objective analysis.
🔬 Differential Diagnosis Architecture: The Systematic Discrimination Matrix
⚕️ Treatment Algorithm Mastery: Evidence-Based Intervention Pathways
Treatment algorithms operate through condition-specific pathways with quantitative success metrics and defined escalation criteria. This systematic approach achieves 85-95% treatment success across vestibular disorders, compared to 40-60% success with empirical treatment approaches.
BPPV Treatment Algorithm achieves >90% success with systematic repositioning protocols:
Primary Treatment Protocol:
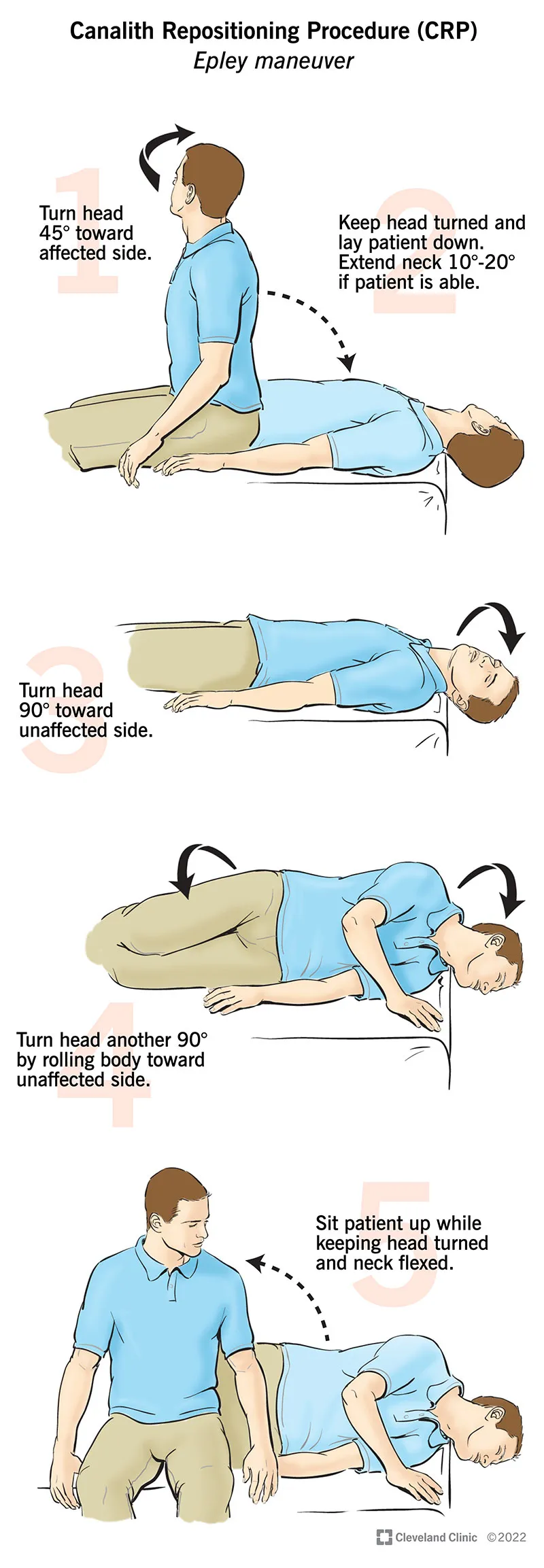
- Posterior Canal BPPV (85% of cases): Epley maneuver
- Success rate: 90-95% with single treatment
- Technique precision: Hold each position 60 seconds minimum
- Post-treatment: 48-hour activity restrictions improve success by 15%
- Horizontal Canal BPPV (10% of cases): Barbecue roll maneuver
- Success rate: 85-90% with 1-2 treatments
- Direction determination: Geotropic vs apogeotropic nystagmus
- Alternative: Gufoni maneuver for apogeotropic variant
Escalation Protocol for Treatment Failures:
- Repeat repositioning after 1 week if symptoms persist
- Alternative maneuvers: Semont or modified Epley techniques
- Surgical intervention: Posterior canal occlusion for refractory cases (<5%)
- Indication: >6 failed repositioning attempts over 6 months
- Success rate: >95% symptom resolution
- Risk: Mild hearing loss in 10-15% of cases
Vestibular Neuritis Treatment Algorithm combines acute management with rehabilitation protocols:
Acute Phase Treatment (0-72 hours):
- Corticosteroids: Prednisone 1mg/kg (max 80mg) for 7 days, then taper
- Evidence: Improves recovery by 40% if started within 72 hours
- Mechanism: Reduces inflammation and promotes neural recovery
- Contraindications: Diabetes, hypertension, psychiatric history
- Symptomatic Relief: Meclizine 25mg TID for maximum 3 days
- Rationale: Longer use delays central compensation
- Alternative: Diazepam 2-5mg for severe symptoms
Subacute Phase Treatment (3 days-6 weeks):
- Early Mobilization: Begin within 48-72 hours of symptom onset
- Vestibular Rehabilitation: Customized exercise program
- Gaze stabilization: VOR adaptation exercises
- Balance training: Progressive difficulty levels
- Habituation: Motion exposure therapy
- Success Metrics: 70-80% functional recovery within 6-12 weeks
| Treatment Phase | Duration | Primary Intervention | Success Rate | Key Metrics |
|---|---|---|---|---|
| Acute | 0-72 hours | Corticosteroids + rest | 40% improvement | Symptom severity reduction |
| Subacute | 3 days-6 weeks | VRT + mobilization | 70-80% recovery | Functional improvement |
| Chronic | >6 weeks | Intensive VRT | 60% improvement | Quality of life measures |
