Back
NEET PG Musculoskeletal & Anti-Inflammatory Pharmacology Glossary 2026: NSAIDs, DMARDs, Anti-Gout Drugs and Muscle Relaxants
Complete NEET PG musculoskeletal pharmacology glossary 2026 covering NSAIDs mechanisms, DMARDs, anti-gout drugs, and muscle relaxants with high-yield MCQ mnemonics for INICET preparation.

NEET PG Musculoskeletal & Anti-Inflammatory Pharmacology Glossary 2026: NSAIDs, DMARDs, Anti-Gout Drugs and Muscle Relaxants
You are staring at your musculoskeletal pharmacology notes, probably thinking "how do I memorize all these drug mechanisms before NEET PG 2026?" NSAIDs blend into each other. DMARD names look like alphabet soup. Anti-gout drugs feel random. And muscle relaxants? You cant even pronounce half of them.
Here's what's different about this glossary — it's the final piece in the NEET PG pharmacology series. While other resources dump information, this gives you the exact mechanism mnemonics, side effect patterns, and MCQ angles that show up repeatedly in NEET PG and INICET 2026. Every drug here has appeared in the last 3 years of Indian PG entrance exams.
The musculoskeletal system touches everything — orthopedics, rheumatology, internal medicine, emergency medicine. Miss these pharmacology concepts and you lose points across multiple subjects. But master these 47 high-yield drugs with their mechanisms and you've locked down 15-20 marks minimum.
This isnt another textbook chapter. It's your final revision weapon.
Drug Classification Overview
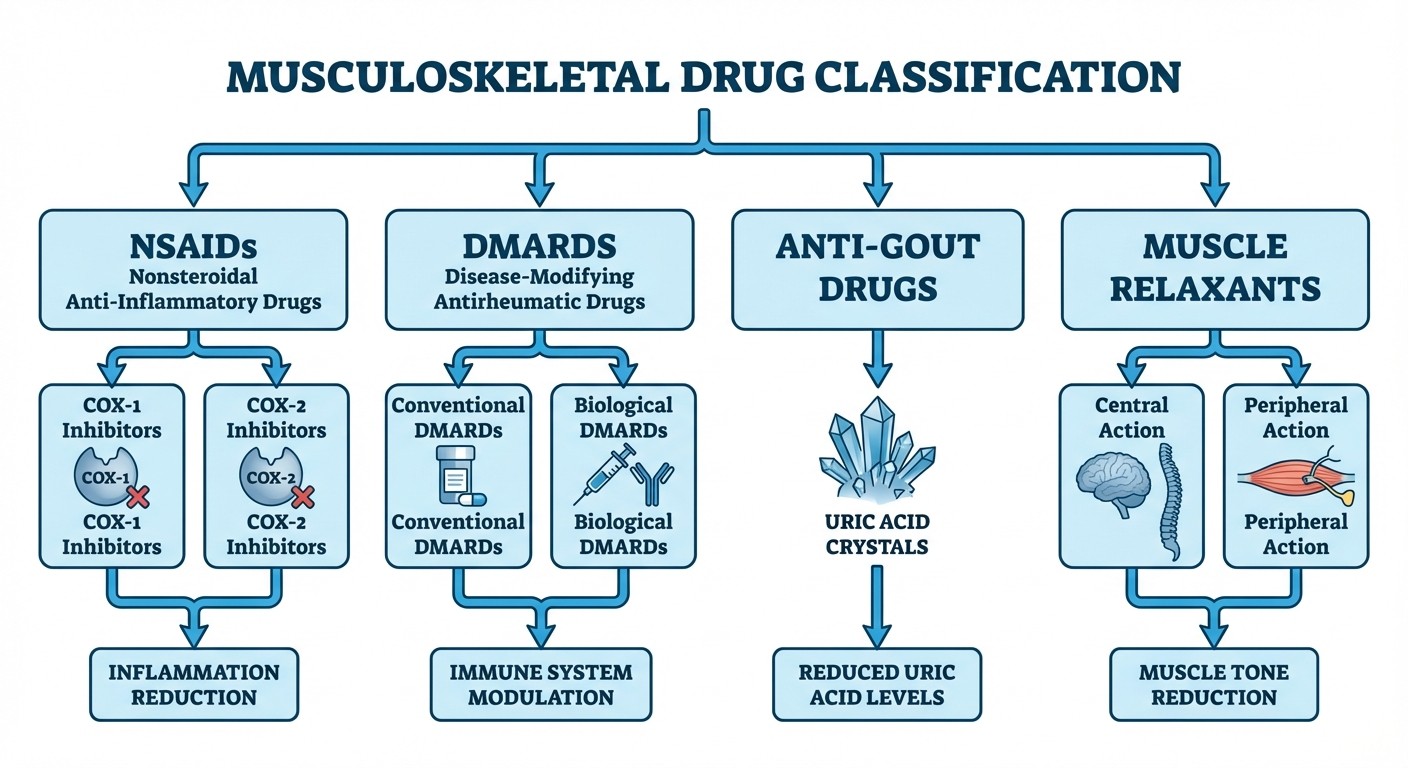
Musculoskeletal pharmacology divides into four major categories:
1. NSAIDs (Non-Steroidal Anti-Inflammatory Drugs) - First-line for inflammation and pain
2. DMARDs (Disease-Modifying Anti-Rheumatic Drugs) - Conventional and biological agents for autoimmune conditions
3. Anti-Gout Drugs - Acute and chronic gout management
4. Muscle Relaxants - Central and peripheral acting agents
Each category has distinct mechanisms, but overlap exists. NSAIDs treat acute gout. Some DMARDs are repurposed cancer drugs. Muscle relaxants work at different sites of the motor pathway. Understanding these connections helps you tackle combination questions that NEET PG loves to ask.
