Psoriasis

On this page
🔬 Psoriasis Unveiled: The Autoimmune Skin Revolution
Psoriasis transforms skin into a battlefield where your own immune system attacks keratinocytes, creating the distinctive silvery plaques that affect 2-3% of the population-but recognizing these lesions is just the beginning. You'll master the pathophysiology driving this chronic inflammatory disease, build systematic approaches to distinguish it from mimics like eczema and fungal infections, and command an expanding therapeutic arsenal from topical agents to biologics. Beyond skin-deep thinking, you'll connect psoriatic disease to cardiovascular risk, metabolic syndrome, and joint destruction, learning when to escalate treatment and how to tailor therapy to disease severity and patient factors.
📌 Remember: PSORIASIS - Plaques with Silvery scales, Oval/round lesions, Red base, Itchy (sometimes), Asymmetric distribution, Symmetric bilateral pattern, Immune-mediated, Systemic associations
Epidemiological Foundation
- Global prevalence: 2-3% of world population
- Bimodal age distribution:
- Early onset: 16-22 years (75% of cases)
- Late onset: 57-60 years (25% of cases)
- Gender distribution: Equal male-to-female ratio
- Genetic component: 30-40% concordance in identical twins
- HLA associations: HLA-Cw6 (strongest association, OR 13.3)

| Psoriasis Type | Prevalence | Age of Onset | Genetic Association | Prognosis | Systemic Risk |
|---|---|---|---|---|---|
| Plaque (Vulgaris) | 85-90% | 20-30 years | HLA-Cw6 positive | Chronic, stable | Moderate CVD risk |
| Guttate | 8-10% | 16-25 years | HLA-Cw6 strong | Often self-limiting | Low systemic risk |
| Pustular | 1-2% | 40-60 years | IL36RN mutations | Severe, episodic | High systemic risk |
| Erythrodermic | 1-3% | 50-60 years | Variable | Life-threatening | Highest mortality |
| Inverse | 3-7% | 40-50 years | Weak HLA association | Chronic, difficult | Moderate risk |
Pathophysiological Architecture
The psoriatic process involves three interconnected pathways:
-
Innate immune activation
- Plasmacytoid dendritic cells produce excessive IFN-α
- Antimicrobial peptides (LL-37, β-defensins) trigger TLR activation
- Neutrophil infiltration creates Munro microabscesses
-
Adaptive immune dysregulation
- Th1/Th17 cell predominance with IL-17A overproduction
- TNF-α levels increase 5-10 fold in lesional skin
- IL-23/IL-17 axis drives chronic inflammation
-
Keratinocyte hyperproliferation
- Ki-67 positivity increases from <5% to >30%
- Epidermal thickness increases 3-5 fold
- Parakeratosis with retained nuclei in stratum corneum

💡 Master This: The IL-23/IL-17 axis represents the central inflammatory pathway in psoriasis, explaining why IL-23 inhibitors (guselkumab, risankizumab) achieve PASI 90 responses in >70% of patients by week 16.
Understanding this autoimmune foundation reveals why psoriasis extends beyond skin manifestations, setting the stage for exploring the clinical presentation patterns that enable rapid diagnostic recognition and severity assessment frameworks that guide therapeutic decision-making.
🔬 Psoriasis Unveiled: The Autoimmune Skin Revolution
🎯 Clinical Recognition Mastery: The Diagnostic Arsenal

Plaque Psoriasis: The Classic Presentation
Plaque psoriasis accounts for 85-90% of cases, presenting with well-demarcated erythematous plaques topped by thick, silvery-white scales. These lesions demonstrate remarkable consistency in morphology while showing predictable distribution patterns.
