Basic Dermatology

On this page
🔬 Dermatological Foundations: The Skin's Architectural Blueprint
The skin is medicine's most visible organ, yet its diseases demand the same rigor as any internal pathology-you'll master the architectural layers that govern barrier function, decode the cellular renewal cycles that underpin inflammatory and neoplastic conditions, and build a systematic framework for distinguishing mimics through morphology and distribution patterns. This lesson equips you with evidence-based treatment algorithms and reveals how cutaneous findings serve as windows into systemic disease, transforming you from observer into diagnostic detective.
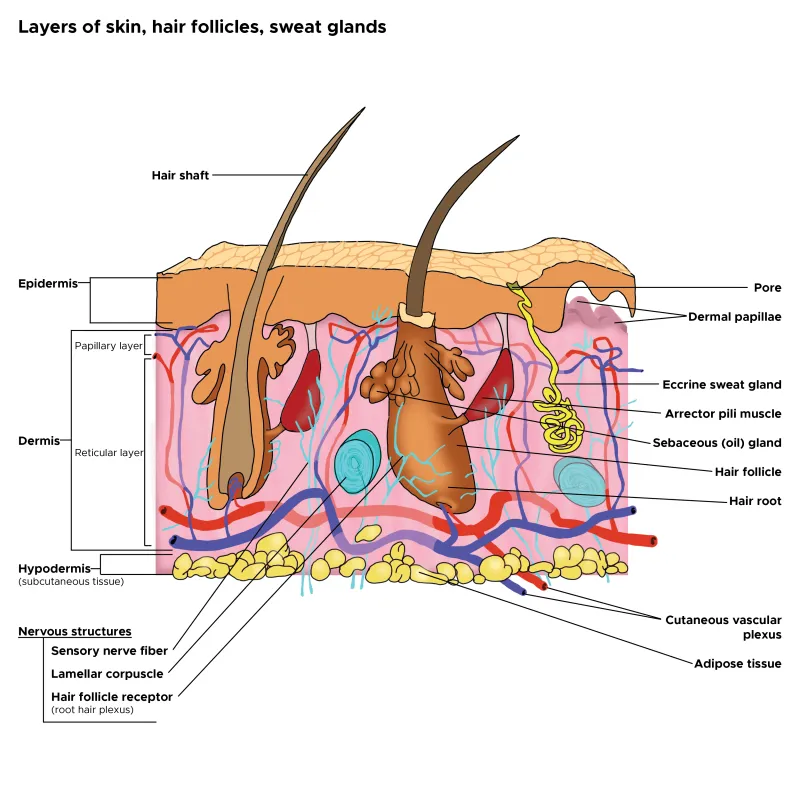
The epidermis consists of 4-5 distinct layers depending on body location, with the stratum corneum containing 15-20 cell layers in thick skin areas like palms and soles. Keratinocytes comprise 95% of epidermal cells, while melanocytes represent only 3-5% but critically determine pigmentation patterns and photoprotection capacity.
📌 Remember: COME Let's Get Sun Burned - Stratum Corneum, Lucidum, Granulosum, Spinosum, Basale (superficial to deep). The stratum lucidum appears only in thick skin (palms/soles), containing 2-3 additional cell layers of anucleate keratinocytes.
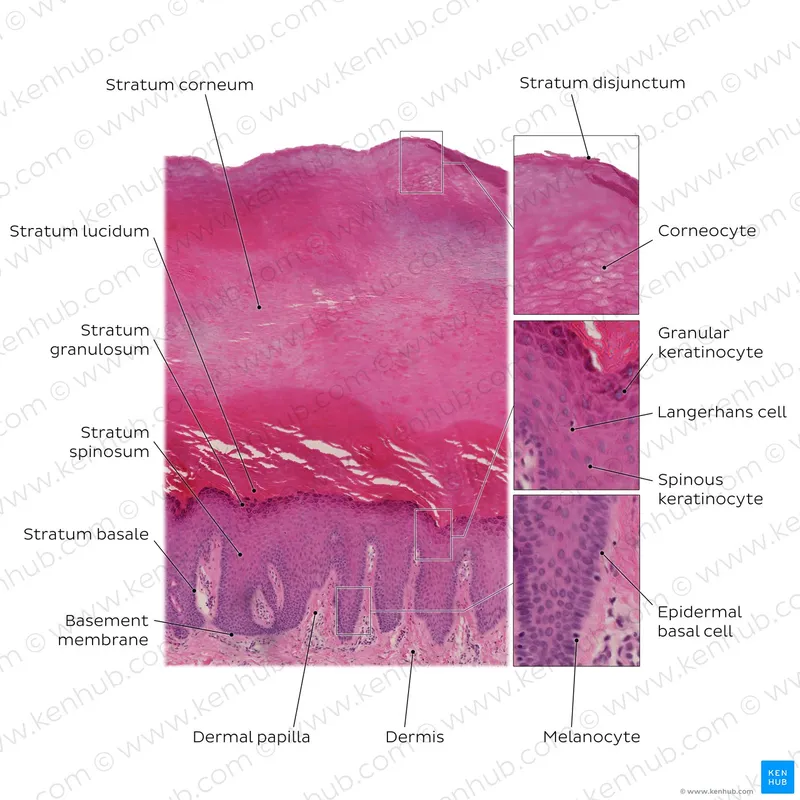
The dermal-epidermal junction creates a basement membrane zone with 4 distinct components: hemidesmosomes, lamina lucida, lamina densa, and sub-basal lamina. This junction amplifies surface area through rete ridges that interdigitate with dermal papillae, increasing adhesion strength by 300-400% compared to flat interfaces.