NSAIDs: Non-Steroidal Anti-Inflammatory Drugs
Mechanism of Action
NSAIDs work by inhibiting cyclooxygenase (COX) enzymes, blocking prostaglandin synthesis. Two COX isoforms exist:
COX-1: Constitutive enzyme, protective prostaglandins in GI tract, kidneys, platelets
COX-2: Inducible enzyme, inflammatory prostaglandins at injury sites
Mnemonic: "COX-1 Protects, COX-2 Inflames"
Non-Selective COX Inhibitors
#### Aspirin (Acetylsalicylic Acid)
Mechanism: Irreversible COX-1 and COX-2 inhibition via acetylation
Clinical Uses: Analgesic (325-650mg), anti-inflammatory (3-6g/day), antiplatelet (75-100mg)
Pharmacokinetics: Zero-order kinetics at high doses, 80% protein bound
Adverse Effects: GI bleeding, Reye syndrome (children), salicylism, hyperuricemia
MCQ Mnemonic: "ASPIRIN = A-cetylation Stops Prostaglandin In Reversible INhibition (it's actually irreversible!)"
#### Ibuprofen
Mechanism: Reversible COX-1/COX-2 inhibition, slight COX-1 selectivity
Dose: 400-800mg TID, maximum 3200mg/day
Advantages: Better GI tolerance than aspirin, available OTC
Adverse Effects: Less GI toxicity, fluid retention, hypertension
Drug Interactions: Reduces antihypertensive effect of ACE inhibitors
#### Diclofenac
Mechanism: Potent COX inhibition, excellent tissue penetration
Routes: Oral, topical, intramuscular, IV
Clinical Uses: Rheumatoid arthritis, osteoarthritis, acute pain
Adverse Effects: Higher cardiovascular risk, hepatotoxicity
MCQ Focus: Often asked about topical formulations and liver monitoring
#### Indomethacin
Mechanism: Potent COX inhibition + additional CNS effects
Unique Uses: Patent ductus arteriosus closure (neonates), Bartter syndrome
Adverse Effects: Severe CNS toxicity (headache, dizziness, confusion)
MCQ Mnemonic: "INDO-methacin = IN-Ductus closure + DO-n't use long-term (CNS toxicity)"
After reviewing NSAID mechanisms, Oncourse's adaptive question bank automatically surfaces targeted MCQs on COX selectivity and drug interactions, helping you identify which specific concepts need more focus.
