Autonomic/CV Drugs

On this page
🎯 Cardiovascular Drug Arsenal: The Command Center
Master the autonomic and cardiovascular pharmacology arsenal, and you possess the power to manipulate every heartbeat, blood vessel, and rhythm disturbance. This lesson builds your foundation from receptor mechanisms through clinical decision-making, integrating 13 drug classes with >50 specific agents. You'll develop pattern recognition for life-threatening toxicities, understand why certain combinations kill while others cure, and build the clinical judgment to choose the right drug at the right moment. Each mechanism connects directly to bedside decisions-from managing cardiogenic shock to terminating lethal arrhythmias.
🔌 The Receptor Landscape: Your Pharmacologic GPS
Understanding cardiovascular drug actions requires mastering the receptor architecture that controls every vessel and myocyte. The autonomic nervous system operates through 5 primary receptor families-alpha-1, alpha-2, beta-1, beta-2, and muscarinic-each triggering distinct cascades that either accelerate or brake cardiac function. Selectivity determines whether a drug saves or kills your patient.
Adrenergic Receptor Architecture
- Beta-1 Receptors (cardiac dominance: 80% myocardial)
- Location: SA node, AV node, ventricular myocytes
- Mechanism: Gs protein → ↑ cAMP → ↑ Ca²⁺ influx
- Effects: ↑ heart rate (+chronotropy), ↑ contractility (+inotropy), ↑ conduction velocity (+dromotropy)
- Clinical threshold: Blockade reduces HR by 15-20%, contractility by 25-30%
- Beta-2 Receptors (vascular/bronchial: 60% smooth muscle)
- Location: Bronchioles, peripheral vessels, uterus
- Mechanism: Gs protein → ↑ cAMP → smooth muscle relaxation
- Effects: Vasodilation, bronchodilation, ↓ peripheral resistance by 20-40%
- Clinical pearl: Stimulation can ↑ HR via reflex tachycardia despite no direct cardiac effect
- Alpha-1 Receptors (vascular tone: 90% arteriolar)
- Location: Arterioles, veins, bladder neck, pupillary dilator
- Mechanism: Gq protein → ↑ IP₃/DAG → ↑ intracellular Ca²⁺
- Effects: Vasoconstriction, ↑ SVR by 40-60%, ↑ BP by 20-50 mmHg
- Clinical use: Shock states requiring peripheral vasoconstriction
📌 Remember: "BATS-1 in the HEART" - Beta-1 receptors dominate cardiac tissue (>75%), while Alpha-1 receptors control Tone in Smooth muscle vessels. Beta-2 receptors relax Bronchi and Blood vessels.
- Alpha-2 Receptors (presynaptic regulation: 70% CNS)
- Location: Presynaptic terminals, CNS (medulla), pancreatic beta cells
- Mechanism: Gi protein → ↓ cAMP → ↓ norepinephrine release
- Effects: Negative feedback, ↓ sympathetic outflow, ↓ BP by 10-20 mmHg
- Clinical relevance: Clonidine exploits this for hypertension management
- Muscarinic Receptors (parasympathetic control)
- M₂ subtype: SA/AV nodes (>80% cardiac muscarinic)
- Mechanism: Gi protein → ↓ cAMP + ↑ K⁺ efflux
- Effects: ↓ HR by 20-30%, ↓ AV conduction, minimal inotropy effect
- Atropine blockade: ↑ HR by 30-40 bpm in healthy adults
⭐ Clinical Pearl: Beta-1 selectivity is dose-dependent-metoprolol is 75% selective at 50 mg but loses selectivity at >200 mg/day, blocking beta-2 receptors and causing bronchospasm in 15-20% of asthmatics.
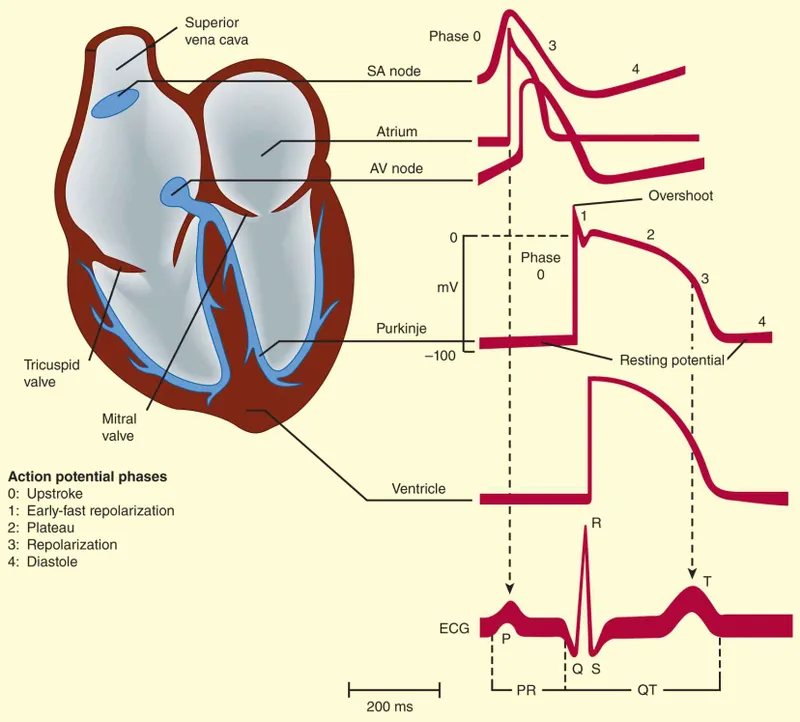
Ion Channel Architecture for Antiarrhythmics
The cardiac action potential depends on 4 sequential ion channel families that create the electrical signature of each heartbeat. Understanding which drugs target which phase predicts both therapeutic effects and toxicities.
