Membrane Biochemistry

On this page
🧬 Membrane Biochemistry: The Cellular Fortress Architecture
Every cell in your body maintains its identity and function through a dynamic barrier just two molecules thick-the plasma membrane. You'll explore how phospholipid bilayers create selective fortresses, how proteins transform these barriers into communication hubs and transport highways, and how electrochemical gradients power cellular work. By connecting membrane architecture to signal transduction and clinical disorders, you'll understand why cystic fibrosis, familial hypercholesterolemia, and drug resistance all trace back to membrane dysfunction, making this knowledge essential for diagnosing and treating disease at the molecular level.
Membrane biochemistry represents the fundamental control system governing cellular existence. Every nutrient entry, waste removal, signal reception, and energy transaction depends on membrane integrity and function. Master these membrane principles, and you unlock the logic behind every metabolic disorder, drug mechanism, and therapeutic intervention.
The membrane's 7-nanometer thickness creates a selective barrier that maintains life's essential gradients while enabling controlled molecular exchange. This architectural marvel integrates 200+ different lipid species, 500+ membrane proteins, and countless regulatory mechanisms into a dynamic, self-organizing system.
Understanding membrane biochemistry transforms clinical reasoning from memorizing isolated facts to recognizing the unified principles underlying cellular pathophysiology. Every disease process ultimately traces back to membrane dysfunction, making this knowledge essential for diagnostic accuracy and therapeutic success.
🧬 Membrane Biochemistry: The Cellular Fortress Architecture
🏗️ The Phospholipid Foundation: Cellular Architecture Blueprints
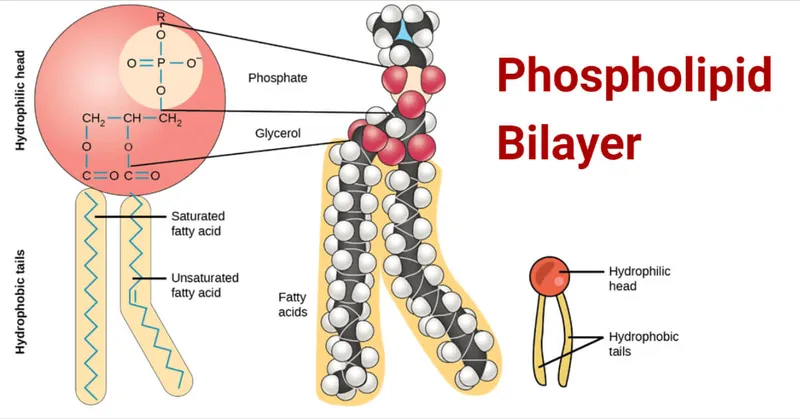
The phospholipid bilayer establishes cellular compartmentalization through amphipathic molecules containing both hydrophilic heads and hydrophobic tails. This dual nature creates spontaneous self-assembly into bilayer structures with 2-3 nanometer thickness per leaflet.
Phospholipid Composition and Distribution
- Phosphatidylcholine (PC): 45-55% of membrane phospholipids
- Primary structural component in most mammalian membranes
- Provides membrane stability and fluidity balance
- Critical for membrane repair and synthesis
- Phosphatidylserine (PS): 10-15% of total phospholipids
- Exclusively located on inner leaflet (>95% asymmetry)
- Exposure signals apoptosis and phagocytosis
- Essential for protein kinase C activation
- Phosphatidylethanolamine (PE): 20-25% of membrane content
- Cone-shaped structure promotes membrane curvature
- Critical for membrane fusion events
- Concentrated in mitochondrial membranes (35-40%)
📌 Remember: CAPS for major phospholipids - Choline (most abundant), Aminophospholipids (PE/PS), Phosphatidylinositol (signaling), Sphingomyelin (rafts). Each serves distinct structural and functional roles with specific membrane distributions.
| Phospholipid | Percentage | Primary Location | Key Function | Clinical Significance |
|---|---|---|---|---|
| Phosphatidylcholine | 45-55% | Both leaflets | Structural integrity | Lecithin deficiency |
| Phosphatidylserine | 10-15% | Inner leaflet only | Apoptosis signaling | Scott syndrome |
| Phosphatidylethanolamine | 20-25% | Both leaflets | Membrane curvature | Mitochondrial disorders |
| Sphingomyelin | 5-10% | Outer leaflet | Lipid raft formation | Niemann-Pick disease |
| Phosphatidylinositol | 2-8% | Inner leaflet | Signal transduction | Lowe syndrome |
Membrane leaflets maintain distinct compositions through active transport mechanisms. ATP-dependent flippases transport aminophospholipids (PS, PE) to the inner leaflet, while floppases move phospholipids outward. This asymmetry requires 2-4 ATP molecules per phospholipid translocation.