Selective COX-2 Inhibitors
#### Celecoxib
Mechanism: Selective COX-2 inhibition, COX-1 sparing
Advantages: Reduced GI ulceration and bleeding
Cardiovascular Risk: Increased thrombotic events (VIGOR trial)
Contraindications: Sulfonamide allergy, recent MI, stroke
MCQ Focus: Questions often contrast GI vs CV risk profiles
#### Etoricoxib
Mechanism: Highly selective COX-2 inhibitor
Half-life: 22 hours (once daily dosing)
Clinical Uses: Osteoarthritis, rheumatoid arthritis, acute gout
Adverse Effects: Hypertension, edema, cardiovascular events
NSAID Adverse Effects & Contraindications
System | Adverse Effects | Mechanism |
|---|---|---|
GI | Peptic ulcers, bleeding, perforation | COX-1 inhibition → decreased PGE2 |
Renal | AKI, fluid retention, hyperkalemia | Decreased PGE2 and PGI2 |
Cardiovascular | MI, stroke, hypertension | COX-2 selectivity → decreased PGI2 |
Hematologic | Antiplatelet effect, bleeding | COX-1 inhibition → decreased TXA2 |
Respiratory | Bronchospasm (aspirin-sensitive) | Shunting to leukotriene pathway |
High-Yield Contraindications:
Active GI bleeding or peptic ulcer disease
Severe renal or hepatic impairment
Heart failure or recent MI (especially COX-2 selective)
Third trimester pregnancy (premature ductus closure)
Aspirin allergy (cross-reactivity risk)
Practice targeted NSAIDs mechanism MCQs to reinforce these concepts.
DMARDs: Disease-Modifying Anti-Rheumatic Drugs
DMARDs slow or halt rheumatoid arthritis progression by targeting specific immune pathways. They divide into conventional synthetic (csDMARDs), targeted synthetic (tsDMARDs), and biological (bDMARDs).
Conventional DMARDs
#### Methotrexate (MTX)
Mechanism: Dihydrofolate reductase inhibition → impaired DNA synthesis in rapidly dividing cells
Dosing: 7.5-25mg weekly (NEVER daily for RA)
Route: Oral or subcutaneous injection
Monitoring: CBC, liver function, pulmonary function
Adverse Effects: Hepatotoxicity, pulmonary fibrosis, mucositis, teratogenicity
Antidote: Leucovorin (folinic acid) rescue
MCQ Mnemonic: "MTX = Must Take X-tra folate (leucovorin) to avoid toxicity"
#### Sulfasalazine
Mechanism: Anti-inflammatory via inhibition of NF-κB, possibly 5-ASA metabolite
Metabolism: Colonic bacteria cleave to sulfapyridine + 5-aminosalicylic acid
Clinical Uses: Rheumatoid arthritis, ankylosing spondylitis, IBD
Adverse Effects: GI upset, oligospermia, hemolysis (G6PD deficiency)
Monitoring: CBC (reversible oligospermia in males)
#### Hydroxychloroquine
Mechanism: Antimalarial with immunomodulatory effects, lysosomal pH alteration
Dosing: 200-400mg daily, weight-based dosing (<6.5mg/kg/day)
Unique Advantage: Lowest toxicity among DMARDs
Adverse Effects: Retinal toxicity (rare but irreversible), QT prolongation
Monitoring: Annual ophthalmologic examination after 5 years
MCQ Focus: Questions emphasize retinal screening protocols
#### Leflunomide
Mechanism: Dihydroorotate dehydrogenase inhibition → pyrimidine synthesis blockade
Active Metabolite: A771726 (extremely long half-life: 2 weeks)
Loading Dose: 100mg daily × 3 days, then 10-20mg daily
Adverse Effects: Hepatotoxicity, teratogenicity, peripheral neuropathy
Washout: Cholestyramine for rapid elimination (pregnancy planning)
MCQ Mnemonic: "LEF-lunomide = LEF-t pyrimidines behind (blocks synthesis)"
Oncourse's spaced repetition flashcards reinforce DMARD mechanisms at scientifically optimal intervals, ensuring these complex pathways stick before your exam.