📌 Remember: PLAQUE features - Predilection for extensors, Large well-demarcated lesions, Auspitz sign positive, Quite symmetric, Uniform silvery scales, Erythematous base
-
Morphological characteristics:
- Size range: 0.5-30 cm diameter
- Scale thickness: 2-5 mm in chronic lesions
- Erythema intensity: Moderate to severe (salmon-pink to deep red)
- Border definition: Sharp, well-demarcated edges
-
Distribution patterns:
- Extensor surfaces: Elbows (90%), knees (85%)
- Scalp involvement: 80% of patients
- Sacral region: 70% of patients
- Bilateral symmetry: >95% of cases
Diagnostic Signs and Phenomena
| Clinical Sign | Technique | Positive Response | Sensitivity | Specificity | Clinical Significance |
|---|---|---|---|---|---|
| Auspitz Sign | Scale removal | Punctate bleeding | 85-90% | 95% | Pathognomonic for psoriasis |
| Candle Wax Sign | Gentle scraping | Waxy scale removal | 90-95% | 80% | Early diagnostic clue |
| Woronoff Ring | Visual inspection | Pale halo around lesion | 60-70% | 90% | Indicates treatment response |
| Koebner Phenomenon | Trauma history | New lesions at injury sites | 25-30% | 95% | Suggests active disease |
| Nail Pitting | Nail examination | Punctate depressions | 50-80% | 85% | Predicts arthritis risk |
Guttate Psoriasis: The Raindrop Pattern
Guttate psoriasis presents as multiple small, drop-like lesions scattered across the trunk and proximal extremities. This variant shows strong association with streptococcal infections and younger age groups.
-
Clinical characteristics:
- Lesion size: 2-10 mm diameter
- Number: Dozens to hundreds of lesions
- Distribution: Trunk (90%), proximal limbs (80%)
- Scale: Thin, minimal compared to plaque type
-
Triggering factors:
- Streptococcal pharyngitis: 80% of cases
- Viral infections: 15% of cases
- Stress/trauma: 10% of cases
- Medication triggers: 5% of cases
💡 Master This: Guttate psoriasis following streptococcal infection shows spontaneous resolution in 60-70% of cases within 3-4 months, but 30-40% progress to chronic plaque psoriasis requiring long-term management.
Severity Assessment Framework
-
Mild psoriasis (70% of patients):
- BSA <3% or PASI <10
- Localized plaques on elbows, knees, scalp
- Minimal functional impairment
- DLQI score <10
-
Moderate psoriasis (20% of patients):
- BSA 3-10% or PASI 10-20
- Multiple body regions involved
- Moderate functional impact
- DLQI score 10-20
-
Severe psoriasis (10% of patients):
- BSA >10% or PASI >20
- Extensive involvement or special sites
- Significant disability
- DLQI score >20
⭐ Clinical Pearl: Special site involvement (face, hands, feet, genitals) automatically qualifies as moderate-to-severe psoriasis regardless of BSA percentage, due to disproportionate functional and psychological impact.
This clinical recognition mastery provides the foundation for understanding pathophysiological mechanisms that drive these diverse presentations and therapeutic approaches that target specific inflammatory pathways for optimal patient outcomes.
🎯 Clinical Recognition Mastery: The Diagnostic Arsenal
🔧 Therapeutic Strategy Engine: Evidence-Based Treatment Algorithms
Treatment Algorithm Framework
Topical Therapy Arsenal
Topical corticosteroids remain the first-line treatment for mild-to-moderate psoriasis, with response rates varying by potency class and anatomical location.
| Potency Class | Representative Agent | Efficacy (PASI 75) | Application Sites | Maximum Duration | Key Limitations |
|---|---|---|---|---|---|
| Class I (Ultra-high) | Clobetasol 0.05% | 85-90% | Body, scalp | 2-4 weeks | Skin atrophy risk |
| Class II (High) | Betamethasone 0.1% | 75-80% | Body, extremities | 4-6 weeks | Moderate atrophy |
| Class III (Medium-high) | Triamcinolone 0.1% | 65-70% | Face, inframammary | 6-8 weeks | Mild atrophy |
| Class IV (Medium) | Hydrocortisone 0.1% | 50-60% | Face, genitals | 12+ weeks | Limited efficacy |
- Vitamin D analogs effectiveness:
- Calcipotriol: PASI 75 in 60-65% at 12 weeks
- Calcitriol: PASI 75 in 55-60% at 12 weeks
- Tacalcitol: PASI 75 in 50-55% at 12 weeks
- Combination therapy (calcipotriol + betamethasone): PASI 75 in 75-80%

Phototherapy Protocols
Narrowband UV-B (NB-UVB) represents the gold standard phototherapy with superior efficacy and safety profile compared to broadband UV-B or PUVA.