⭐ Clinical Pearl: PS externalization occurs within 30-60 minutes of apoptosis initiation, serving as an "eat-me" signal for macrophages. Annexin V binding to externalized PS provides >95% specificity for detecting early apoptosis in clinical assays.
The energy cost of maintaining asymmetry represents 15-20% of cellular ATP consumption, highlighting its critical importance for cellular function. Loss of asymmetry occurs in pathological conditions including ischemia, oxidative stress, and membrane aging.
💡 Master This: Membrane phospholipid composition directly determines physical properties including fluidity, permeability, and protein function. Understanding these relationships predicts how lipid disorders affect cellular metabolism and drug distribution.
Connect these foundational phospholipid principles through membrane protein integration to understand how structure enables sophisticated cellular functions.
🏗️ The Phospholipid Foundation: Cellular Architecture Blueprints
⚡ Membrane Protein Integration: The Functional Command Centers
Integral Membrane Protein Architecture
- Single-pass proteins: 20-25% of membrane proteins
- Single transmembrane domain spanning 20-25 amino acids
- Include many receptor proteins and cell adhesion molecules
- Examples: EGFR, integrins, immunoglobulin superfamily
- Multi-pass proteins: 60-70% of membrane proteins
- Multiple transmembrane domains (2-12 typical range)
- Include transporters, channels, and complex receptors
- G-protein coupled receptors contain 7 transmembrane domains
- Membrane-associated proteins: 10-15% of total
- Lipid-anchored or peripherally associated
- Include GPI-anchored proteins and prenylated proteins
📌 Remember: TIMES for membrane protein types - Transporters (move stuff), Ion channels (selective gates), Markers (identification), Enzymes (catalysis), Signal receptors (communication). Each type requires specific membrane integration mechanisms.
Protein-Lipid Interactions and Regulation
Membrane proteins require specific lipid environments for optimal function. Annular lipids directly contact protein surfaces, while non-annular lipids occupy specific binding sites within protein structures.
| Protein Type | Lipid Requirement | Functional Impact | Clinical Example |
|---|---|---|---|
| Na+/K+-ATPase | Phosphatidylserine | 50% activity loss without PS | Cardiac glycoside sensitivity |
| Cytochrome oxidase | Cardiolipin | 80% activity reduction | Barth syndrome |
| GPCR | Cholesterol | Altered ligand binding | Statin effects on receptors |
| Ion channels | PIP2 | Gating modulation | Epilepsy channelopathies |
| Transporters | Membrane fluidity | Transport kinetics | Temperature-sensitive mutations |
Membrane Protein Quality Control
The endoplasmic reticulum maintains strict quality control for membrane protein folding and assembly. Misfolded proteins trigger ER-associated degradation (ERAD) within 2-4 hours of synthesis.
- Chaperone systems: BiP, calnexin, calreticulin
- Monitor protein folding for 6-8 hours maximum
- Facilitate proper disulfide bond formation
- Prevent aggregation during membrane insertion
- Degradation pathways: Proteasomal and lysosomal
- Remove 15-20% of newly synthesized membrane proteins
- Prevent accumulation of toxic aggregates
- Maintain membrane protein homeostasis
💡 Master This: Membrane protein integration requires coordination between synthesis, folding, lipid environment, and quality control systems. Disruption of any component leads to protein mislocalization and cellular dysfunction.
Connect these protein integration principles through transport mechanism analysis to understand how membrane proteins enable selective molecular trafficking.
⚡ Membrane Protein Integration: The Functional Command Centers
🚛 Transport Mechanism Mastery: Selective Molecular Highways

Membrane transport mechanisms determine cellular composition, energy status, and response capabilities. Understanding these systems reveals the logic behind metabolic regulation, drug action, and pathophysiological processes.
Passive Transport Kinetics and Selectivity
- Simple diffusion: Lipophilic molecules <500 Da
- Rate proportional to concentration gradient
- No saturation kinetics or competitive inhibition
- Examples: O₂, CO₂, steroid hormones, anesthetics
- Permeability coefficient: 10⁻⁶ to 10⁻⁴ cm/s
- Facilitated diffusion: Hydrophilic molecules
- Michaelis-Menten kinetics with Km values
- Competitive inhibition and saturation
- 100-1000x faster than simple diffusion
- Examples: glucose via GLUT, ions via channels
📌 Remember: FAST for transport types - Facilitated (carrier-mediated), Active (energy-requiring), Simple (direct diffusion), Transport (bulk movement). Each mechanism serves specific molecular size and polarity requirements.