| Phase | Primary Current | Channel Type | Duration | Drug Classes | Clinical Effect |
|---|---|---|---|---|---|
| 0 (Depolarization) | INa (fast sodium) | Nav1.5 | 1-2 ms | Class I (Na⁺ blockers) | ↓ Conduction velocity, ↓ automaticity |
| 1 (Early repolarization) | Ito (transient outward K⁺) | Kv4.3 | 10-20 ms | None clinically | Minimal therapeutic target |
| 2 (Plateau) | ICaL (L-type Ca²⁺) | Cav1.2 | 100-200 ms | Class IV (CCBs) | ↓ AV conduction, ↓ contractility |
| 3 (Repolarization) | IKr, IKs (delayed rectifier K⁺) | hERG, KCNQ1 | 200-300 ms | Class III (K⁺ blockers) | ↑ Refractory period, ↑ QT interval |
| 4 (Resting) | IK1 (inward rectifier K⁺) | Kir2.1 | Variable | Digoxin (indirect) | ↓ Automaticity in SA node |
💡 Master This: Class I drugs subdivide by recovery kinetics from sodium channel blockade-Class IA recovers in intermediate time (200-1000 ms), IB recovers fast (<200 ms, targets ischemic tissue), IC recovers slow (>1000 ms, dangerous in structural heart disease). This explains why lidocaine (IB) works in MI but flecainide (IC) kills post-MI patients.
- Vaughan Williams Classification (mechanism-based)
- Class I: Sodium channel blockade
- IA: Moderate blockade + ↑ APD (quinidine, procainamide, disopyramide)
- IB: Weak blockade + ↓ APD (lidocaine, mexiletine)
- IC: Strong blockade, no APD change (flecainide, propafenone)
- Class II: Beta-adrenergic blockade (metoprolol, esmolol, propranolol)
- Class III: Potassium channel blockade (amiodarone, sotalol, dofetilide, ibutilide)
- Class IV: Calcium channel blockade (verapamil, diltiazem)
- Other: Adenosine, digoxin, magnesium
- Class I: Sodium channel blockade
⚠️ Warning: The CAST trial (1989) showed Class IC agents (flecainide, encainide) increased mortality by 2.5-fold in post-MI patients despite suppressing >80% of PVCs. Mechanism: Slowed conduction creates reentrant circuits in scarred myocardium. Never use IC agents in structural heart disease.
Connect these receptor and channel foundations through the clinical drug classes that exploit them for therapeutic benefit, starting with agents that manipulate contractility and rhythm.
🔌 The Receptor Landscape: Your Pharmacologic GPS
💪 Inotropic Agents: The Contractility Command Center
When cardiac output fails, inotropic agents become life-saving tools that boost myocardial contractility through 3 distinct mechanisms: increasing intracellular calcium, enhancing calcium sensitivity, or preventing cAMP breakdown. Each mechanism carries unique benefits and toxicities that determine clinical selection.
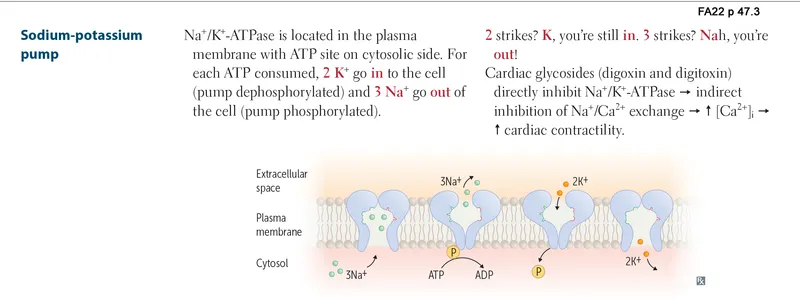
Cardiac Glycosides: The Ancient Pump Inhibitor
Digoxin, derived from Digitalis purpurea, remains relevant after >200 years through its unique mechanism: Na⁺/K⁺-ATPase inhibition → ↑ intracellular Na⁺ → ↓ Na⁺/Ca²⁺ exchanger activity → ↑ intracellular Ca²⁺ → ↑ contractility by 15-25%.
- Pharmacokinetics (narrow therapeutic index: 0.5-2.0 ng/mL)
- Bioavailability: 70-80% oral (tablets), 90-100% IV
- Half-life: 36-48 hours (renal elimination 70-80%)
- Volume of distribution: 5-7 L/kg (extensive tissue binding)
- Loading dose: 0.5-1.0 mg IV divided over 24 hours
- Maintenance: 0.125-0.25 mg daily (adjust for renal function)
- Clinical indications (limited modern use)
- Heart failure with reduced EF: Improves symptoms, no mortality benefit
- Atrial fibrillation rate control: Target HR <80 bpm at rest, <110 bpm with activity
- Systolic dysfunction + AF: Reduces hospitalizations by 28% (DIG trial)
- Mechanism of AV nodal effects
- Direct: ↑ vagal tone (muscarinic stimulation) → ↓ AV conduction
- Indirect: ↓ sympathetic outflow from baroreceptor sensitization
- Net effect: Prolongs AV nodal refractory period, ↓ ventricular rate by 20-30%
📌 Remember: "DIG TOXICITY" - Dysrhythmias (bidirectional VT, pathognomonic), Increased automaticity (PAT with block), GI symptoms (nausea >80%, anorexia), Threshold lowered (hypokalemia <3.5 mEq/L ↑ toxicity risk 3-fold), Ocular changes (yellow-green halos), Xanthopsia (yellow vision), Increased PR interval, Confusion, Infusion of digoxin-specific Fab, Treated with K⁺ replacement, Yellow digoxin level >2.0 ng/mL.