-
NB-UVB treatment parameters:
- Initial dose: 70% of minimal erythema dose (MED)
- Dose escalation: 10-20% increase per session
- Treatment frequency: 3 times weekly
- Total sessions: 25-30 for optimal response
- PASI 75 achievement: 75-80% of patients
-
Response timeline:
- Initial improvement: Week 4-6
- Significant clearing: Week 8-12
- Maintenance phase: Once weekly for 3-6 months
- Relapse prevention: Monthly treatments long-term
⭐ Clinical Pearl: NB-UVB achieves PASI 75 in 75-80% of patients with guttate psoriasis within 8-10 weeks, making it the preferred first-line therapy for this variant over systemic agents.
Systemic Therapy Selection
Conventional systemic agents provide cost-effective options for moderate-to-severe psoriasis with established safety profiles and monitoring protocols.
-
Methotrexate dosing strategy:
- Starting dose: 7.5-10 mg weekly
- Dose escalation: 2.5-5 mg every 4 weeks
- Maximum dose: 25 mg weekly
- Folic acid supplementation: 5 mg daily (except MTX day)
- PASI 75 achievement: 60-70% at 16 weeks
-
Cyclosporine protocols:
- Initial dose: 2.5-3 mg/kg/day divided BID
- Maximum dose: 5 mg/kg/day
- Treatment duration: Maximum 12-24 months
- PASI 75 achievement: 70-80% at 12 weeks
- Rapid onset: Improvement within 2-4 weeks
💡 Master This: Methotrexate requires comprehensive monitoring including CBC, LFTs, creatinine every 4-8 weeks initially, then every 12 weeks once stable. Hepatic fibrosis risk increases with cumulative doses >1.5-2 grams.
Biologic Therapy Revolution
Biologic agents have transformed psoriasis management with unprecedented efficacy and targeted mechanisms addressing specific inflammatory pathways.
-
TNF-α inhibitor efficacy (16-week PASI 75 rates):
- Adalimumab: 75-80%
- Etanercept: 45-50%
- Infliximab: 80-85%
- Certolizumab: 75-80%
-
IL-17 inhibitor superiority (16-week PASI 90 rates):
- Secukinumab: 70-75%
- Ixekizumab: 80-85%
- Brodalumab: 75-80%
-
IL-23 inhibitor excellence (16-week PASI 90 rates):
- Guselkumab: 75-80%
- Risankizumab: 85-90%
- Tildrakizumab: 65-70%
This therapeutic mastery framework enables precision medicine approaches that optimize individual patient outcomes while minimizing treatment-related risks and healthcare costs through evidence-based decision algorithms.
🔧 Therapeutic Strategy Engine: Evidence-Based Treatment Algorithms
🔍 Differential Diagnosis Matrix: Systematic Disease Discrimination
Primary Differential Considerations
Seborrheic dermatitis represents the most common diagnostic challenge, particularly in scalp and facial presentations where clinical overlap creates diagnostic uncertainty.
| Feature | Psoriasis | Seborrheic Dermatitis | Atopic Dermatitis | Lichen Planus | Cutaneous Lupus |
|---|---|---|---|---|---|
| Scale Character | Thick, silvery | Greasy, yellowish | Fine, dry | Minimal scaling | Adherent, thick |
| Border Definition | Sharp, well-demarcated | Ill-defined, blurred | Poorly defined | Well-demarcated | Well-demarcated |
| Distribution | Extensors, scalp | Seborrheic areas | Flexural areas | Wrists, ankles | Sun-exposed areas |
| Auspitz Sign | Positive (85%) | Negative | Negative | Negative | Negative |
| Pruritus Intensity | Mild-moderate | Mild | Severe | Severe | Minimal |
Scalp Psoriasis Discrimination
Scalp involvement occurs in 80% of psoriasis patients, creating diagnostic challenges with seborrheic dermatitis, tinea capitis, and alopecia areata.