Active Transport Energetics and Coupling
Active transport systems overcome concentration gradients using various energy sources with distinct thermodynamic requirements.
| Transport Type | Energy Source | Coupling Ratio | Example | Clinical Relevance |
|---|---|---|---|---|
| Primary active | ATP hydrolysis | 1-3 ions/ATP | Na+/K+-ATPase | Digitalis toxicity |
| Secondary active | Ion gradients | 1-3 substrates/ion | SGLT1 glucose | Diabetes therapy |
| Tertiary active | Multiple gradients | Variable coupling | NCX exchanger | Cardiac contractility |
| Group translocation | Phosphorylation | 1:1 substrate:PEP | PTS system | Bacterial metabolism |
| ABC transporters | ATP binding/hydrolysis | 2 ATP/cycle | P-glycoprotein | Drug resistance |

Transport Protein Regulation and Pathophysiology
Transport protein activity responds to multiple regulatory mechanisms operating on different timescales:
- Allosteric regulation: Millisecond to second responses
- Substrate and product inhibition
- Cofactor and ion dependencies
- pH and membrane potential effects
- Covalent modification: Second to minute responses
- Phosphorylation by protein kinases (>50 sites on CFTR)
- Ubiquitination for degradation signaling
- Glycosylation affecting trafficking
- Transcriptional control: Hour to day responses
- Hormone-responsive element activation
- Stress response pathway induction
- Circadian rhythm synchronization
💡 Master This: Transport protein dysfunction underlies major disease categories including diabetes (glucose transporters), cystic fibrosis (chloride channels), and hypertension (sodium transporters). Therapeutic strategies target specific regulatory mechanisms.
Connect these transport principles through membrane potential analysis to understand how electrochemical gradients drive cellular energetics and signaling.
🚛 Transport Mechanism Mastery: Selective Molecular Highways
⚡ Electrochemical Gradient Analysis: The Cellular Power Grid
Ion Gradient Establishment and Maintenance
- Sodium gradient: [Na+]out/[Na+]in = 10-15:1
- Extracellular: 140-145 mM, Intracellular: 10-15 mM
- Maintained by Na+/K+-ATPase consuming 25-40% cellular ATP
- Provides driving force for >100 secondary transport systems
- Equilibrium potential: +60 to +70 mV
- Potassium gradient: [K+]in/[K+]out = 30-40:1
- Intracellular: 140-150 mM, Extracellular: 3.5-5.0 mM
- Primary determinant of resting membrane potential
- Equilibrium potential: -90 to -95 mV
- Calcium gradient: [Ca2+]out/[Ca2+]in = 10,000:1
- Extracellular: 1-2 mM, Intracellular: 100-200 nM
- Steepest gradient requiring multiple transport systems
- Equilibrium potential: +120 to +130 mV
📌 Remember: PACK for major ion gradients - Potassium (high inside), All others low inside, Calcium (steepest gradient), Keep with ATP pumps. Each gradient serves specific physiological functions and requires distinct maintenance mechanisms.
Membrane Potential Determinants and Regulation
The Goldman-Hodgkin-Katz equation describes membrane potential based on ion permeabilities and concentrations:
$$V_m = \frac{RT}{F} \ln\left(\frac{P_K[K^+]o + P{Na}[Na^+]o + P{Cl}[Cl^-]_i}{P_K[K^+]i + P{Na}[Na^+]i + P{Cl}[Cl^-]_o}\right)$$
| Cell Type | Resting Potential | Primary Determinant | Key Channels | Clinical Significance |
|---|---|---|---|---|
| Neurons | -70 to -80 mV | K+ permeability | Kir, KCNQ | Epilepsy, neuropathy |
| Cardiac myocytes | -85 to -95 mV | K+ permeability | IK1, IKr, IKs | Arrhythmias, sudden death |
| Skeletal muscle | -80 to -90 mV | K+ permeability | Kir2.1 | Periodic paralysis |
| Smooth muscle | -40 to -60 mV | Mixed permeability | Various | Hypertension, asthma |
| Epithelial cells | -30 to -70 mV | Variable | ENaC, CFTR | Cystic fibrosis, Liddle syndrome |
Gradient-Driven Transport Energetics
Secondary active transport systems harness ion gradients to drive substrate accumulation against concentration gradients. The maximum concentration ratio achievable depends on the driving ion gradient:
- Sodium-coupled transporters: Maximum 100-1000:1 ratios
- SGLT1: 2 Na+ : 1 glucose stoichiometry
- Can concentrate glucose >200-fold above equilibrium
- Provides >90% of renal glucose reabsorption
- Proton-coupled systems: Maximum 10,000:1 ratios
- Mitochondrial pH gradient: 0.5-1.0 pH units
- Drives ATP synthesis with >95% efficiency
- Essential for oxidative phosphorylation
💡 Master This: Electrochemical gradient disruption rapidly compromises cellular function, explaining why ischemia, hypoxia, and metabolic poisons cause immediate cellular dysfunction. Understanding gradient energetics predicts therapeutic targets and toxicity mechanisms.