Biological DMARDs
#### TNF-α Inhibitors
Infliximab (Chimeric Monoclonal Antibody)
Mechanism: TNF-α neutralization, prevents inflammatory cascade
Route: IV infusion at weeks 0, 2, 6, then every 8 weeks
Clinical Uses: RA, IBD, psoriasis, ankylosing spondylitis
Adverse Effects: Infection risk, lymphoma, lupus-like syndrome
Contraindications: Active TB, hepatitis B reactivation risk
Adalimumab (Fully Human Monoclonal)
Route: Subcutaneous injection every 2 weeks
Advantage: Lower immunogenicity than chimeric antibodies
Pre-treatment Screening: TB, hepatitis B, CBC, LFTs
Etanercept (Fusion Protein)
Mechanism: Soluble TNF receptor that binds and neutralizes TNF-α
Route: Subcutaneous twice weekly or weekly
Unique Feature: Also binds lymphotoxin-α
MCQ Mnemonic for TNF Inhibitors: "TNF = Tumor Necrosis Factor → Terrible iNfection Fear"
#### IL-6 Receptor Antagonist
Tocilizumab
Mechanism: IL-6 receptor blockade → reduced acute phase reactants
Route: IV monthly or subcutaneous weekly
Monitoring: CRP may be unreliable (blocks IL-6 pathway)
Adverse Effects: Elevated transaminases, neutropenia, GI perforation
#### B-Cell Depletion
Rituximab
Mechanism: Anti-CD20 monoclonal antibody → B-cell depletion
Dosing: Two IV infusions separated by 2 weeks
Duration: B-cell depletion lasts 6-9 months
Pre-medication: Methylprednisolone, antihistamines (infusion reactions)
Targeted Synthetic DMARDs (JAK Inhibitors)
#### Tofacitinib
Mechanism: JAK1/JAK3 inhibition → blocks cytokine signaling
Route: Oral (major advantage over biologics)
Adverse Effects: Increased infection risk, thromboembolism, elevated cholesterol
Monitoring: CBC, lipid profile, liver function
Explore comprehensive DMARD pharmacology lessons for detailed mechanism coverage.
Anti-Gout Drugs
Gout management has two phases: acute attack treatment and chronic uric acid lowering. Different drugs target different points in the uric acid pathway.
Acute Gout Treatment
#### Colchicine
Mechanism: Microtubule polymerization inhibition → neutrophil dysfunction
Dosing: 1.2mg at first sign, then 0.6mg 1 hour later (max 1.8mg/24h)
Pharmacokinetics: Hepatic metabolism, renal elimination
Adverse Effects: Diarrhea (dose-limiting), myopathy, bone marrow suppression
Drug Interactions: CYP3A4 inhibitors increase toxicity
MCQ Mnemonic: "COL-chicine = COL-lapses microtubules → COL-on problems (diarrhea)"
#### NSAIDs for Acute Gout
First Choice: Indomethacin 50mg TID × 3-5 days
Alternatives: Ibuprofen, diclofenac, naproxen
Contraindications: Renal disease, heart failure, GI bleeding
#### Corticosteroids
Indications: NSAIDs and colchicine contraindicated/ineffective
Routes: Oral prednisolone, intra-articular injection, IV methylprednisolone
Dosing: Prednisolone 30-40mg daily × 5-7 days
Chronic Uric Acid Lowering Therapy
#### Allopurinol
Mechanism: Xanthine oxidase inhibition → reduces uric acid production
Dosing: Start 100mg daily, titrate to serum urate <6mg/dL
Pharmacokinetics: Active metabolite oxypurinol (longer half-life)
Adverse Effects: Severe cutaneous reactions (HLA-B*5801 association)
Drug Interactions: Increases toxicity of azathioprine, 6-MP
MCQ Focus: Often asked about azathioprine interaction mechanism
#### Febuxostat
Mechanism: Selective xanthine oxidase inhibition (non-purine analog)
Advantages: Can use in mild-moderate renal impairment
Dosing: 40-80mg daily, more potent than allopurinol
Adverse Effects: Cardiovascular events (FDA warning)
#### Probenecid
Mechanism: Uric acid reabsorption inhibition in proximal tubule
Indication: Underexcretion of uric acid (normal production)
Contraindications: CKD (GFR <50), history of kidney stones
Drug Interactions: Reduces penicillin excretion
MCQ Mnemonic: "PRO-benecid = PRO-motes uric acid elimination"
#### Rasburicase
Mechanism: Recombinant urate oxidase → converts uric acid to allantoin
Indication: Tumor lysis syndrome prevention
Contraindications: G6PD deficiency (hemolysis risk)
Route: IV infusion in oncology settings
Practice anti-gout drug MCQs to master these high-yield mechanisms.