- Digoxin toxicity management (potentially lethal)
- Predisposing factors: Hypokalemia <3.5 mEq/L (↑ binding), hypomagnesemia, hypercalcemia, renal failure (↓ clearance 50-70%)
- ECG findings: ↑ PR interval, ↓ QT interval, ST "scooping" (therapeutic), bradycardia, heart block (toxic)
- Arrhythmias: PAT with 2:1 block (classic), accelerated junctional rhythm, bidirectional VT (pathognomonic)
- Antidote: Digoxin-specific Fab fragments (DigiFab)
- Indication: Life-threatening arrhythmia, K⁺ >5.5 mEq/L, digoxin >10 ng/mL acute ingestion
- Dose: # vials = (serum level × weight in kg) / 100
- Response time: 20-30 minutes for arrhythmia resolution
⭐ Clinical Pearl: Quinidine, verapamil, and amiodarone double digoxin levels through P-glycoprotein inhibition-reduce digoxin dose by 50% when starting these drugs. Spironolactone interferes with digoxin assays, causing falsely elevated levels by 0.4-0.8 ng/mL.
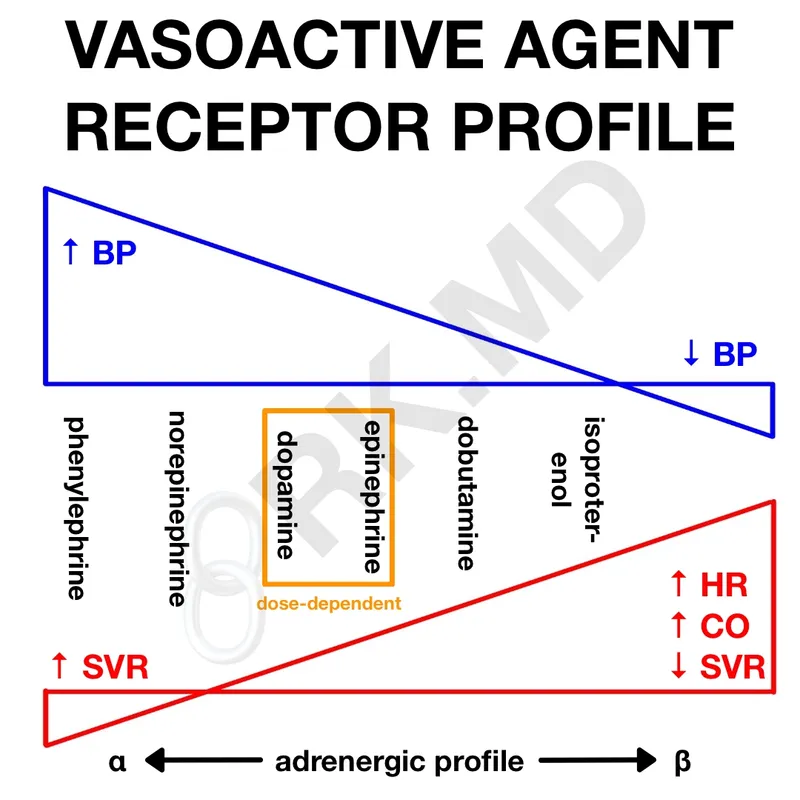
Beta-Adrenergic Agonists: The Catecholamine Cavalry
Sympathomimetics boost contractility through beta-1 receptor stimulation, with selectivity determining side effect profiles. Critical in cardiogenic shock and acute decompensated heart failure.
| Agent | Beta-1 | Beta-2 | Alpha-1 | Dose Range | Contractility ↑ | Clinical Use |
|---|---|---|---|---|---|---|
| Dobutamine | ++++ | ++ | + | 2.5-20 mcg/kg/min | +40-60% | Cardiogenic shock, low CO states |
| Dopamine | +++ (high dose) | + | +++ (high dose) | 2-20 mcg/kg/min | +30-50% | Shock with hypotension |
| Epinephrine | ++++ | ++++ | ++++ | 0.05-0.5 mcg/kg/min | +60-80% | Cardiac arrest, anaphylaxis |
| Isoproterenol | ++++ | ++++ | 0 | 2-10 mcg/min | +50-70% | Bradycardia, heart block |
| Norepinephrine | +++ | + | ++++ | 0.05-0.3 mcg/kg/min | +20-30% | Septic shock (vasoconstriction primary) |
- Dobutamine (synthetic catecholamine)
- Mechanism: Beta-1 >> beta-2 > alpha-1 (net vasodilation at therapeutic doses)
- Hemodynamics: ↑ CO by 40-60%, ↓ PCWP by 30-40%, minimal BP change
- Tachyphylaxis: Develops in 48-72 hours (downregulation of beta receptors 40-50%)
- Adverse effects: Tachycardia (>20% patients), ectopy (15-20%), ↑ myocardial O₂ demand
- Contraindication: Obstructive cardiomyopathy (↑ gradient), tachyarrhythmias
- Dopamine (dose-dependent receptor activation)
- Low dose (2-5 mcg/kg/min): D₁ receptors → renal/splanchnic vasodilation (controversial benefit)
- Medium dose (5-10 mcg/kg/min): Beta-1 >> alpha-1 → ↑ inotropy + ↑ HR
- High dose (>10 mcg/kg/min): Alpha-1 >>> beta-1 → vasoconstriction dominates
- Clinical pearl: No longer recommended for "renal protection"-zero evidence for preventing AKI
💡 Master This: Dobutamine increases contractility without significant vasoconstriction, making it ideal for cardiogenic shock with adequate BP (SBP >90 mmHg). Dopamine causes more tachycardia and arrhythmias (30% vs 15%) but provides vasoconstriction when BP is critically low. In septic shock, norepinephrine is first-line over dopamine due to lower mortality (SOAP II trial: 28-day mortality 49% vs 52%, p=0.03).