-
Psoriasis scalp features:
- Thick, adherent scales extending beyond hairline
- Well-demarcated plaques with sharp borders
- Hair shaft involvement without hair loss
- Silvery-white scale color
- Bleeding upon scale removal (Auspitz sign)
-
Seborrheic dermatitis scalp features:
- Greasy, yellowish scales confined to scalp
- Ill-defined erythema with diffuse distribution
- Minimal hair involvement
- Easy scale removal without bleeding
- Associated dandruff and facial involvement

⭐ Clinical Pearl: Scalp psoriasis extending >1 cm beyond the hairline occurs in 70% of cases and is pathognomonic for psoriasis, distinguishing it from seborrheic dermatitis which rarely extends beyond hair-bearing areas.
Nail Involvement Patterns
Nail psoriasis affects 50-80% of psoriasis patients and provides crucial diagnostic clues for disease confirmation and arthritis prediction.
-
Nail matrix involvement (proximal nail bed):
- Nail pitting: 1-2 mm punctate depressions
- Leukonychia: White spots or transverse bands
- Red spots in lunula: Dilated capillaries
- Crumbling: Nail plate fragmentation
-
Nail bed involvement (distal nail bed):
- Oil drop sign: Yellow-brown discoloration
- Onycholysis: Nail separation from nail bed
- Subungual hyperkeratosis: Thick debris accumulation
- Splinter hemorrhages: Linear blood streaks
-
Nail fold involvement:
- Paronychia: Chronic inflammation
- Periungual scaling: Psoriatic plaques
Histopathological Discrimination
Histological examination provides definitive diagnosis when clinical features remain ambiguous or atypical presentations occur.
-
Psoriasis histological hallmarks:
- Regular acanthosis with uniform rete ridge elongation
- Parakeratosis with retained nuclei in stratum corneum
- Munro microabscesses: Neutrophil collections in corneal layer
- Spongiform pustules of Kogoj: Neutrophils in spinous layer
- Dilated dermal capillaries with tortuous morphology
- Suprapapillary thinning: Epidermis <3-4 cell layers thick
-
Inflammatory infiltrate characteristics:
- Perivascular lymphocytic infiltrate in superficial dermis
- Neutrophil predominance in acute lesions
- T-cell infiltration with CD4+ and CD8+ populations
- Minimal eosinophils (distinguishes from eczema)
💡 Master This: Munro microabscesses and spongiform pustules of Kogoj are pathognomonic for psoriasis, occurring in >90% of active lesions and distinguishing psoriasis from all other papulosquamous disorders.
Response Pattern Analysis
Therapeutic response patterns provide retrospective diagnostic confirmation and guide ongoing management decisions.
-
Psoriasis treatment responses:
- Topical corticosteroids: Rapid improvement within 2-4 weeks
- Vitamin D analogs: Gradual improvement over 8-12 weeks
- Phototherapy: Sustained improvement with minimal relapse
- Systemic therapy: Dramatic improvement with appropriate agents
-
Seborrheic dermatitis responses:
- Antifungal agents: Rapid improvement within 1-2 weeks
- Low-potency steroids: Quick resolution with frequent recurrence
- Calcineurin inhibitors: Sustained control without atrophy risk
This systematic discrimination framework enables confident diagnosis even in challenging presentations, ensuring appropriate therapy selection and optimal patient outcomes through evidence-based clinical reasoning.