| Layer | Thickness | Cell Types | Key Functions | Clinical Significance |
|---|---|---|---|---|
| Stratum Corneum | 10-40 μm | Corneocytes | Barrier function | 80% of barrier capacity |
| Stratum Granulosum | 3-5 cell layers | Keratinocytes with keratohyalin | Lipid barrier formation | Filaggrin mutations → atopic dermatitis |
| Stratum Spinosum | 8-10 cell layers | Keratinocytes with desmosomes | Structural integrity | Pemphigus targets desmoglein |
| Stratum Basale | Single cell layer | Keratinocytes, melanocytes | Proliferation, pigmentation | Melanoma origin site |
| Papillary Dermis | 100-300 μm | Fibroblasts, vessels | Nutrition, sensation | Capillary loop patterns |
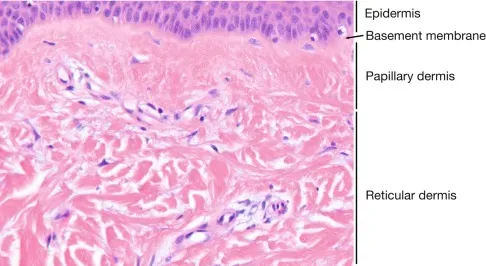
Dermal architecture divides into papillary and reticular layers, with the papillary dermis containing type I collagen (80%) and type III collagen (15%). The reticular dermis provides tensile strength through thick collagen bundles oriented parallel to skin surface, explaining Langer's lines that guide surgical incision placement.
-
Papillary Dermis Architecture:
- Thickness: 100-300 μm (varies by location)
- Collagen: Fine type I and III fibers in loose arrangement
- Vasculature: Superficial plexus at 150-200 μm depth
- Capillary loops extend 100-200 μm into dermal papillae
- Arteriovenous anastomoses enable thermoregulation
- Cellular components: Fibroblasts, mast cells, dendritic cells
-
Reticular Dermis Foundation:
- Thickness: 1-4 mm (location-dependent)
- Collagen: Thick type I bundles in parallel orientation
- Elastic fibers: 2-4% of dermal volume, peak at mid-dermis
- Oxytalan fibers: Superficial, perpendicular to surface
- Elaunin fibers: Mid-dermis, mixed orientation
- Elastic fibers: Deep dermis, parallel to surface
💡 Master This: Collagen synthesis requires vitamin C, iron, and α-ketoglutarate as cofactors. Scurvy disrupts collagen cross-linking, causing perifollicular hemorrhages and corkscrew hairs - pathognomonic signs resulting from defective prolyl-4-hydroxylase activity.
Skin appendages demonstrate remarkable engineering precision. Hair follicles cycle through anagen (2-7 years), catagen (2-3 weeks), and telogen (2-4 months) phases, with 85-90% of scalp hairs in anagen simultaneously. Sebaceous glands produce 1-2 grams of sebum daily, with facial sebum production reaching 150-300 μg/cm²/hour.
⭐ Clinical Pearl: Eccrine sweat glands number 2-4 million total, producing up to 10 liters of sweat daily under extreme conditions. Palmar/plantar eccrine density reaches 600-700 glands/cm², explaining hyperhidrosis severity in these locations.
The subcutaneous layer contains adipocytes organized in lobules separated by fibrous septa. Subcutaneous thickness varies from 1-3 cm (face) to 5-7 cm (abdomen), providing thermal insulation and mechanical protection. This layer houses the deep vascular plexus and larger nerve trunks essential for skin function.
📌 Remember: STEP for skin functions - Sensation, Thermoregulation, Excretion, Protection. Each function requires integration of multiple anatomical components working in precise coordination to maintain homeostasis.
Understanding this architectural foundation enables prediction of pathological patterns and guides therapeutic interventions. The skin's barrier function depends on corneocyte-lipid matrix organization, while immune surveillance relies on Langerhans cell networks comprising 2-3% of epidermal cells. Connect these structural principles through functional integration to master dermatological pattern recognition.
🔬 Dermatological Foundations: The Skin's Architectural Blueprint
⚙️ Cellular Dynamics: The Skin's Renewal Engine
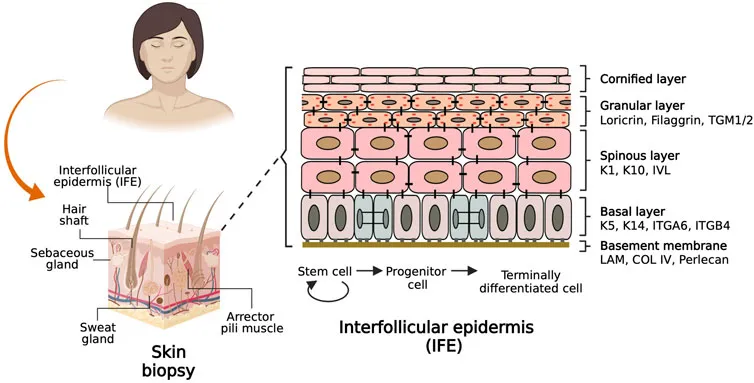
Basal layer keratinocytes express keratin 5 and 14, providing cytoskeletal structure for proliferative cells. Upon differentiation commitment, cells upregulate keratin 1 and 10 (>100-fold increase) while downregulating proliferation markers like Ki-67 and PCNA. This keratin switch occurs within 24-48 hours of leaving the basal layer.
📌 Remember: K5/K14 → K1/K10 transition marks commitment to differentiation. Epidermolytic hyperkeratosis results from K1 or K10 mutations, causing cytolysis and hyperkeratosis due to defective intermediate filament assembly.
Spinous layer development involves desmosome maturation and keratin filament bundling. Desmoglein 1 and 3 expression increases 5-10 fold, while desmocollin 1 becomes the predominant isoform. Involucrin synthesis begins, representing early cornified envelope precursor formation.
| Differentiation Stage | Duration | Key Proteins | Morphological Changes | Clinical Correlations |
|---|---|---|---|---|
| Basal | Continuous | K5/K14, integrins | Columnar, mitotic | Melanoma, BCC origin |
| Spinous | 7-10 days | K1/K10, involucrin | Polygonal, desmosomes | Pemphigus target layer |
| Granular | 2-3 days | Filaggrin, loricrin | Flattened, keratohyalin | Ichthyosis mutations |
| Cornified | 14-21 days | Cornified envelope | Anucleate, lipid matrix | Barrier dysfunction |
Granular layer formation represents terminal differentiation commitment. Keratohyalin granules contain filaggrin and trichohyalin, which aggregate keratin filaments into dense bundles. Lamellar bodies (Odland bodies) release lipid contents (ceramides, cholesterol, fatty acids) into intercellular spaces, forming the permeability barrier.