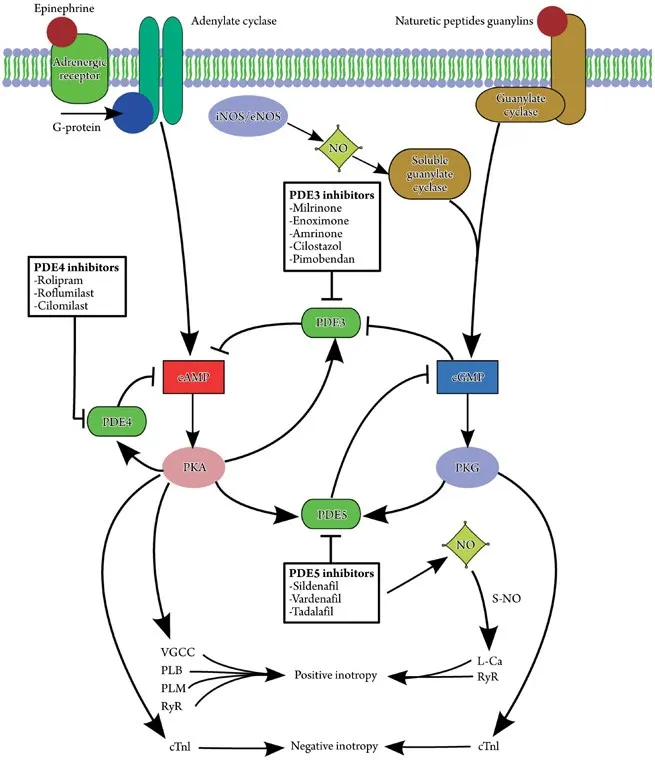
Phosphodiesterase-3 Inhibitors: The cAMP Lifeguards
PDE-3 inhibitors block cAMP degradation, causing dual inotropic-vasodilator effects ("inodilators"). Unlike beta-agonists, they bypass beta-receptors and avoid tachyphylaxis.
- Milrinone (primary agent in clinical use)
- Mechanism: PDE-3 inhibition → ↑ cAMP → ↑ Ca²⁺ influx (myocytes) + ↓ Ca²⁺ (smooth muscle)
- Hemodynamics: ↑ CO by 30-40%, ↓ SVR by 20-30%, ↓ PCWP by 30-40%
- Loading dose: 50 mcg/kg over 10 minutes (optional, causes hypotension in 20-30%)
- Maintenance: 0.375-0.75 mcg/kg/min (adjust for renal function, 85% renal elimination)
- Half-life: 2-3 hours (prolonged to 6-8 hours in renal failure)
- Clinical applications
- Acute decompensated heart failure: Short-term use (<48-72 hours)
- Bridge to transplant or LVAD: Continuous infusion for weeks
- Right heart failure post-cardiac surgery: Reduces PVR by 25-35%
- Contraindication: Severe aortic/pulmonic stenosis (afterload reduction worsens gradient)
- Inamrinone (formerly amrinone, rarely used)
- Similar mechanism to milrinone
- Dose: 0.75 mg/kg bolus, then 5-10 mcg/kg/min
- Adverse effects: Thrombocytopenia (10-15%, reversible), hepatotoxicity (rare)
- Largely replaced by milrinone due to side effect profile
⚠️ Warning: Long-term PDE-3 inhibitor use (oral milrinone) increased mortality by 28% in PROMISE trial-reserved for acute use only or palliative bridge to transplant. Mechanism: ↑ intracellular Ca²⁺ → ↑ arrhythmias + ↑ myocardial O₂ demand.
⭐ Clinical Pearl: Milrinone is superior to dobutamine in patients on chronic beta-blockers because it bypasses beta-receptors. Combination therapy (dobutamine + milrinone) provides additive effects through different mechanisms, useful in refractory cardiogenic shock.
Connect inotropic mechanisms to the rhythm control agents that prevent dangerous arrhythmias while preserving contractility.
💪 Inotropic Agents: The Contractility Command Center
⚡ Sodium Channel Blockers: The Class I Rhythm Controllers
Class I antiarrhythmics suppress arrhythmias by blocking cardiac sodium channels, but subclass distinctions determine which arrhythmias respond and which patients die. The Vaughan Williams classification subdivides Class I by recovery kinetics and action potential duration (APD) effects.