| Refractory | >6 months | Multidisciplinary care | Variable | Disability reduction |
Conservative Management (First-line, 60-70% response):
- Dietary Modification: Low sodium (<2g/day), caffeine restriction
- Evidence: Reduces attack frequency by 30-40%
- Mechanism: Decreases endolymphatic pressure
- Diuretics: Hydrochlorothiazide 25mg daily or acetazolamide 250mg BID
- Success rate: 40-60% reduction in attack frequency
- Monitoring: Electrolytes, kidney function every 3 months
Medical Escalation (Second-line):
- Betahistine: 16mg TID (not FDA approved in US)
- Evidence: Modest benefit in European studies
- Mechanism: Histamine H3 antagonist, improves microcirculation
- Intratympanic Steroids: Dexamethasone 4mg/ml
- Success rate: 60-70% vertigo control
- Procedure: 3 injections over 6 weeks
- Advantage: Preserves hearing compared to gentamicin
Surgical Intervention (Third-line, refractory cases):
- Intratympanic Gentamicin: Chemical labyrinthectomy
- Indication: Failed medical management, serviceable hearing
- Success rate: 85-95% vertigo control
- Risk: Hearing loss in 15-30% of patients
- Endolymphatic Sac Surgery: Decompression or shunt placement
- Success rate: 60-70% (controversial efficacy)
- Advantage: Hearing preservation
- Labyrinthectomy: Complete vestibular ablation
- Indication: Non-serviceable hearing, failed other treatments
- Success rate: >95% vertigo control
- Consequence: Complete hearing loss on treated side
⭐ Clinical Pearl: Intratympanic gentamicin requires careful titration with low-dose protocols to minimize hearing loss. The "gentamicin holiday" approach uses single injection followed by 4-6 week observation before additional doses. This reduces hearing loss risk from 30% to <15% while maintaining >90% vertigo control.
Vestibular Migraine Treatment Algorithm mirrors migraine prophylaxis protocols:
Acute Treatment:
- Triptans: Sumatriptan 50-100mg for acute episodes
- Antiemetics: Ondansetron 4-8mg for nausea control
Prophylactic Treatment (>2 episodes/month):
- First-line: Propranolol 80-160mg daily or topiramate 50-100mg daily
- Success rate: 60-80% reduction in episode frequency
- Monitoring: Blood pressure, weight, cognitive function
- Second-line: Amitriptyline 25-75mg or venlafaxine 75-150mg
- Lifestyle Modifications: Trigger avoidance, sleep hygiene, stress management
💡 Master This: Treatment success depends on accurate diagnosis and appropriate algorithm selection. The "Treatment Response Rule" guides management: >50% improvement within expected timeframe confirms diagnosis and continues current therapy, while <25% improvement suggests diagnostic reconsideration or algorithm escalation. Quantitative outcome measures (DHI scores, attack frequency, functional assessments) provide objective treatment monitoring and guide therapeutic decisions.