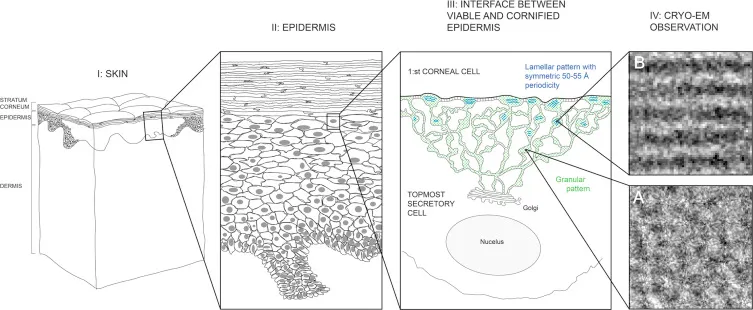
-
Cornified Envelope Assembly:
- Involucrin provides structural scaffold (85% of envelope mass)
- Loricrin contributes glycine-rich domains (70% glycine content)
- Small proline-rich proteins enable flexibility and elasticity
- SPR1 and SPR2 families provide cross-linking sites
- Transglutaminase 1 catalyzes ε(γ-glutamyl)lysine bonds
- Filaggrin aggregates keratin filaments into macrofibrils
-
Lipid Barrier Organization:
- Ceramides (50% of lipid mass): 12 distinct species identified
- Cholesterol (25%): Free cholesterol, not cholesterol esters
- Free fatty acids (15%): C16-C26 chain lengths predominate
- Linoleic acid essential for barrier competence
- Omega-hydroxyceramides provide covalent lipid-protein links
💡 Master This: Transglutaminase 1 deficiency causes lamellar ichthyosis with collodion baby presentation. Enzyme replacement through retinoid therapy partially restores cornified envelope formation, improving barrier function by 40-60%.
Desquamation requires controlled proteolysis of corneodesmosomes. Kallikrein 5 and 7 cleave desmoglein 1 and corneodesmosin, enabling cell shedding. Protease activity is pH-dependent, with optimal activity at pH 5.5 (skin surface pH).
Melanocyte function integrates with keratinocyte differentiation through dendrite-keratinocyte interactions. Single melanocytes supply 30-40 keratinocytes via dendritic processes. Melanin transfer occurs through melanosome phagocytosis, with melanin distribution determining skin phototype and UV protection capacity.
⭐ Clinical Pearl: Melanosome size and distribution patterns vary by ethnicity. African skin contains larger, singly-dispersed melanosomes, providing superior UV protection (SPF 13.4) compared to Caucasian skin (SPF 3.4) with smaller, clustered melanosomes.
Langerhans cells comprise 2-3% of epidermal cells, serving as antigen-presenting cells. Birbeck granules (tennis racket-shaped organelles) characterize these dendritic cells, which capture antigens and migrate to lymph nodes for T-cell activation. Langerhans cell density decreases 50% with chronic UV exposure.
📌 Remember: CALM for epidermal cell types - Corneocytes, Antigen-presenting cells (Langerhans), Langerhans cells, Melanocytes. Keratinocytes comprise 95%, melanocytes 3-5%, Langerhans cells 2-3%, Merkel cells <1%.
Understanding cellular dynamics enables prediction of pathological disruptions and therapeutic targets. Psoriasis accelerates keratinocyte turnover to 3-5 days, while atopic dermatitis involves barrier dysfunction from filaggrin deficiency. Connect these cellular mechanisms through inflammatory mediators to master dermatological pathophysiology.
⚙️ Cellular Dynamics: The Skin's Renewal Engine
🎯 Pattern Recognition: The Clinical Detective Framework
Primary lesion identification forms the diagnostic foundation. Macules (<1 cm flat lesions) versus patches (>1 cm flat lesions) represent color changes without surface elevation. Papules (<1 cm raised lesions) and plaques (>1 cm raised lesions) indicate epidermal thickening or dermal infiltration. Vesicles (<1 cm fluid-filled) and bullae (>1 cm fluid-filled) suggest intraepidermal or subepidermal separation.
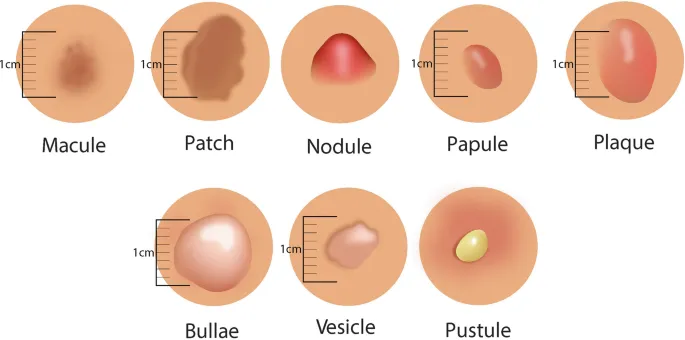
📌 Remember: MVPN for flat lesions - Macule (<1 cm), Vesicle (<1 cm fluid), Patch (>1 cm flat), Nodule (deep, >1 cm). Size cutoffs at 1 cm distinguish localized from extensive lesions with different diagnostic implications.