Connect these gradient principles through signal transduction analysis to understand how membrane potential changes enable rapid cellular communication.
⚡ Electrochemical Gradient Analysis: The Cellular Power Grid
🎯 Signal Transduction Integration: Membrane Communication Networks
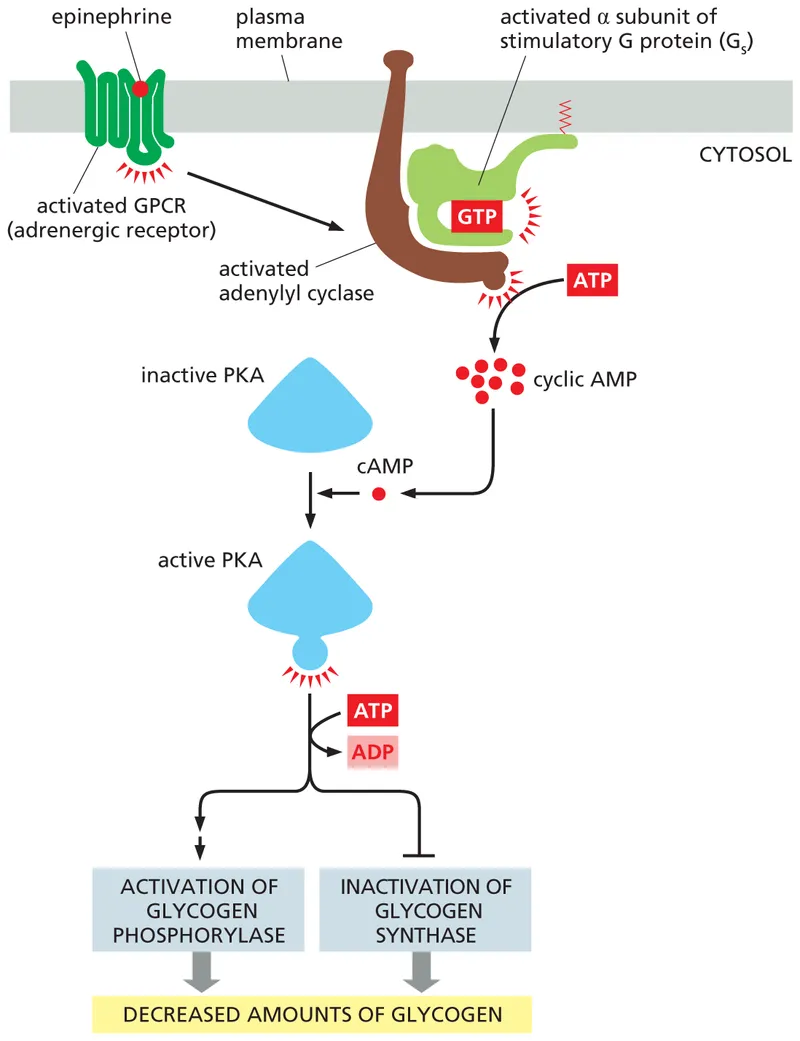
Signal transduction transforms extracellular information into intracellular responses through membrane-bound receptor systems. These networks enable cellular adaptation, coordination, and survival in complex multicellular environments.