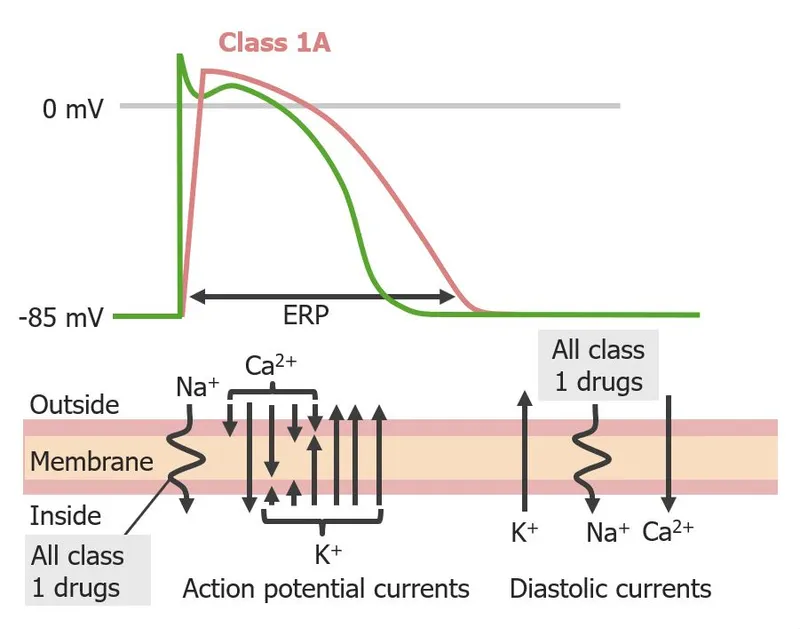
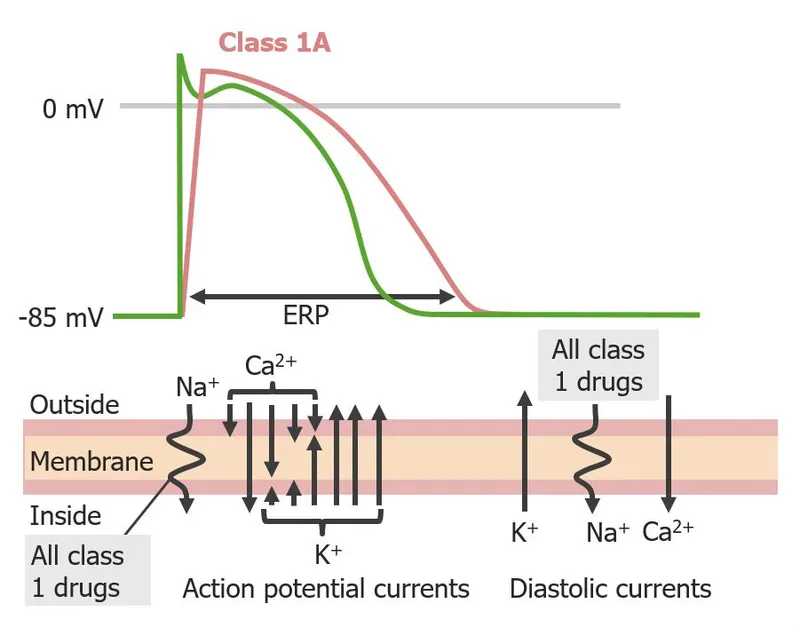
Class IA: The APD Prolongers
Class IA agents block sodium channels with intermediate recovery kinetics (200-1000 ms) and prolong APD through additional potassium channel blockade. This dual action creates both therapeutic benefit and proarrhythmic risk.
- Quinidine (the original antiarrhythmic, rarely used)
- Mechanism: INa blockade + IKr blockade → ↑ QRS by 10-20%, ↑ QT by 15-25%
- Indications: Atrial fibrillation conversion (50-60% success), Brugada syndrome (off-label)
- Adverse effects: Cinchonism (tinnitus, blurred vision, >30% at therapeutic levels), diarrhea (35%)
- Proarrhythmia: Torsades de pointes in 2-8% (QT prolongation, ↓ K⁺)
- Drug interactions: P-glycoprotein inhibitor → ↑ digoxin levels 2-fold
- Procainamide (IV preferred for acute conversion)
- Mechanism: INa + IKr blockade, also blocks ganglionic transmission
- Dose: 15-18 mg/kg IV load over 30-60 min (max 50 mg/min), then 1-4 mg/min infusion
- Conversion rate: 60-80% for atrial fibrillation/flutter, 40-60% for VT
- Metabolism: N-acetylprocainamide (NAPA, active metabolite with Class III properties)
- Toxicity: Drug-induced lupus (20-30% with chronic use, 60-70% develop ANA), agranulocytosis (0.5%)
📌 Remember: "QUEEN PROCLAIMS DISOPYRAMIDE" for Class IA agents - QUinidine causes cinEhonism and diErrhea (35%), NAPA is PROCainamide's active metabolite causing Lupus (ANA positive 60-70%), Anticholinergic effects dominate with dIsopyraMide (urinary retention 10-15%, dry Mouth 40%), all prolong QT and cause Torsades (2-8%).
- Disopyramide (negative inotrope with anticholinergic effects)
- Mechanism: INa + IKr blockade + muscarinic receptor blockade
- Unique property: Negative inotropy (↓ contractility 20-30%)
- Indications: Hypertrophic cardiomyopathy with obstruction (↓ gradient 30-50%)
- Adverse effects: Anticholinergic (dry mouth 40%, urinary retention 10-15%, constipation 20%)
- Contraindication: Heart failure (worsens EF), glaucoma, BPH
Class IB: The Ischemia Specialists
Class IB agents have fast recovery kinetics (<200 ms) and shorten APD, selectively targeting ischemic or depolarized tissue where sodium channels remain partially inactivated. This selectivity makes them safe in acute MI.