Treatment algorithms succeed through systematic application of evidence-based interventions with defined success criteria and escalation pathways. This approach maximizes therapeutic outcomes while minimizing unnecessary interventions and treatment delays.
⚕️ Treatment Algorithm Mastery: Evidence-Based Intervention Pathways
🧠 Multi-System Integration Hub: The Vestibular-Neural Network Convergence

Vestibular integration occurs through five major neural networks: vestibulo-ocular, vestibulo-spinal, vestibulo-autonomic, vestibulo-cognitive, and vestibulo-emotional pathways. These networks process information within 10-50 milliseconds, creating real-time spatial awareness and coordinated motor responses essential for survival and function.
Vestibulo-Ocular Integration maintains visual stability during head movement through precise neural computation:
- Direct VOR pathway: 3-neuron arc with 16-millisecond latency
- Primary neurons: Vestibular ganglion cells
- Secondary neurons: Vestibular nuclei (medial, superior)
- Motor neurons: Oculomotor, trochlear, abducens nuclei
- VOR gain calibration: Cerebellar modulation maintains gain = 1.0
- Flocculus: Short-term adaptation (minutes to hours)
- Nodulus: Long-term plasticity (days to weeks)
- Paraflocculus: Context-dependent modulation
Vestibulo-Spinal Integration coordinates postural responses through multiple descending pathways:
- Lateral vestibulospinal tract: Ipsilateral excitation of extensor muscles
- Origin: Lateral vestibular nucleus (Deiters' nucleus)
- Function: Anti-gravity support, limb extension
- Latency: 25-30 milliseconds for postural responses
- Medial vestibulospinal tract: Bilateral neck muscle control
- Origin: Medial vestibular nucleus
- Function: Head stabilization, cervico-collic reflexes
- Integration: Coordinates with cervical proprioception
Advanced Vestibulo-Cognitive Integration reveals cutting-edge understanding:
- Spatial Navigation Networks: Hippocampal place cells receive vestibular input
- Grid cells: Entorhinal cortex integrates path integration signals
- Head direction cells: Anterior thalamus processes compass information
- Border cells: Subicular complex defines spatial boundaries
- Vestibular Cortical Areas: Multiple cortical regions process vestibular information
- PIVC (Parieto-Insular Vestibular Cortex): Primary vestibular cortex
- Posterior parietal cortex: Spatial reference frame integration
- Temporal cortex: Vestibular memory and learning
Vestibulo-Autonomic Integration controls cardiovascular and respiratory responses:
- Orthostatic Responses: Vestibular-sympathetic reflexes maintain blood pressure
- Otolith input: Linear acceleration detection during postural changes
- Sympathetic activation: Vasoconstriction and heart rate increase
- Response time: 2-3 seconds for cardiovascular adjustment
- Motion Sickness Pathways: Vestibular-vagal connections trigger nausea
- Area postrema: Chemoreceptor trigger zone integration
- Nucleus tractus solitarius: Autonomic coordination center
- Dorsal motor nucleus: Vagal efferent control
| Integration Network | Primary Function | Response Time | Clinical Relevance | Dysfunction Effects |
|---|---|---|---|---|
| Vestibulo-Ocular | Gaze stabilization | 16 ms | Oscillopsia, diplopia | Visual instability |
| Vestibulo-Spinal | Postural control | 25-30 ms | Falls, imbalance | Postural instability |
| Vestibulo-Autonomic | Cardiovascular control | 2-3 seconds | Orthostatic symptoms | Syncope, presyncope |
| Vestibulo-Cognitive | Spatial navigation | 50-100 ms | Disorientation | Spatial memory loss |
| Vestibulo-Emotional | Anxiety responses | Variable | Panic, agoraphobia | Psychiatric symptoms |
- Synaptic Plasticity: LTP/LTD in vestibular nuclei and cerebellum
- NMDA receptors: Activity-dependent synaptic strengthening
- Calcium signaling: Intracellular cascades modify synaptic efficacy
- Time course: Minutes to hours for initial adaptation
- Structural Plasticity: Dendritic remodeling and synaptogenesis
- BDNF expression: Brain-derived neurotrophic factor promotes growth
- Glial activation: Microglial and astrocytic support for remodeling
- Time course: Days to weeks for structural changes
📌 Remember: SPACE for vestibular integration - Spatial navigation (hippocampus), Postural control (spinal), Autonomic responses (brainstem), Cortical processing (parietal), Eye movements (oculomotor). Each network operates at different time scales from milliseconds (reflexes) to minutes (adaptation) to weeks (compensation).