Gout Drug Selection Algorithm
Clinical Scenario | First Choice | Alternative | Contraindication |
|---|---|---|---|
Acute monoarticular | Colchicine 1.2mg→0.6mg | Indomethacin 50mg TID | Renal failure |
Acute polyarticular | NSAIDs | Prednisolone 40mg | Active GI bleeding |
Chronic prophylaxis | Allopurinol 100mg→target | Febuxostat 40-80mg | HLA-B*5801 positive |
Renal impairment | Febuxostat | Probenecid (if GFR>50) | Severe CKD |
Tumor lysis syndrome | Rasburicase | Allopurinol (less effective) | G6PD deficiency |
Muscle Relaxants
Muscle relaxants work at different levels of motor control. Central agents modify CNS pathways, while peripheral agents target the neuromuscular junction or muscle fibers directly.
Centrally Acting Muscle Relaxants
#### Baclofen
Mechanism: GABA-B receptor agonist in spinal cord
Clinical Uses: Spasticity (cerebral palsy, spinal cord injury), trigeminal neuralgia
Route: Oral or intrathecal pump
Pharmacokinetics: Crosses blood-brain barrier poorly (hence intrathecal route)
Adverse Effects: Sedation, weakness, withdrawal seizures (abrupt discontinuation)
MCQ Mnemonic: "BAC-lofen = BAC-k pain via GABA-B receptors"
#### Tizanidine
Mechanism: α2-adrenergic agonist → reduced presynaptic excitation
Clinical Uses: Muscle spasticity, tension headaches
Pharmacokinetics: Short half-life (2.5 hours), hepatic metabolism
Adverse Effects: Hypotension, sedation, hepatotoxicity
Monitoring: Liver function tests (risk of hepatotoxicity)
#### Cyclobenzaprine
Mechanism: CNS depression, structurally similar to tricyclic antidepressants
Clinical Uses: Acute muscle spasm, fibromyalgia
Pharmacokinetics: Long half-life (18 hours), extensive first-pass metabolism
Adverse Effects: Anticholinergic effects (dry mouth, urinary retention)
Contraindications: Recent MAO inhibitor use, hyperthyroidism
Performance analytics through Oncourse help identify which muscle relaxant mechanisms you're missing most frequently, enabling targeted revision in your final weeks.
Peripherally Acting Muscle Relaxants
#### Dantrolene
Mechanism: Blocks ryanodine receptors → prevents calcium release from sarcoplasmic reticulum
Unique Indication: Malignant hyperthermia treatment and prevention
Other Uses: Spasticity (cerebral palsy, multiple sclerosis)
Route: Oral for spasticity, IV for malignant hyperthermia
Adverse Effects: Hepatotoxicity (major concern), muscle weakness
MCQ Focus: Always think malignant hyperthermia when you see dantrolene
MCQ Mnemonic: "DAN-trolene = DAN-gerous hyperthermia rescue"
Neuromuscular Junction Blockers (Brief Review)
While not traditional muscle relaxants, these appear in musculoskeletal pharmacology sections:
#### Succinylcholine
Mechanism: Depolarizing NMJ blocker (ACh receptor agonist)
Clinical Use: Rapid sequence intubation
Adverse Effects: Malignant hyperthermia, hyperkalemia
Reversal: None (wait for metabolism)
#### Rocuronium
Mechanism: Non-depolarizing NMJ blocker (ACh receptor competitive antagonist)
Advantages: Rapid onset, predictable duration
Reversal: Sugammadex (specific reversal agent)
Review comprehensive pharmacology concepts to connect these mechanisms with clinical applications.