🔍 Differential Diagnosis Matrix: Systematic Disease Discrimination
⚖️ Advanced Management Protocols: Precision Treatment Algorithms
Biologic Selection Algorithm
Biologic Efficacy Comparison Matrix
IL-23 inhibitors demonstrate superior long-term efficacy with extended dosing intervals and excellent safety profiles, representing the current gold standard for moderate-to-severe psoriasis.
| Biologic Class | Representative Agent | PASI 90 (Week 16) | PASI 100 (Week 52) | Dosing Interval | Injection Site Reactions | Serious Infections |
|---|---|---|---|---|---|---|
| IL-23 Inhibitors | Risankizumab | 85-90% | 60-65% | Every 12 weeks | <5% | <1% |
| IL-23 Inhibitors | Guselkumab | 75-80% | 55-60% | Every 8 weeks | <5% | <1% |
| IL-17A Inhibitors | Ixekizumab | 80-85% | 50-55% | Every 12 weeks | 10-15% | 1-2% |
| IL-17A Inhibitors | Secukinumab | 70-75% | 45-50% | Every 12 weeks | 10-15% | 1-2% |
| TNF Inhibitors | Adalimumab | 50-55% | 30-35% | Every 2 weeks | 15-20% | 2-3% |
Treatment Optimization Strategies
Combination therapy approaches enhance treatment efficacy while minimizing individual drug toxicities through synergistic mechanisms and dose-sparing effects.
-
Methotrexate + biologic combinations:
- Adalimumab + MTX: PASI 75 in 85-90% vs 75-80% monotherapy
- Infliximab + MTX: Reduces immunogenicity by 60-70%
- Anti-drug antibody formation: Decreased from 15% to <5%
- Dose optimization: MTX 7.5-15 mg weekly sufficient
-
Topical + systemic synergy:
- Calcipotriol + systemic therapy: Additional 15-20% improvement
- Localized resistant plaques: Intralesional triamcinolone 5-10 mg/mL
- Scalp-specific combinations: Calcipotriol/betamethasone foam
⭐ Clinical Pearl: Methotrexate co-therapy with TNF inhibitors reduces anti-drug antibody formation by 60-70% and extends drug survival by 18-24 months compared to biologic monotherapy.
Monitoring and Safety Protocols
Comprehensive monitoring strategies ensure early detection of treatment-related adverse events while optimizing therapeutic outcomes through systematic assessment protocols.
-
Pre-biologic screening requirements:
- Tuberculosis screening: CXR, IGRA/TST, clinical assessment
- Hepatitis B/C testing: HBsAg, anti-HBc, anti-HCV
- Complete blood count: Baseline values for comparison
- Liver function tests: AST, ALT, bilirubin
- Live vaccine updates: Complete 4 weeks before initiation
-
Ongoing monitoring schedules:
- Months 1-6: CBC, LFTs every 3 months
- Months 6-12: Laboratory monitoring every 6 months
- Year 2+: Annual comprehensive assessment
- Clinical evaluation: Every 3-4 months initially
-
Safety signal recognition:
- Serious infections: Incidence 1-3% across biologic classes
- Malignancy risk: No increased risk with modern biologics
- IBD development: Rare with IL-17/IL-23 inhibitors
- Cardiovascular events: Potential protective effect with IL-23 inhibitors
Treatment Failure Management
Systematic approach to treatment failures involves mechanism-based switching and optimization strategies rather than random therapeutic trials.
-
Primary failure (<50% improvement at 16 weeks):
- Dose optimization: Increase frequency or dose
- Mechanism switching: Different pathway targeting
- Combination therapy: Add complementary agent
- Adherence assessment: Patient education and support
-
Secondary failure (loss of response after initial success):
- Anti-drug antibody testing: If available
- Dose escalation: Shorten intervals or increase dose
- Class switching: Different mechanism preferred
- Immunosuppressive co-therapy: Methotrexate addition
💡 Master This: IL-23 inhibitor failure should prompt switching to IL-17A inhibitors rather than TNF inhibitors, as cross-resistance between IL-23 and TNF pathways is minimal, while IL-17A inhibitors target downstream effectors with >80% response rates in IL-23 failures.