Receptor Classification and Signal Amplification
- G-protein coupled receptors (GPCRs): >800 human subtypes
- Seven transmembrane domain architecture
- Signal amplification: 1 receptor → 100+ G-proteins
- Response time: Milliseconds to minutes
- Examples: β-adrenergic, muscarinic, dopamine receptors
- Receptor tyrosine kinases (RTKs): >60 human subtypes
- Single transmembrane domain with kinase activity
- Autophosphorylation creates docking sites
- Signal amplification: 1 receptor → 1000+ substrates
- Examples: insulin, EGF, PDGF receptors
- Ligand-gated ion channels: >50 major subtypes
- Direct coupling of binding to ion flux
- Signal amplification: 10^6-10^8 ions per opening
- Response time: Microseconds to milliseconds
- Examples: nicotinic, GABA, glutamate receptors
📌 Remember: GREAT for receptor types - GPCRs (7TM amplifiers), RTKs (kinase cascades), Enzyme-linked (direct catalysis), Acetylcholine-type (ligand-gated), Transport-coupled (nutrient sensing). Each provides distinct signal processing capabilities.
Second Messenger Systems and Integration
Second messenger systems amplify and integrate signals from multiple receptor types with distinct kinetic and spatial properties.
| Second Messenger | Source | Target | Amplification | Duration | Clinical Relevance |
|---|---|---|---|---|---|
| cAMP | Adenylyl cyclase | PKA, EPAC | 100-1000x | Seconds-minutes | Cholera toxin, caffeine |
| cGMP | Guanylyl cyclase | PKG, PDEs | 50-500x | Seconds-minutes | Nitric oxide, sildenafil |
| IP3 | PLC activation | Ca2+ release | 10-100x | Seconds | Lithium, bipolar disorder |
| DAG | PLC activation | PKC isoforms | 10-100x | Minutes | Phorbol esters, cancer |
| Ca2+ | Multiple sources | >100 targets | 1000-10000x | Milliseconds-hours | Calcium channel blockers |
Membrane Receptor Regulation and Desensitization
Receptor systems employ multiple mechanisms to prevent signal saturation and maintain responsiveness:
- Homologous desensitization: Seconds to minutes
- Receptor phosphorylation by specific kinases (GRKs, RTK autophosphorylation)
- β-arrestin binding and receptor internalization
- 50-90% reduction in responsiveness
- Heterologous desensitization: Minutes to hours
- Cross-phosphorylation by PKA, PKC
- Affects multiple receptor subtypes
- 20-50% reduction in responsiveness
- Receptor downregulation: Hours to days
- Decreased receptor synthesis
- Increased receptor degradation
- 70-95% reduction in receptor number
💡 Master This: Signal transduction dysfunction underlies major disease categories including diabetes (insulin resistance), depression (neurotransmitter imbalance), and cancer (growth factor dysregulation). Therapeutic strategies must account for receptor regulation and cross-talk.
Connect these signaling principles through clinical membrane pathology to understand how membrane dysfunction manifests in human disease.
🎯 Signal Transduction Integration: Membrane Communication Networks
🏥 Clinical Membrane Mastery: Pathophysiology and Therapeutic Targets
Membrane Structural Disorders and Clinical Presentations
- Hereditary spherocytosis: 1 in 2,000-5,000 prevalence
- Spectrin, ankyrin, or band 3 protein defects
- Membrane surface area loss: 20-30% reduction
- Clinical features: hemolytic anemia, splenomegaly, gallstones
- Osmotic fragility test: >95% diagnostic sensitivity
- Duchenne muscular dystrophy: 1 in 3,500 male births
- Dystrophin deficiency disrupts membrane-cytoskeleton linkage
- Membrane permeability increases 10-100 fold
- CK elevation: >10,000 U/L (normal <200 U/L)
- Progressive muscle weakness beginning age 2-5 years
- Hereditary elliptocytosis: 1 in 2,500 prevalence
- α-spectrin or protein 4.1 mutations
- Membrane mechanical stability reduced >50%
- Usually asymptomatic unless severe variants
📌 Remember: SHED for membrane structural disorders - Spherocytosis (spectrin/ankyrin), Hemolysis (membrane fragility), Elliptocytosis (spectrin defects), Dystrophy (dystrophin loss). Each involves specific membrane protein defects with characteristic clinical patterns.