High-Yield Drug Interactions & Contraindications
Major Drug Interactions
Drug | Interacting Agent | Mechanism | Clinical Significance |
|---|---|---|---|
Methotrexate | NSAIDs | Reduced renal clearance | Increased MTX toxicity |
Allopurinol | Azathioprine | Xanthine oxidase inhibition | 6-MP toxicity |
NSAIDs | ACE inhibitors | Reduced prostaglandin synthesis | Hypertension, renal dysfunction |
Colchicine | CYP3A4 inhibitors | Increased plasma levels | Severe toxicity |
Warfarin | NSAIDs | Protein binding displacement | Bleeding risk |
Pregnancy & Lactation Considerations
Contraindicated in Pregnancy:
Methotrexate (Category X - teratogenic)
Leflunomide (Category X - requires washout)
NSAIDs in third trimester (ductus arteriosus closure)
Mycophenolate (Category D - teratogenic)
Relatively Safe Options:
Sulfasalazine (Category B - folate supplementation)
Hydroxychloroquine (Category C - continue if needed)
Prednisolone (Category B - crosses placenta minimally)
Low-dose aspirin (Category B up to 32 weeks)
MCQ Strategy & High-Yield Topics
Most Frequently Tested Concepts
1. NSAID mechanisms and selectivity - Distinguish COX-1 vs COX-2 effects
2. Methotrexate monitoring - Know leucovorin rescue and contraindications
3. Allopurinol interactions - Especially with azathioprine and 6-MP
4. Malignant hyperthermia - Dantrolene mechanism and indication
5. TNF inhibitor screening - TB and hepatitis B reactivation risks
6. Colchicine toxicity - Diarrhea and drug interactions
7. Pregnancy categories - Which DMARDs are safe/contraindicated
Question Pattern Recognition
NEET PG loves to test:
Mechanism of action with clinical correlation
Adverse effect patterns and monitoring requirements
Drug interactions with clinical significance
Contraindications in specific populations
Dosing schedules and administration routes
INICET frequently asks:
First-line therapy selection for specific conditions
Monitoring requirements for high-risk drugs
Alternative agents when first choice is contraindicated
Combination therapy rationales
Study these patterns with targeted pharmacology flashcards that use spaced repetition to lock in these concepts.
Frequently Asked Questions
Which NSAIDs are safest for patients with cardiovascular disease?
Non-selective NSAIDs like ibuprofen carry less CV risk than COX-2 selective agents. However, naproxen has the best cardiovascular safety profile among NSAIDs. Consider topical preparations to minimize systemic exposure.
How long should DMARDs be continued in rheumatoid arthritis?
DMARDs are typically lifelong therapy. Methotrexate remains first-line, often combined with biologics in severe disease. Stopping DMARDs usually leads to disease flare within months.
Can allopurinol be started during an acute gout attack?
No, starting allopurinol during acute gout can worsen the attack. Wait until inflammation resolves, then start with colchicine prophylaxis for 3-6 months while titrating allopurinol to target uric acid levels.
What is the antidote for methotrexate toxicity?
Leucovorin (folinic acid) rescue. Give within 24-48 hours of MTX overdose. Dose depends on MTX levels - higher MTX levels require higher leucovorin doses and longer duration.
Which muscle relaxant is used for malignant hyperthermia?
Dantrolene is the specific treatment for malignant hyperthermia. It blocks calcium release from sarcoplasmic reticulum, preventing the hypermetabolic crisis. IV administration is critical - have it readily available in ORs.
Are biologics safe during pregnancy?
Most biologics are Category B or C. TNF inhibitors like adalimumab and infliximab can be continued through pregnancy if disease activity is high. Rituximab should be avoided due to B-cell depletion in the fetus.
This comprehensive glossary gives you the mechanism clarity and MCQ strategy needed to excel in musculoskeletal pharmacology. The key is connecting drug mechanisms to clinical applications - exactly what NEET PG and INICET test most heavily.
Prepare smarter with Oncourse AI — adaptive MCQs, spaced repetition, and AI explanations built for NEET PG 2026. Download free on Android and iOS.