This precision management framework enables individualized treatment strategies that maximize therapeutic success while minimizing adverse events through evidence-based protocols and systematic monitoring approaches.
⚖️ Advanced Management Protocols: Precision Treatment Algorithms
🔗 Systemic Integration Hub: Multi-Organ Disease Connections

Cardiovascular Risk Amplification
Psoriasis patients demonstrate significantly elevated cardiovascular risk through shared inflammatory pathways involving TNF-α, IL-17, and IL-6 that accelerate atherosclerosis and increase thrombotic events.
-
Cardiovascular risk quantification:
- Myocardial infarction risk: Increased 1.5-2.0 fold
- Stroke incidence: Elevated 1.2-1.6 fold
- Cardiovascular mortality: Increased 1.3-1.8 fold
- Severe psoriasis (BSA >10%): Risk amplification 2.5-3.0 fold
- Young patients (<40 years): Highest relative risk increase
-
Mechanistic cardiovascular connections:
- Endothelial dysfunction: IL-17 reduces nitric oxide bioavailability
- Accelerated atherosclerosis: TNF-α promotes foam cell formation
- Prothrombotic state: Elevated fibrinogen and platelet activation
- Insulin resistance: Chronic inflammation impairs glucose metabolism
| Cardiovascular Parameter | Psoriasis Patients | Control Population | Relative Risk | Clinical Significance | Monitoring Frequency |
|---|---|---|---|---|---|
| Coronary Artery Disease | 15-20% | 8-10% | 1.8-2.2 | Screen at diagnosis | Every 2-3 years |
| Hypertension | 45-50% | 25-30% | 1.6-1.8 | Annual BP monitoring | Every visit |
| Diabetes Mellitus | 20-25% | 8-12% | 1.8-2.1 | HbA1c screening | Annually |
| Dyslipidemia | 40-45% | 20-25% | 1.8-2.0 | Lipid panel required | Every 1-2 years |
| Metabolic Syndrome | 35-40% | 15-20% | 2.0-2.5 | Comprehensive assessment | Annually |

Psoriatic Arthritis Integration
Psoriatic arthritis (PsA) develops in 20-30% of psoriasis patients, representing a distinct inflammatory arthropathy with unique clinical patterns and progressive joint destruction if untreated.
-
PsA development timeline:
- Skin-first presentation: 85% of cases
- Joint-first presentation: 10% of cases
- Simultaneous onset: 5% of cases
- Average delay: 7-10 years from skin to joint involvement
- Nail involvement: Strongest predictor (OR 2.5-3.0)
-
Clinical pattern recognition:
- Oligoarticular pattern (<5 joints): 50-60% of cases
- Polyarticular pattern (≥5 joints): 25-30% of cases
- Axial involvement: 20-25% of cases
- Enthesitis: 30-40% of patients
- Dactylitis ("sausage digits"): 15-20% of patients
-
Screening and early detection:
- PEST questionnaire: Sensitivity 92%, specificity 87%
- Nail examination: Critical for risk assessment
- Entheseal ultrasound: Subclinical inflammation detection
- MRI imaging: Early erosive changes before radiographic evidence
⭐ Clinical Pearl: Nail psoriasis increases PsA risk by 2.5-3.0 fold, with nail matrix involvement showing stronger association than nail bed changes. Annual joint screening is mandatory for nail psoriasis patients.
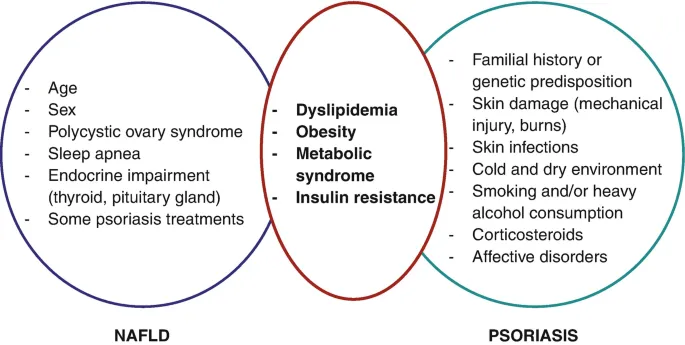
Metabolic Syndrome Constellation
Metabolic dysfunction in psoriasis results from chronic systemic inflammation that disrupts insulin signaling, promotes adipose tissue dysfunction, and alters lipid metabolism.