Membrane Transport Disorders and Metabolic Consequences
Transport protein dysfunction creates specific metabolic signatures enabling targeted diagnosis and treatment.
| Disorder | Defective Protein | Prevalence | Key Clinical Features | Diagnostic Test | Treatment |
|---|---|---|---|---|---|
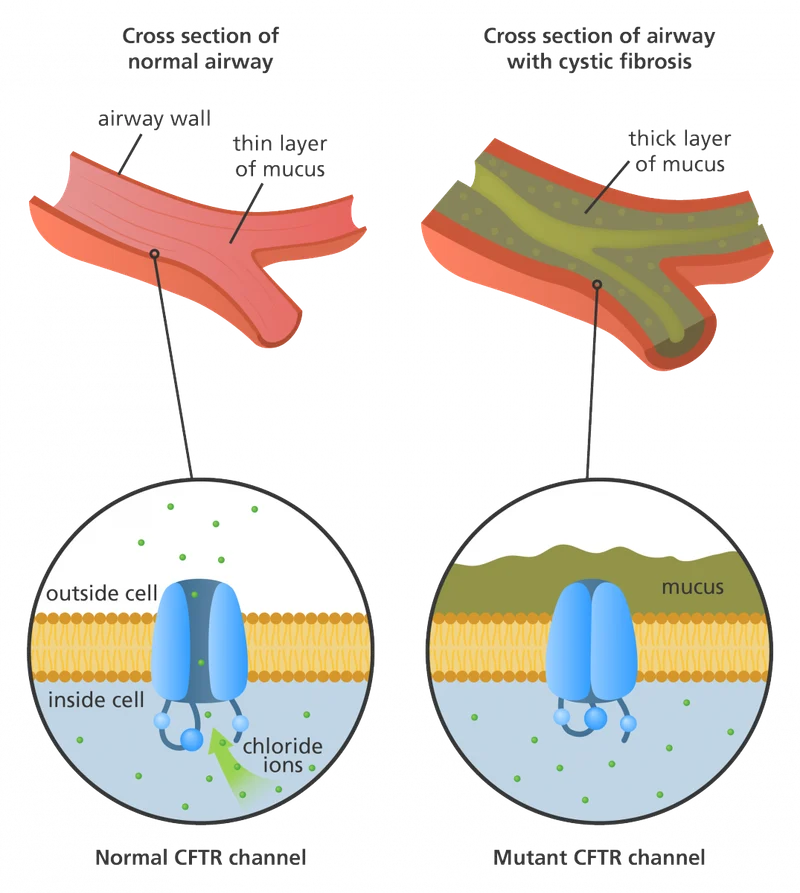
| Cystic fibrosis | CFTR chloride channel | 1 in 2,500 | Pulmonary/GI dysfunction | Sweat chloride >60 mM | CFTR modulators |
| Glucose-galactose malabsorption | SGLT1 transporter | 1 in 43,000 | Severe diarrhea, dehydration | Glucose tolerance test | Fructose-based diet |
| Hartnup disease | Neutral amino acid transporter | 1 in 30,000 | Pellagra-like symptoms | Aminoaciduria | Nicotinamide supplementation |
| Bartter syndrome | NKCC2, ROMK channels | 1 in 1,000,000 | Hypokalemic alkalosis | Genetic testing | Electrolyte replacement |
| Gitelman syndrome | NCCT transporter | 1 in 40,000 | Hypomagnesemia, hypocalciuria | Thiazide-like effects | Magnesium supplementation |
Membrane Receptor Disorders and Signal Transduction Defects
Receptor dysfunction creates characteristic clinical syndromes with specific therapeutic implications:
- Nephrogenic diabetes insipidus: V2 receptor or aquaporin-2 defects
- Urine osmolality: <300 mOsm/kg despite dehydration
- Daily urine output: >3-4 liters in infants
- Treatment: thiazide diuretics, amiloride
- Pseudohypoparathyroidism: Gs protein defects
- PTH resistance with >10-fold elevated PTH levels
- Albright hereditary osteodystrophy features
- Treatment: calcitriol and calcium supplementation
- Familial hypercholesterolemia: LDL receptor defects
- Cholesterol levels: >300 mg/dL (heterozygotes), >600 mg/dL (homozygotes)
- Premature coronary disease: 50% by age 50 (heterozygotes)
- Treatment: statins, PCSK9 inhibitors, LDL apheresis
💡 Master This: Membrane disorders require systematic analysis of structure-function relationships, inheritance patterns, and metabolic consequences. Understanding these relationships enables precision diagnosis and targeted therapy selection for optimal patient outcomes.
This comprehensive membrane biochemistry framework provides the essential knowledge base for understanding cellular pathophysiology, drug mechanisms, and therapeutic interventions across all medical specialties.
🏥 Clinical Membrane Mastery: Pathophysiology and Therapeutic Targets
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more
Have doubts about this lesson?
Ask Rezzy, your AI Study Mate, to explain anything you didn't understand
Everything you need for NEET-PG prep
Get full Oncourse access with lessons, practice questions, flashcards and AI study tools.
Scan to download app
