Clinical Integration Disorders result from network dysfunction:
- Persistent Postural-Perceptual Dizziness (PPPD): Maladaptive plasticity
- Prevalence: 15-20% of vestibular patients develop chronic symptoms
- Mechanism: Excessive reliance on visual and proprioceptive inputs
- Treatment: Vestibular rehabilitation with cognitive behavioral therapy
- Vestibular Paroxysmia: Vascular compression of 8th cranial nerve
- Symptoms: Brief episodes (seconds) of vertigo and hearing loss
- Diagnosis: MRI shows neurovascular contact in 60-80% of cases
- Treatment: Carbamazepine or microvascular decompression
⭐ Clinical Pearl: Vestibular rehabilitation success depends on network-specific training. Gaze stabilization exercises target VOR adaptation, balance training enhances vestibulo-spinal integration, and habituation exercises promote central compensation. Customized protocols based on specific network deficits achieve 85-90% success compared to 60-70% with generic exercises.
Emerging Integration Concepts reveal novel therapeutic targets:
- Vestibular-Immune Interactions: Inflammatory mediators affect vestibular function
- Cytokines: IL-1β, TNF-α modulate vestibular neuron excitability
- Autoimmune vestibular disorders: Cogan's syndrome, AIED
- Treatment implications: Immunosuppressive therapy for refractory cases
- Vestibular-Circadian Connections: Temporal processing of vestibular signals
- Suprachiasmatic nucleus: Circadian modulation of vestibular sensitivity
- Clinical relevance: Time-of-day effects on vestibular symptoms
- Therapeutic potential: Chronotherapy for vestibular disorders
💡 Master This: Vestibular integration represents convergent evolution of multiple neural networks optimized for survival advantage. Understanding network-specific dysfunction guides targeted interventions and predicts treatment outcomes. The "Integration Principle" states that vestibular symptoms reflect network-wide dysfunction, not just peripheral damage, requiring comprehensive rehabilitation addressing all affected systems for optimal recovery.
Multi-system integration mastery transforms vestibular medicine from organ-specific treatment to network-based therapy, achieving superior outcomes through systems-level understanding of vestibular function and dysfunction.
🧠 Multi-System Integration Hub: The Vestibular-Neural Network Convergence
🎯 Clinical Mastery Arsenal: Rapid-Fire Diagnostic & Treatment Protocols
The Vestibular Rapid Assessment Protocol (V-RAP) enables complete evaluation within 5 minutes:
60-Second History Screen:
- "TIMING" questions: When (onset), How long (duration), How often (frequency)
- "TRIGGERS" assessment: Position, movement, stress, spontaneous
- "ASSOCIATED" symptoms: Hearing, neurological, headache, autonomic
3-Minute Physical Examination:
- HINTS examination (90 seconds): Head impulse, nystagmus, skew deviation
- Dix-Hallpike test (60 seconds): Posterior canal BPPV screening
- Romberg testing (30 seconds): Proprioceptive vs vestibular dysfunction
📌 Remember: V-HINTS-D for rapid assessment - Vital signs, Head impulse, Instant nystagmus, Neurological signs, Test of skew, Spontaneous symptoms, Dix-Hallpike. This 7-step protocol achieves >90% diagnostic accuracy in <5 minutes.