Distribution patterns provide crucial diagnostic clues. Symmetric distributions suggest systemic causes (drug reactions, autoimmune diseases), while asymmetric patterns indicate external factors (contact dermatitis, infections). Photodistribution affects sun-exposed areas (face, neck, dorsal hands), sparing photoprotected sites (nasolabial folds, postauricular areas).
| Distribution Pattern | Typical Conditions | Key Features | Diagnostic Accuracy |
|---|---|---|---|
| Symmetric | Psoriasis, eczema | Bilateral involvement | >90% specificity |
| Photodistributed | Lupus, drug reactions | Sun-exposed areas | 85% sensitivity |
| Dermatomal | Herpes zoster | Unilateral nerve distribution | >95% specificity |
| Flexural | Atopic dermatitis | Antecubital, popliteal | 80% in children |
| Acral | Hand-foot-mouth, syphilis | Palms, soles involved | High specificity |
Configuration analysis reveals lesion arrangement patterns. Annular (ring-shaped) lesions suggest fungal infections or granuloma annulare. Linear arrangements indicate external contact (poison ivy) or Koebner phenomenon. Grouped (herpetiform) vesicles characterize herpes simplex infections.
-
Morphological Assessment Framework:
- Surface characteristics: Smooth, rough, verrucous, umbilicated
- Border definition: Well-demarcated vs ill-defined
- Color variations: Uniform vs variegated pigmentation
- Melanin: Brown-black pigmentation
- Hemoglobin: Red-purple coloration
- Carotene: Yellow-orange hues
- Consistency: Soft, firm, hard, fluctuant
-
Secondary Change Recognition:
- Scale: Fine (pityriasis) vs thick (psoriasis)
- Crust: Honey-colored (impetigo) vs hemorrhagic
- Erosion: Partial epidermal loss (heals without scarring)
- Ulceration: Full-thickness loss (heals with scarring)
- Excoriation: Linear erosions from scratching
- Lichenification: Thickened skin from chronic rubbing
💡 Master This: Auspitz sign (pinpoint bleeding after scale removal) occurs in psoriasis due to dilated capillaries in elongated dermal papillae. This sign has 70-80% sensitivity for psoriatic plaques but low specificity (also positive in seborrheic dermatitis).
Age-related pattern recognition enhances diagnostic accuracy. Neonatal presentations include transient conditions (erythema toxicum, milia) and serious infections (herpes simplex, bacterial sepsis). Pediatric patterns favor atopic dermatitis (flexural distribution), viral exanthems, and bacterial infections.
Adult presentations shift toward inflammatory dermatoses (psoriasis, eczema) and early neoplasms (actinic keratoses, melanoma). Elderly patients develop increased malignancy risk (basal cell carcinoma incidence >2000/100,000 after age 80) and xerosis from decreased sebum production (50% reduction by age 70).
⭐ Clinical Pearl: Melanoma in children most commonly presents as amelanotic nodules (40-50% lack pigmentation) rather than classic ABCDE criteria. Pediatric melanoma accounts for <1% of childhood cancers but requires high clinical suspicion in atypical pigmented lesions.
Diagnostic algorithms integrate morphology, distribution, and clinical context. Vesiculobullous diseases require differentiation between intraepidermal (pemphigus, HSV) and subepidermal (bullous pemphigoid, dermatitis herpetiformis) blistering through clinical features and confirmatory testing.
📌 Remember: HELPS for vesiculobullous workup - Histology (level of split), Eosinophils (allergic vs autoimmune), Location (mucosal involvement), Patient age (pemphigus >50), Salt-split skin (autoantibody localization). Direct immunofluorescence provides definitive diagnosis in >95% of autoimmune blistering diseases.
Understanding pattern recognition principles enables systematic diagnostic approaches and reduces diagnostic errors. Morphological assessment combined with distribution analysis achieves diagnostic accuracy comparable to dermatopathology in common conditions. Connect these recognition patterns through pathophysiological mechanisms to master clinical dermatological reasoning.
🎯 Pattern Recognition: The Clinical Detective Framework
🔍 Diagnostic Precision: The Systematic Discrimination Matrix
Papulosquamous disorders present overlapping morphologies requiring systematic differentiation. Psoriasis demonstrates well-demarcated plaques with silvery scale, Auspitz sign positivity (70-80% sensitivity), and predilection for extensor surfaces. Seborrheic dermatitis shows greasy, yellow scales in sebaceous-rich areas with Malassezia colonization >10⁴ organisms/cm².
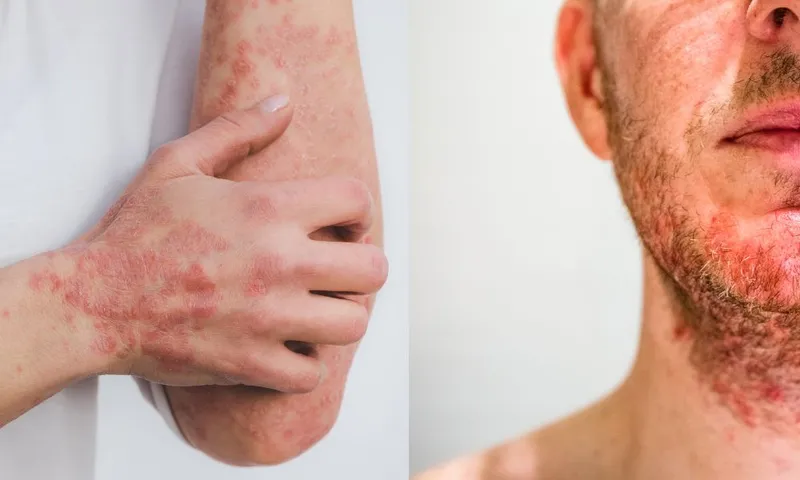
| Condition | Scale Character | Distribution | Associated Features | Histological Markers |
|---|---|---|---|---|
| Psoriasis | Silvery, thick | Extensor surfaces | Nail pitting (50%) | Munro microabscesses |
| Seborrheic Dermatitis | Greasy, yellow | Sebaceous areas | Dandruff (90%) | Spongiosis, yeast |
| Lichen Planus | Minimal scale | Flexor surfaces | Wickham striae | Band-like infiltrate |
| Pityriasis Rosea | Collarette scale | Trunk, Christmas tree | Herald patch (80%) | Focal parakeratosis |
| Tinea Corporis | Fine, peripheral | Annular lesions | KOH positive (85%) | Fungal elements |
Vesiculobullous disease discrimination requires precise anatomical localization of blister formation. Intraepidermal blistering (pemphigus group) shows acantholysis with intact basement membrane, while subepidermal blistering (pemphigoid group) demonstrates basement membrane disruption with dermal-epidermal separation.