-
Metabolic parameter alterations:
- Insulin resistance: HOMA-IR elevated 1.5-2.0 fold
- Visceral adiposity: Waist circumference increased 5-8 cm
- Triglyceride elevation: >150 mg/dL in 45-50%
- HDL reduction: <40 mg/dL (men), <50 mg/dL (women) in 40%
- Inflammatory markers: CRP elevated 3-5 fold
-
Weight management strategies:
- Weight loss >5%: Improves PASI scores by 20-30%
- Bariatric surgery: PASI improvement >50% in morbidly obese
- Mediterranean diet: Anti-inflammatory effects with PASI reduction
- Exercise programs: Cardiovascular benefits plus psoriasis improvement
Psychological and Quality of Life Impact
Psoriasis significantly impairs quality of life with psychological morbidity comparable to major medical conditions like diabetes, heart disease, and cancer.
-
Psychological comorbidity prevalence:
- Depression: 15-20% vs 5-8% general population
- Anxiety disorders: 20-25% vs 8-12% general population
- Suicidal ideation: 2.5-3.0 fold increased risk
- Social isolation: 40-50% report significant impairment
- Work productivity: Reduced by 25-30%
-
Quality of life assessment tools:
- DLQI scores: Mean 8-12 (moderate-severe impact)
- Skindex-29: Comprehensive symptom assessment
- PDI (Psoriasis Disability Index): Functional impairment
- PLSI (Psoriasis Life Stress Inventory): Stress quantification
💡 Master This: Biologic therapy not only improves skin clearance but also significantly reduces cardiovascular risk markers, metabolic dysfunction, and psychological morbidity through systemic anti-inflammatory effects, making early aggressive treatment both dermatologically and systemically beneficial.
This systemic integration understanding transforms psoriasis management from isolated skin treatment to comprehensive inflammatory disease management that addresses multiple organ systems and improves overall patient health outcomes.
🔗 Systemic Integration Hub: Multi-Organ Disease Connections
🎯 Clinical Mastery Arsenal: Rapid Assessment and Decision Tools

Rapid Severity Assessment Matrix
PASI (Psoriasis Area and Severity Index) remains the gold standard for clinical trials and treatment decisions, but simplified tools enable rapid clinical assessment without complex calculations.
| Assessment Tool | Time Required | Clinical Utility | Sensitivity | Specificity | Best Application |
|---|---|---|---|---|---|
| BSA (Body Surface Area) | 30 seconds | Treatment eligibility | 85% | 90% | Initial screening |
| PGA (Physician Global Assessment) | 15 seconds | Overall severity | 80% | 85% | Routine monitoring |
| PASI-75/90/100 | 2-3 minutes | Treatment response | 95% | 95% | Clinical trials |
| DLQI (Quality of Life) | 2-3 minutes | Patient impact | 90% | 85% | Comprehensive care |
| NAPSI (Nail Assessment) | 1-2 minutes | Nail involvement | 85% | 90% | Arthritis screening |
Treatment Decision Algorithm
-
Mild psoriasis management (BSA <3%):
- First-line: Topical corticosteroids + vitamin D analogs
- Scalp: Calcipotriol/betamethasone foam or solution
- Face/intertriginous: Calcineurin inhibitors or low-potency steroids
- Maintenance: Vitamin D analogs 3-4 times weekly
- Response timeline: Improvement by week 4-6
-
Moderate psoriasis approach (BSA 3-10%):
- Combination therapy: Topicals + phototherapy
- NB-UVB: 3 times weekly for 8-12 weeks
- Systemic consideration: If topicals + phototherapy fail
- Methotrexate: First-line systemic option
- Monitoring: CBC, LFTs every 4-8 weeks
-
Severe psoriasis strategy (BSA >10%):
- Immediate systemic therapy: Biologics preferred
- IL-23 inhibitors: First-line choice for naive patients
- Rapid control: Cyclosporine for emergency situations
- Combination approach: Systemic + topical for resistant areas
- Target: PASI 90 or complete clearance
Monitoring Excellence Framework
Systematic monitoring protocols ensure optimal treatment outcomes while minimizing adverse events through evidence-based surveillance strategies.