Essential Clinical Thresholds for instant decision-making:
| Parameter | Normal | Abnormal | Critical | Action Required |
|---|---|---|---|---|
| VOR Gain | 0.8-1.2 | <0.6 or >1.4 | <0.3 | Immediate VRT |
| Caloric Response | 6-50°/sec | <6 or >50 | Absent | Neuroimaging |
| Episode Duration | Variable | >24 hours | Continuous | Emergency eval |
| Hearing Loss | <20 dB | >25 dB | Sudden onset | Urgent ENT |
| Nystagmus Velocity | <5°/sec | >10°/sec | >30°/sec | Central concern |
BPPV Protocol (2-minute deployment):
- Confirm diagnosis: Dix-Hallpike positive with typical nystagmus
- Execute Epley: 4 positions, 60 seconds each, smooth transitions
- Success criteria: Symptom resolution + negative repeat Dix-Hallpike
- Follow-up: 48-hour restrictions, return if symptoms persist
Acute Vertigo Protocol (5-minute assessment):
- HINTS examination: Differentiate peripheral vs central
- If peripheral: Symptomatic treatment + early mobilization
- If central: Immediate neuroimaging + neurology consultation
- Red flags: Normal head impulse + acute vertigo = stroke until proven otherwise
Master Clinician Decision Trees:
The "Rule of 3s" for Vestibular Urgency:
- 3 seconds: Normal head impulse in acute vertigo = central lesion
- 3 minutes: Maximum BPPV episode duration (longer suggests other causes)
- 3 days: Peak vestibular neuritis symptoms (improvement expected after)
- 3 weeks: Expected compensation timeline (refer if no improvement)
- 3 months: Chronic vestibular syndrome threshold (comprehensive evaluation)
High-Yield Clinical Pearls for instant expertise:
⭐ Pearl 1: Spontaneous nystagmus that changes direction with gaze indicates central pathology with >95% specificity. Peripheral nystagmus is unidirectional and suppressed by fixation.
⭐ Pearl 2: BPPV in patients >65 years has 40% recurrence rate within 2 years. Vitamin D deficiency increases recurrence risk by 300% - check 25-OH vitamin D levels.
⭐ Pearl 3: Vestibular migraine responds to magnesium supplementation (400mg daily) in 60% of patients, providing non-pharmacological prophylaxis option.
Rapid Medication Protocols:
Acute Symptom Control (choose one):
- Meclizine 25mg: Mild-moderate symptoms, minimal sedation
- Diazepam 2-5mg: Severe symptoms, short-term use only (<3 days)
- Ondansetron 4-8mg: Nausea predominant, no vestibular suppression
Chronic Management Algorithms:
- Ménière's disease: Low sodium (<2g/day) + HCTZ 25mg daily
- Vestibular migraine: Propranolol 80-160mg or topiramate 50-100mg daily
- Persistent dizziness: VRT + CBT combination therapy
Emergency Red Flags requiring immediate action:
- Sudden hearing loss + vertigo = AICA stroke (MRI within 6 hours)
- Vertical nystagmus = Central lesion (neuroimaging immediately)
- Diplopia + vertigo = Brainstem pathology (emergency neurology)
- Severe headache + vertigo = Posterior fossa lesion (urgent imaging)
💡 Master This: Clinical mastery combines pattern recognition speed with protocol precision. The "5-Minute Rule" states that experienced clinicians should identify disorder category within 5 minutes and initiate appropriate treatment within 10 minutes. This efficiency comes from systematic approach + threshold memorization + protocol automation through deliberate practice.
Quality Metrics for Vestibular Mastery:
- Diagnostic accuracy: >90% with initial assessment
- Treatment success: >85% symptom improvement within expected timeframes
- Patient satisfaction: >95% understanding of diagnosis and treatment plan
- Efficiency: Complete evaluation within 15 minutes for routine cases
- Safety: Zero missed central lesions through systematic HINTS application
The clinical mastery arsenal transforms vestibular medicine from time-consuming uncertainty to rapid-fire precision, enabling optimal patient outcomes through systematic excellence and evidence-based protocols.
🎯 Clinical Mastery Arsenal: Rapid-Fire Diagnostic & Treatment Protocols
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more
Have doubts about this lesson?
Ask Rezzy, your AI Study Mate, to explain anything you didn't understand
Everything you need for NEET-PG prep
Get full Oncourse access with lessons, practice questions, flashcards and AI study tools.
Scan to download app