⭐ Clinical Pearl: Nikolsky sign demonstrates different mechanisms in pemphigus versus toxic epidermal necrolysis. Pemphigus shows lateral pressure causing intraepidermal separation due to desmoglein antibodies, while TEN exhibits tangential pressure causing full-thickness epidermal necrosis.
Inflammatory dermatoses require discrimination based on cellular infiltrate patterns and tissue reaction types. Eczematous dermatitis shows spongiosis with lymphocytic infiltration, while psoriasiform dermatitis demonstrates acanthosis with neutrophilic collections. Lichenoid reactions exhibit band-like lymphocytic infiltrates at the dermal-epidermal junction.
-
Eczematous Pattern Recognition:
- Acute: Spongiosis, vesiculation with lymphocytic infiltrate
- Subacute: Moderate spongiosis with scale-crust formation
- Chronic: Acanthosis, hyperkeratosis with lichenification
- Fibroblast proliferation in papillary dermis
- Increased collagen deposition from chronic inflammation
-
Psoriasiform Discrimination:
- Regular acanthosis with elongated rete ridges
- Parakeratosis with retained nuclei in stratum corneum
- Munro microabscesses: Neutrophil collections in cornified layer
- Spongiform pustules: Neutrophils in spinous layer
- Dilated capillaries in dermal papillae (Auspitz sign)
💡 Master This: Direct immunofluorescence patterns provide definitive discrimination in autoimmune blistering diseases. Pemphigus shows intercellular IgG deposition (chicken wire pattern), while bullous pemphigoid demonstrates linear basement membrane IgG with >95% diagnostic accuracy.
Pigmentary disorder differentiation requires understanding melanin distribution and melanocyte function. Vitiligo shows complete melanocyte absence with sharp borders and Wood's lamp enhancement. Post-inflammatory hypopigmentation demonstrates decreased melanin transfer with indistinct borders and partial repigmentation potential.
| Pigmentary Disorder | Melanocyte Status | Border Character | Wood's Lamp | Histological Features |
|---|---|---|---|---|
| Vitiligo | Absent | Sharp, convex | Enhanced | No melanocytes |
| PIH | Present, decreased | Indistinct | Not enhanced | Reduced melanin |
| Tinea Versicolor | Normal | Fine scale | Yellow-green | Yeast and hyphae |
| Idiopathic Guttate | Decreased | Punctate | Enhanced | Focal melanocyte loss |
| Chemical Leukoderma | Destroyed | Irregular | Enhanced | Melanocyte necrosis |
Neoplastic versus inflammatory discrimination requires assessment of cellular atypia, growth patterns, and architectural disruption. Seborrheic keratoses demonstrate benign hyperplasia with horn cysts and pseudohorn cysts, while squamous cell carcinoma shows invasive growth with cellular atypia and keratin pearl formation.
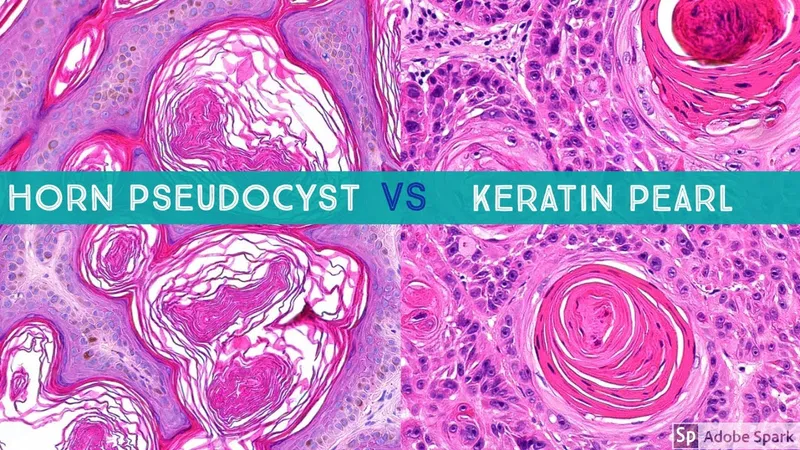
📌 Remember: ABCDE criteria for melanoma detection achieve >85% sensitivity when properly applied. Asymmetry, Border irregularity, Color variation, Diameter >6mm, Evolving characteristics. Dermoscopy increases diagnostic accuracy to >90% by revealing subsurface structures invisible to naked eye examination.
Understanding systematic discrimination principles enables precise differential diagnosis and appropriate management decisions. Morphological analysis combined with histopathological correlation and immunological markers provides diagnostic certainty in complex cases. Connect these discrimination techniques through evidence-based algorithms to master dermatological diagnostic precision.
🔍 Diagnostic Precision: The Systematic Discrimination Matrix
⚖️ Treatment Algorithms: The Evidence-Based Intervention Matrix
Topical therapy optimization follows vehicle selection principles and penetration enhancement strategies. Ointments provide maximal occlusion and drug penetration but poor cosmetic acceptance (<60% patient preference). Creams offer balanced efficacy and tolerability with moderate penetration and good patient compliance (>85% preference). Gels enable alcohol-based delivery for hairy areas with rapid absorption but potential drying effects.
| Vehicle Type | Penetration | Occlusion | Patient Preference | Optimal Use |
|---|---|---|---|---|
| Ointment | Highest | Maximum | 60% | Thick plaques, xerosis |
| Cream | Moderate | Intermediate | 85% | General use, face |
| Gel | Variable | Minimal | 75% | Hairy areas, scalp |
| Lotion | Low | None | 90% | Large areas, maintenance |
| Foam | Moderate | Low | 80% | Scalp, intertriginous |
Corticosteroid potency classification guides systematic treatment escalation. Class I (superpotent) steroids like clobetasol propionate 0.05% achieve >90% improvement in psoriatic plaques within 2-4 weeks but carry significant atrophy risk with >4 weeks continuous use. Class VII (least potent) preparations provide maintenance therapy with minimal side effects.