-
Clinical response milestones:
- Week 4: Initial improvement expected with all therapies
- Week 12: Significant response (PASI 75) with topicals
- Week 16: Major response (PASI 90) with biologics
- Week 52: Sustained remission assessment and maintenance planning
-
Laboratory monitoring schedules:
- Methotrexate: CBC, LFTs, creatinine every 4 weeks × 3 months, then every 12 weeks
- Cyclosporine: CBC, comprehensive metabolic panel, BP every 2 weeks × 1 month, then monthly
- Biologics: CBC, LFTs every 3 months × 1 year, then every 6 months
- Infection screening: Annual TB screening, hepatitis monitoring
⭐ Clinical Pearl: PASI 90 achievement by week 16 predicts long-term treatment success with >90% probability of sustained response at 2 years, making this the critical decision timepoint for treatment continuation vs switching.
Emergency Recognition Protocols
Psoriatic emergencies require immediate recognition and aggressive intervention to prevent life-threatening complications.
-
Erythrodermic psoriasis (medical emergency):
- Clinical criteria: >90% BSA involvement with erythema and scaling
- Systemic complications: Hypothermia, fluid loss, high-output heart failure
- Immediate management: Hospitalization, IV fluids, temperature control
- Avoid triggers: Systemic steroid withdrawal, lithium, antimalarials
- Treatment: Cyclosporine 3-5 mg/kg/day or infliximab
-
Generalized pustular psoriasis (life-threatening):
- Clinical features: Widespread sterile pustules with systemic toxicity
- Laboratory findings: Leukocytosis, elevated CRP, hypoalbuminemia
- Complications: Sepsis-like syndrome, organ failure
- Treatment: Immediate systemic therapy with cyclosporine or biologics
- Monitoring: ICU-level care may be required
Patient Education Essentials
-
Disease understanding:
- Chronic condition: Lifelong management required
- Not contagious: Autoimmune disorder, not infectious
- Genetic component: Family history relevant
- Trigger avoidance: Stress, infections, medications
-
Treatment expectations:
- Realistic timelines: Improvement takes weeks to months
- Adherence importance: Consistent use critical for success
- Side effect recognition: When to contact healthcare provider
- Monitoring compliance: Regular follow-up essential
-
Lifestyle modifications:
- Weight management: 5-10% weight loss improves treatment response
- Smoking cessation: Reduces treatment resistance
- Alcohol moderation: Enhances medication safety
- Stress management: Psychological support and relaxation techniques
💡 Master This: Early aggressive treatment achieving PASI 90 within 16 weeks not only optimizes skin clearance but also reduces long-term cardiovascular risk, prevents psoriatic arthritis development, and improves quality of life - making rapid, complete control the therapeutic imperative rather than gradual improvement.
This clinical mastery arsenal provides practical tools for immediate implementation in clinical practice, enabling evidence-based decision-making and optimal patient outcomes through systematic assessment and monitoring protocols.
🎯 Clinical Mastery Arsenal: Rapid Assessment and Decision Tools
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more
Have doubts about this lesson?
Ask Rezzy, your AI Study Mate, to explain anything you didn't understand
Everything you need for NEET-PG prep
Get full Oncourse access with lessons, practice questions, flashcards and AI study tools.
Scan to download app
