- Lidocaine (IV only, first-pass metabolism >70%)
- Mechanism: Preferential binding to open/inactivated sodium channels → use-dependent blockade
- Indications: Ventricular arrhythmias (VT, VF) in acute MI setting
- Dose: 1-1.5 mg/kg IV bolus, then 1-4 mg/min infusion
- Therapeutic level: 1.5-5.0 mcg/mL (toxicity >5 mcg/mL)
- CNS toxicity: Paresthesias, drowsiness, seizures (>8 mcg/mL), respiratory arrest (>10 mcg/mL)
- Metabolism: Hepatic (CYP3A4) → reduce dose 50% in liver disease or heart failure (↓ hepatic flow)
- Mexiletine (oral lidocaine analog)
- Mechanism: Identical to lidocaine, oral bioavailability 80-90%
- Indications: Chronic ventricular arrhythmias, neuropathic pain (off-label)
- Dose: 150-300 mg TID (start 150 mg TID, titrate q3-5 days)
- Adverse effects: GI upset (40%, take with food), tremor (15%), dizziness (25%)
💡 Master This: Class IB agents shorten APD and don't prolong QT-opposite of Class IA/IC. This makes them safe in ischemia where prolonged repolarization increases vulnerability. Lidocaine's use-dependent blockade means it preferentially blocks rapidly firing cells (ectopic foci) while sparing normal conduction, explaining efficacy in VT without causing heart block.
Class IC: The Dangerous Rhythm Killers
Class IC agents cause profound sodium channel blockade with slow recovery kinetics (>1000 ms) and no APD change. Powerful but deadly in structural heart disease.
- Flecainide (most potent Na⁺ blocker)
- Mechanism: Marked ↓ conduction velocity (↑ QRS 20-30%), minimal APD effect
- Indications: Paroxysmal atrial fibrillation in structurally normal hearts ("pill-in-pocket" 70-80% success)
- Dose: 50-200 mg BID (start 50 mg BID, max 400 mg/day)
- Contraindication: Any structural heart disease (post-MI, LV dysfunction, LVH)
- CAST trial: ↑ mortality 2.5-fold post-MI despite suppressing >80% PVCs
- Propafenone (additional beta-blocking properties)
- Mechanism: INa blockade + weak beta-blockade + weak Ca²⁺ blockade
- Indications: Atrial fibrillation, PSVT in normal hearts
- Dose: 150-300 mg TID (immediate-release), 225-425 mg BID (extended-release)
- Metabolism: CYP2D6 (poor metabolizers have 2-fold higher levels + more beta-blockade)
- Adverse effects: Metallic taste (5-10%), bronchospasm (avoid in asthma)
⚠️ Warning: CAST trial (1989, NEJM) terminated early when flecainide/encainide caused 2.5-fold mortality increase post-MI. Mechanism: Slowed conduction in scarred tissue → unidirectional block → reentrant VT. Absolute contraindication: structural heart disease, CAD, LV dysfunction, LVH.
| Class IA | Class IB | Class IC |
|---|---|---|
| Intermediate recovery (200-1000 ms) | Fast recovery (<200 ms) | Slow recovery (>1000 ms) |
| Prolongs APD + QT | Shortens APD, no QT change | No change in APD/QT |
| ↑ QRS 10-20%, ↑ QT 15-25% | Minimal QRS widening | ↑ QRS 20-30%, no QT change |
| Torsades risk 2-8% | No torsades risk | No torsades, but VT/VF in structural disease |
| Atrial + ventricular arrhythmias | Ventricular only (ischemia-selective) | Atrial fibrillation in normal hearts |
| Quinidine, procainamide, disopyramide | Lidocaine, mexiletine | Flecainide, propafenone |
Connect sodium channel blockade principles to the beta-blockers that control rhythm through rate reduction and sympathetic antagonism.
⚡ Sodium Channel Blockers: The Class I Rhythm Controllers
🛡️ Beta-Blockers: The Class II Protective Shield
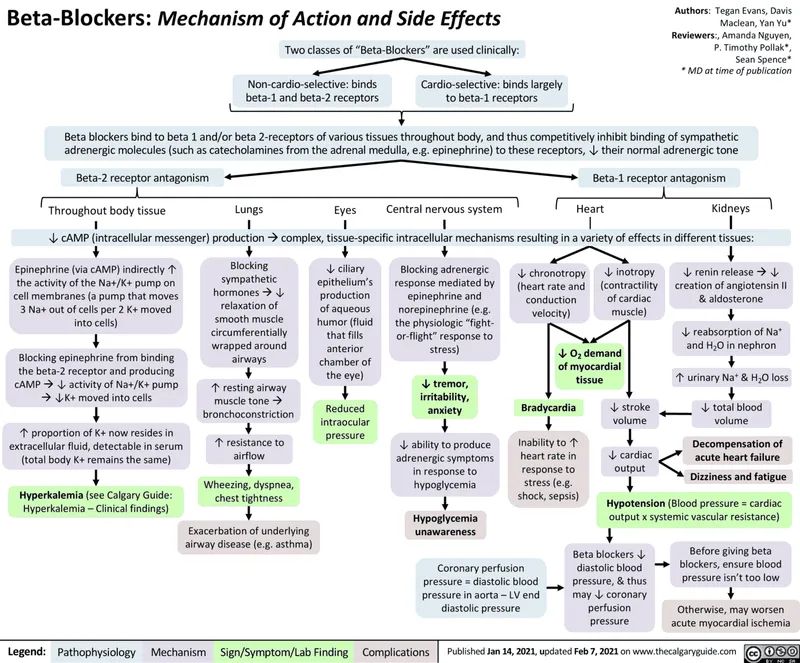
Beta-adrenergic antagonists prevent arrhythmias by blocking sympathetic drive to the heart, reducing automaticity, conduction velocity, and triggered activity. Unlike sodium channel blockers, beta-blockers provide mortality benefit in heart failure and post-MI patients.