⭐ Clinical Pearl: Tachyphylaxis develops with continuous potent steroid use within 2-3 weeks, reducing efficacy by 40-60%. Pulse therapy (2 days on, 5 days off) or weekend therapy maintains therapeutic response while minimizing tolerance development.
Systemic therapy algorithms integrate disease severity scores and patient comorbidities. Methotrexate achieves PASI 75 response in 60-70% of psoriasis patients at 15-25 mg weekly doses. Monitoring requirements include CBC, liver function tests every 4-8 weeks, and annual chest X-ray due to pneumonitis risk (<1% incidence).
-
Methotrexate Protocol:
- Starting dose: 7.5-10 mg weekly with folic acid 5mg daily
- Dose escalation: 2.5-5 mg increments every 4-6 weeks
- Maximum dose: 25 mg weekly (higher doses no additional benefit)
- Contraindications: Pregnancy, liver disease, renal impairment
- Drug interactions: Trimethoprim, NSAIDs increase toxicity risk
- Monitoring schedule: Baseline then monthly for 3 months, then quarterly
-
Biologic Therapy Selection:
- TNF-α inhibitors: First-line for moderate-severe psoriasis
- IL-17 inhibitors: Superior efficacy (PASI 90: 70-80% vs 50-60%)
- IL-23 inhibitors: Longest durability (sustained response >1 year)
- Screening requirements: Tuberculosis, hepatitis B/C, malignancy
- Cost considerations: $50,000-80,000 annually
💡 Master This: Biologic selection depends on patient phenotype and comorbidities. Psoriatic arthritis favors TNF-α inhibitors, inflammatory bowel disease contraindicates IL-17 inhibitors, and cardiovascular disease benefits from IL-23 inhibitors due to anti-inflammatory effects.
Antimicrobial therapy requires organism-specific targeting and resistance pattern awareness. Staphylococcus aureus causes 80-90% of bacterial skin infections, with MRSA prevalence reaching 20-30% in community settings. First-line oral therapy includes cephalexin 500mg QID for susceptible strains or clindamycin 300mg TID for MRSA coverage.
| Organism | First-Line Therapy | Alternative Options | Resistance Rates | Treatment Duration |
|---|---|---|---|---|
| S. aureus (MSSA) | Cephalexin 500mg QID | Dicloxacillin | <5% to beta-lactams | 7-10 days |
| S. aureus (MRSA) | Clindamycin 300mg TID | Doxycycline, TMP-SMX | 15-20% to clindamycin | 7-10 days |
| Streptococcus | Penicillin V 500mg QID | Erythromycin | <1% to penicillin | 10 days |
| P. aeruginosa | Ciprofloxacin 500mg BID | Ceftazidime IV | 20-25% to fluoroquinolones | 10-14 days |
Antifungal therapy selection depends on organism identification and infection location. Dermatophyte infections respond to terbinafine 250mg daily with >90% cure rates for onychomycosis after 12-16 weeks therapy. Candida infections require azole antifungals with fluconazole 150mg weekly for recurrent vulvovaginal candidiasis.
📌 Remember: FAST for antifungal selection - Fluconazole (Candida), Azoles (broad spectrum), Systemic terbinafine (dermatophytes), Topical therapy (localized infections). Liver function monitoring required for systemic azoles due to hepatotoxicity risk (1-2% incidence).
Treatment response monitoring utilizes validated severity scores and objective measurements. PASI score (Psoriasis Area and Severity Index) incorporates erythema, induration, desquamation, and body surface area to generate 0-72 point scale. PASI 75 (75% improvement) represents clinically meaningful response achieved by >80% of patients on biologic therapy.
Understanding evidence-based treatment algorithms enables optimal therapeutic outcomes while minimizing adverse effects. Systematic approach to drug selection, dosing, and monitoring improves patient safety and treatment adherence. Connect these therapeutic principles through personalized medicine approaches to master dermatological treatment optimization.
⚖️ Treatment Algorithms: The Evidence-Based Intervention Matrix
🔗 Multi-System Integration: The Dermatological Connectome
Cutaneous immunology interfaces with systemic immune networks through shared cellular populations and cytokine pathways. Langerhans cells migrate from epidermis to regional lymph nodes within 24-48 hours of antigen encounter, initiating T-cell responses that influence both local and systemic immunity. Skin-associated lymphoid tissue (SALT) contains 2x10¹⁰ T-cells, representing 20% of total body T-cell population.
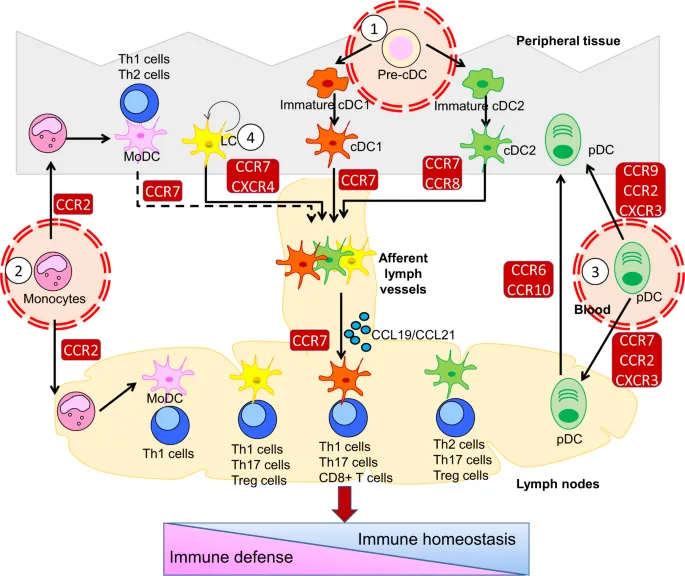
Psoriasis exemplifies multi-system inflammatory disease with cardiovascular, metabolic, and arthritic comorbidities. TNF-α overexpression (5-10 fold elevation) drives not only skin inflammation but also insulin resistance, endothelial dysfunction, and accelerated atherosclerosis. Psoriasis patients demonstrate 40-50% increased risk of myocardial infarction and 20-30% increased risk of diabetes mellitus.