Mechanism of Antiarrhythmic Action
- SA node effects
- ↓ Phase 4 slope (↓ If current) → ↓ automaticity → ↓ HR by 15-25%
- ↓ Catecholamine-triggered ectopy → prevents exercise-induced arrhythmias
- AV node effects
- Prolongs AV nodal refractoriness → ↓ ventricular rate in AF/flutter by 25-35%
- Terminates AVNRT/AVRT by blocking anterograde limb (success 30-50%, inferior to adenosine)
- Ventricular effects
- ↓ Triggered activity from ↓ intracellular Ca²⁺ → prevents catecholamine-induced VT
- ↓ Myocardial O₂ demand → ↓ ischemia-related arrhythmias
- Post-MI mortality reduction: 23% (meta-analysis, primarily through arrhythmia prevention)
Beta-Blocker Selection: Selectivity and Properties
Selectivity, lipophilicity, and additional properties determine clinical applications and side effect profiles.
| Agent | Beta-1 Selectivity | Lipophilicity | Half-Life | Unique Properties | Arrhythmia Dose |
|---|---|---|---|---|---|
| Metoprolol | Moderate (75% at low dose) | Lipophilic | 3-7 hours | None | 25-100 mg BID (PO), 2.5-5 mg q5min (IV) |
| Esmolol | High (beta-1:beta-2 = 33:1) | Lipophilic | 9 minutes | Ultra-short acting | 500 mcg/kg load, 50-300 mcg/kg/min |
| Propranolol | None (nonselective) | Highly lipophilic | 3-6 hours | Membrane stabilizing | 10-80 mg TID-QID (PO), 1-3 mg slow IV |
| Atenolol | Moderate (beta-1:beta-2 = 20:1) | Hydrophilic | 6-9 hours | Renal elimination | 25-100 mg daily |
| Carvedilol | None + alpha-1 blockade | Lipophilic | 7-10 hours | Vasodilation, antioxidant | 3.125-25 mg BID |
| Sotalol | None + Class III (K⁺ block) | Hydrophilic | 12 hours | QT prolongation, torsades risk | 80-160 mg BID |
- Esmolol (ultra-short acting, IV only)
- Indication: Acute rate control in perioperative/ICU settings
- Metabolism: Red blood cell esterases → 9-minute half-life
- Advantage: Titratable, rapid offset if adverse effects occur
- Dose: 500 mcg/kg load over 1 min, then 50-300 mcg/kg/min (titrate by 50 mcg/kg/min q5min)
- Use case: Intraoperative tachycardia, acute AF with RVR, aortic dissection
- Metoprolol (first-line for most arrhythmias)
- Selectivity: Beta-1 selective at doses ≤100 mg/day (75% selective)
- Formulations: Tartrate (immediate-release, BID-TID) vs succinate (extended-release, daily)
- IV dose: 2.5-5 mg over 2 min, repeat q5min to max 15 mg
- Conversion: IV to PO ratio 1:2.5 (5 mg IV ≈ 12.5 mg PO)
- Post-MI: Reduces mortality 23%, start within 24 hours if hemodynamically stable
- Propranolol (nonselective, lipophilic)
- Properties: Crosses BBB → CNS effects (tremor reduction, migraine prophylaxis)
- Membrane stabilizing: "Quinidine-like" at high doses (not clinically significant)
- Arrhythmia use: Long QT syndrome, thyroid storm, catecholaminergic polymorphic VT
- Dose: 10-80 mg TID-QID (PO), 1-3 mg IV at 1 mg/min
📌 Remember: "BEAM" for beta-blocker benefits - Blockade prevents post-MI death (23% ↓), Esmolol for acute control (9-min half-life), Atenolol is renal (adjust in CKD), Metoprolol is first-line (beta-1 selective 75%).
Clinical Applications in Arrhythmias
- Atrial fibrillation/flutter rate control
- Target: Resting HR <80 bpm, exercise HR <110 bpm (RACE II trial)
- First-line agents: Metoprolol 25-100 mg BID, atenolol 25-100 mg daily
- Acute IV control: Metoprolol 2.5-5 mg IV q5min or esmolol infusion
- Combination: With digoxin for additive effect (different mechanisms)
- Supraventricular tachycardia (SVT)
- AVNRT/AVRT: Terminates 30-50% (inferior to adenosine 90%)
- Prophylaxis: Prevents recurrence in 50-70% of patients
- Dose: Metoprolol 25-50 mg BID or propranolol 10-40 mg TID
- Ventricular arrhythmias
- Catecholamine-induced VT: Propranolol first-line (nonselective)
- Long QT syndrome: ↓ adrenergic-triggered events by 60-70%
- Post-MI PVCs: Suppress ectopy, ↓ sudden cardiac death 23%
⭐ Clinical Pearl: Beta-blockers are the only antiarrhythmic class with mortality benefit in heart failure (MERIT-HF: 34% mortality reduction) and post-MI (23% reduction). Mechanism: Prevents catecholamine-induced arrhythmias + ↓ myocardial O₂ demand + reverse remodeling. Start low, go slow (metoprolol 12.5 mg BID, uptitrate q2 weeks).
Adverse Effects and Contraindications
- Cardiovascular
- Bradycardia: 10-15% (HR <50 bpm), dose-dependent
- Hypotension: 5-10% (SBP <90 mmHg), more with carvedilol (alpha-blockade)
- AV block: Worsens existing conduction disease, avoid in 2nd/3rd degree block
- Heart failure exacerbation: 5-10% during initiation (transient, improves with time)
- Respiratory
- Bronchospasm: 15-20% in asthmatics with nonselective agents
- Selectivity lost at high doses: Metoprolol >200 mg/day blocks beta-2
- Relative contraindication: Severe asthma/COPD (use cardioselective cautiously)
- Metabolic
- Masks hypoglycemia symptoms: Blunts tachycardia, tremor (↑ risk in diabetics on insulin)
- Glucose awareness preserved: Sweating (cholinergic) still occurs
- Lipid effects: ↑ TG by 10-20%, ↓ HDL by 5-10% (nonselective > selective)
- CNS (lipophilic agents cross BBB)
- Fatigue: 10-20%, depression: 5-10%, vivid dreams/nightmares: 5%
- Sexual dysfunction: 5-10% (erectile dysfunction, ↓ libido)
⚠️ Warning: Never abruptly discontinue beta-blockers-rebound sympathetic surge causes tachycardia, hypertension, MI risk ↑ 3-fold, arrhythmias within 24-72 hours. Taper over 1-2 weeks minimum. Mechanism: Upregulation of beta-receptors (40-50% increase) during chronic blockade.
Connect beta-blocker rate control to calcium channel blockers that provide alternative nodal suppression without beta-receptor effects.
🛡️ Beta-Blockers: The Class II Protective Shield
🚪 Calcium Channel Blockers: The Class IV Nodal Gatekeepers
Non-dihydropyridine calcium channel blockers (verapamil, diltiazem) slow AV nodal conduction by blocking L-type calcium channels, providing rate control and rhythm termination without beta-receptor antagonism. Dihydropyridines (amlodipine, nifedipine) lack antiarrhythmic properties due to vascular selectivity.
Mechanism of Class IV Antiarrhythmic Action
- AV nodal calcium channels (Cav1.2, Cav1.3)
- Phase 0 depolarization in SA/AV nodes depends on ICaL (not INa like ventricles)
- CCB blockade → ↓ upstroke velocity → ↓ conduction → ↑ AV nodal refractoriness
- Net effect: Prolongs PR interval by 20-40%, slows ventricular rate in AF by 25-35%
- Negative inotropy
- Blocks myocyte L-type channels → ↓ Ca²⁺ influx → ↓ contractility by 15-25%
- Verapamil > diltiazem for negative inotropy (verapamil 25% ↓, diltiazem 15% ↓)
- Contraindication: Severe LV dysfunction (EF <35%), cardiogenic shock
- Vascular effects
- Peripheral vasodilation → ↓ afterload → ↓ BP by 10-20 mmHg
- Coronary vasodilation → ↑ coronary flow (antianginal effect)
- Dihydropyridines: Vascular-selective (minimal cardiac effects, no antiarrhythmic use)
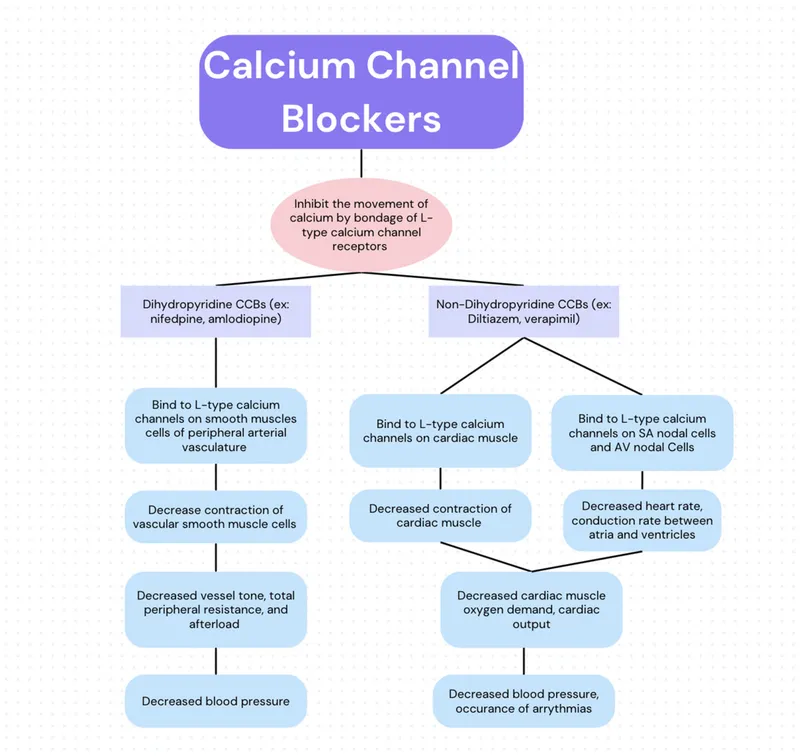
Verapamil vs Diltiazem: Clinical Distinctions
| Property | Verapamil | Diltiazem |
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more
Have doubts about this lesson?
Ask Rezzy, your AI Study Mate, to explain anything you didn't understand
Everything you need for USMLE prep
Get full Oncourse access with lessons, practice questions, flashcards and AI study tools.
Scan to download app



