📌 Remember: IMPACT for psoriasis comorbidities - Inflammatory bowel disease (3-4x risk), Metabolic syndrome (2-3x risk), Psychiatric disorders (depression 2x), Arthritis (30% prevalence), Cardiovascular disease (40-50% increased MI), Tumor risk (lymphoma 2-3x).
Endocrine-dermatological interactions manifest through hormone-responsive skin changes and cutaneous hormone production. Sebaceous glands express androgen receptors and 5α-reductase, converting testosterone to dihydrotestosterone (DHT) locally. Acne severity correlates with sebum production rates (150-300 μg/cm²/hour in affected individuals vs 50-100 μg/cm²/hour in controls).
| Endocrine Condition | Cutaneous Manifestations | Mechanism | Prevalence |
|---|---|---|---|
| Diabetes Mellitus | Acanthosis nigricans | Insulin resistance | 75% in type 2 DM |
| Thyroid Disease | Pretibial myxedema | Hyaluronic acid deposition | 5% in Graves disease |
| Cushing Syndrome | Purple striae | Collagen breakdown | 90% of cases |
| PCOS | Hirsutism, acne | Androgen excess | 70-80% of patients |
| Addison Disease | Hyperpigmentation | ACTH elevation | >90% of cases |
Neurological-cutaneous connections operate through shared embryological origins and ongoing neural regulation. Neural crest cells give rise to melanocytes, Schwann cells, and peripheral neurons, explaining neurocutaneous syndromes like neurofibromatosis and tuberous sclerosis. Cutaneous innervation includes sensory, autonomic, and peptidergic components affecting inflammation, vascular tone, and appendageal function.
💡 Master This: Substance P and CGRP (calcitonin gene-related peptide) mediate neurogenic inflammation in skin. Stress-induced release causes vasodilation, plasma extravasation, and mast cell degranulation, explaining stress-triggered exacerbations of atopic dermatitis and psoriasis in 60-70% of patients.
Gastrointestinal-skin axis demonstrates bidirectional communication through microbiome interactions and shared inflammatory pathways. Inflammatory bowel disease associates with pyoderma gangrenosum (1-2% prevalence), erythema nodosum (3-10% prevalence), and aphthous stomatitis (5-20% prevalence). Gut microbiome diversity influences skin barrier function and atopic dermatitis severity.
-
Microbiome-Skin Interactions:
- Gut dysbiosis reduces short-chain fatty acid production
- Decreased butyrate impairs regulatory T-cell development
- Systemic inflammation increases skin barrier dysfunction
- Filaggrin expression decreases 30-40% in dysbiotic states
- Ceramide synthesis reduces 20-30% affecting barrier integrity
- Probiotic supplementation improves atopic dermatitis in 40-50% of pediatric patients
-
Hepatic-Cutaneous Connections:
- Chronic liver disease causes spider angiomata (>5 lesions in 80% of cirrhotic patients)
- Palmar erythema occurs in 60-70% of chronic liver disease
- Pruritus affects 70-80% of primary biliary cholangitis patients
- Bile acid accumulation activates pruritogenic pathways
- Autotaxin elevation (5-10 fold) correlates with itch severity
⭐ Clinical Pearl: Dermatitis herpetiformis represents cutaneous manifestation of celiac disease in 15-25% of gluten-sensitive patients. IgA anti-tissue transglutaminase antibodies cross-react with epidermal transglutaminase 3, causing subepidermal blistering with pathognomonic granular IgA deposits.
Cardiovascular-dermatological relationships extend beyond shared risk factors to direct pathophysiological connections. Chronic inflammatory skin diseases elevate C-reactive protein (2-5 fold), interleukin-6 (3-8 fold), and TNF-α (5-10 fold), promoting endothelial dysfunction and accelerated atherosclerosis. Psoriasis treatment with biologics reduces cardiovascular events by 20-30% independent of traditional risk factors.
📌 Remember: HEART for cardiovascular-skin connections - Hypertension (increased in psoriasis), Endothelial dysfunction (inflammatory mediators), Atherosclerosis (accelerated), Risk factors (shared metabolic), Treatment benefits (anti-inflammatory therapy). Dermatological inflammation serves as cardiovascular risk marker requiring integrated management.
Renal-cutaneous manifestations reflect shared filtration and excretory functions. Chronic kidney disease causes uremic pruritus in 60-90% of dialysis patients through accumulation of pruritogenic toxins and altered skin barrier function. Nephrogenic systemic fibrosis develops in <5% of CKD patients exposed to gadolinium contrast, causing progressive skin fibrosis and joint contractures.
Understanding multi-system integration enables comprehensive patient care and recognition of systemic disease manifestations. Dermatological findings often provide early clues to systemic conditions, while skin diseases influence overall health outcomes. Connect these integration principles through personalized medicine approaches to master holistic dermatological practice.
🔗 Multi-System Integration: The Dermatological Connectome
🎯 Clinical Mastery Arsenal: The Rapid-Fire Reference Matrix
Essential Diagnostic Thresholds form the numerical foundation for clinical decision-making. Melanoma thickness determines staging and prognosis: <1mm (Stage IA, >95% 5-year survival), 1-2mm (Stage IB-IIA, 85-90% survival), 2-4mm (Stage IIB-IIC, 70-80% survival), >4mm (Stage III-IV, <50% survival). Breslow depth measurement accuracy to ±0.1mm significantly impacts treatment recommendations.
📌 Remember: ABCDE Plus for melanoma detection - Asymmetry, Border irregularity, Color variation, Diameter >6mm, Evolving characteristics, Plus ugly duckling sign (lesion different from others). Dermoscopy increases sensitivity to >90% and specificity to >80%.
Critical Laboratory Values guide systemic therapy monitoring and disease activity assessment. Methotrexate toxicity monitoring requires CBC and liver enzymes every 4-8 weeks, with dose reduction for AST/ALT >2x normal or platelet count <100,000. Cyclosporine necessitates creatinine monitoring every 2 weeks initially, with dose reduction for >30% increase from baseline.
| Medication | Monitoring Parameter | Frequency | Action Threshold | Clinical Significance |
|---|---|---|---|---|
| Methotrexate | AST/ALT | Monthly x3, then quarterly | >2x normal | Hepatotoxicity risk |
| Cyclosporine | Creatinine | Biweekly x2, then monthly | >30% increase | Nephrotoxicity |
| Acitretin | Lipids | Monthly x3, then quarterly | Triglycerides >400 | Pancreatitis risk |
| Biologics | CBC | Quarterly | ANC <1000 | Infection risk |
| Hydroxychloroquine | Ophthalmologic exam | Annually | Retinal changes | Irreversible toxicity |
Rapid Treatment Algorithms enable systematic therapeutic escalation. Psoriasis management follows step-wise approach: topical therapy (mild disease, <3% BSA), phototherapy (moderate disease, 3-10% BSA), systemic therapy (severe disease, >10% BSA or significant impact). Treatment failure defined as <50% improvement after adequate trial (8-12 weeks for topicals, 12-16 weeks for systemics).
💡 Master This: Body Surface Area estimation uses patient's palm (including fingers) = 1% BSA. Rule of 9s applies for extensive involvement: head/neck 9%, each arm 9%, anterior trunk 18%, posterior trunk 18%, each leg 18%, genitalia 1%.
Emergency Recognition Patterns require immediate identification and urgent intervention. Stevens-Johnson Syndrome/Toxic Epidermal Necrolysis presents with >30% BSA involvement (TEN) or <10% BSA (SJS), positive Nikolsky sign, and mucosal involvement (>90% cases). Mortality rates: SJS 5-10%, SJS-TEN overlap 10-35%, TEN 25-50%.
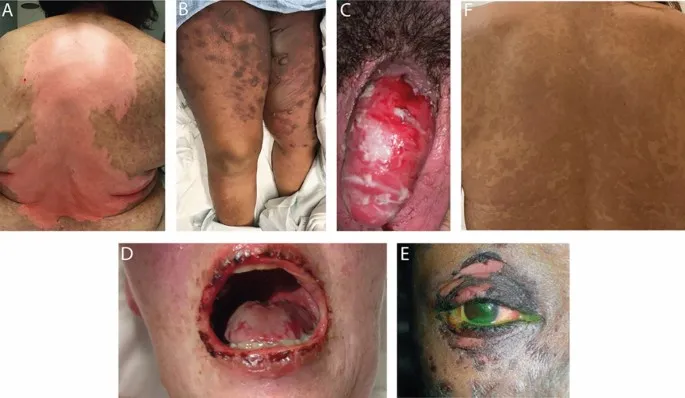
- Dermatological Emergency Checklist:
- Necrotizing fasciitis: Rapid spread (>1 cm/hour), severe pain, systemic toxicity
- Staphylococcal scalded skin syndrome: Widespread erythema, positive Nikolsky, neonates/children
- Drug reaction with eosinophilia: Fever, eosinophilia >1000, organ involvement
- DRESS syndrome: 2-8 week onset, facial edema, lymphadenopathy
- Mortality 5-10% without prompt steroid therapy
- Erythroderma: >90% BSA involvement, thermoregulatory failure, high-output cardiac failure
⭐ Clinical Pearl: SCORTEN (Severity-of-illness Score for TEN) predicts mortality using 7 variables: age >40, malignancy, tachycardia >120, initial BSA >10%, BUN >28, bicarbonate <20, glucose >252. Score 0-1: 3% mortality, Score 5+: >90% mortality.
Prognostic Indicators enable accurate patient counseling and treatment planning. Basal cell carcinoma demonstrates <1% metastasis rate overall, but high-risk features (perineural invasion, >2cm diameter, aggressive histology) increase recurrence risk to 5-15%. Squamous cell carcinoma metastasis risk varies: <2% (sun-exposed sites) to >20% (immunosuppressed patients, high-risk locations).
| Cancer Type | 5-Year Survival | High-Risk Features | Metastasis Rate | Follow-up Protocol |
|---|---|---|---|---|
| Melanoma in situ | >99% | Lentigo maligna subtype | 0% | Annual exam x5 years |
| Melanoma <1mm | >95% | Ulceration, mitoses | <5% | Every 6 months x5 years |
| BCC | >99% | Perineural invasion | <1% | Annual exam |
| SCC | >95% | Perineural invasion, >2cm | 2-20% | Every 6 months x2 years |
| Merkel cell | 50-70% | >2cm, node positive | 30-50% | Every 3 months x2 years |
Understanding clinical mastery tools enables rapid, accurate decision-making in complex dermatological scenarios. Systematic approach to diagnostic thresholds, treatment algorithms, and prognostic assessment optimizes patient outcomes while minimizing errors. These reference frameworks provide the foundation for expert-level dermatological practice and confident clinical decision-making.
🎯 Clinical Mastery Arsenal: The Rapid-Fire Reference Matrix
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more
Have doubts about this lesson?
Ask Rezzy, your AI Study Mate, to explain anything you didn't understand
Everything you need for NEET-PG prep
Get full Oncourse access with lessons, practice questions, flashcards and AI study tools.
Scan to download app
























