Free Radical Generation - The Radical Starters
- Radiation: Ionizing radiation (UV, X-rays) splits water ($H_2O$) into hydroxyl radicals ($'''•OH'''$).
- Inflammation: NADPH oxidase in neutrophils generates superoxide ($O_2^{•-}$) during respiratory burst to kill microbes.
- Metabolism: Normal mitochondrial respiration (OxPhos) can leak electrons to $O_2$, creating superoxide.
- Drugs & Chemicals: Metabolism of toxins (e.g., $CCl_4$) or drugs by the P450 system.
- Transition Metals: Iron ($Fe^{2+}$) and copper ($Cu^{2+}$) donate electrons in the Fenton reaction.
⭐ The Fenton Reaction is a key source of the highly reactive hydroxyl radical ($'''•OH'''). It involves the donation of an electron from ferrous iron ($Fe^{2+}$) to hydrogen peroxide ($H_2O_2$).
Mechanisms of Damage - Cellular Wrecking Crew
Free radicals target key cellular macromolecules, leading to widespread damage. The hydroxyl radical ($\cdot OH$) is the most reactive and destructive.
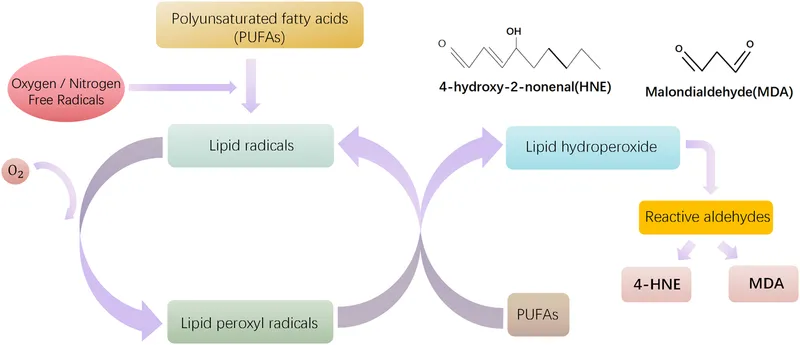
- Lipid Peroxidation:
- Attacks double bonds in polyunsaturated fatty acids of cell & organelle membranes.
- Initiates a self-propagating chain reaction, causing loss of membrane integrity.
- Protein Oxidation & Cross-Linking:
- Oxidizes amino acid side chains (e.g., sulfhydryl groups), leading to protein cross-linking.
- Results in enzyme inactivation and degradation of structural proteins.
- DNA Damage:
- Causes single and double-strand breaks, base modifications, and DNA-protein cross-links.
- Implicated in cellular aging and malignant transformation.
⭐ The Fenton reaction ($Fe^{2+} + H_2O_2 \rightarrow Fe^{3+} + \cdot OH + OH^-$) is a major source of the highly damaging hydroxyl radical in the body, linking iron overload (hemochromatosis) to free radical injury.
Antioxidant Defenses - The Radical Scavengers
Cells neutralize reactive oxygen species (ROS) via enzymatic and non-enzymatic systems to prevent oxidative damage. Key endogenous and exogenous molecules act as radical scavengers.
-
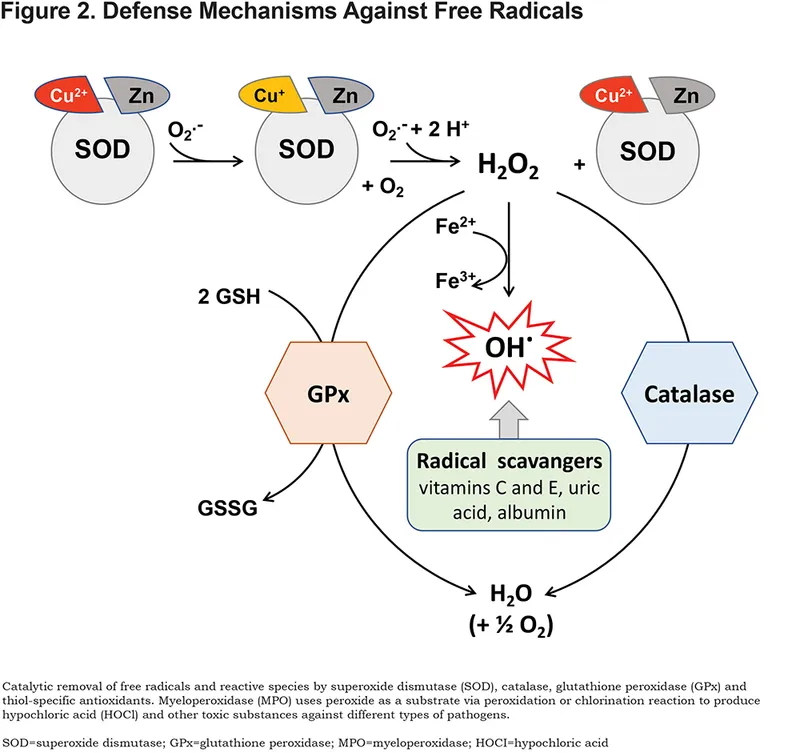
Enzymatic Scavengers
- Superoxide Dismutase (SOD): Converts superoxide into hydrogen peroxide ($2O_2^{•-} + 2H^+ \rightarrow H_2O_2 + O_2$).
- Exists as Mn-SOD (mitochondria) and Cu-Zn SOD (cytosol).
- Catalase: Found in peroxisomes; degrades hydrogen peroxide ($2H_2O_2 \rightarrow 2H_2O + O_2$).
- Glutathione (GSH) Peroxidase: Cytosolic & mitochondrial enzyme; breaks down H₂O₂ and lipid peroxides ($H_2O_2 + 2GSH \rightarrow GSSG + 2H_2O$).
- Superoxide Dismutase (SOD): Converts superoxide into hydrogen peroxide ($2O_2^{•-} + 2H^+ \rightarrow H_2O_2 + O_2$).
-
Non-Enzymatic Scavengers
- Vitamins E, A, and C are potent antioxidants.
⭐ Selenium is an essential mineral cofactor for glutathione peroxidase. Deficiency impairs ROS defense, potentially leading to cardiomyopathy (Keshan disease) and skeletal muscle myopathy.
Pathological Examples - When Radicals Run Riot
- Ischemia-Reperfusion Injury: Restored O₂ to ischemic tissue (e.g., MI, stroke) paradoxically causes damage via an ROS burst.
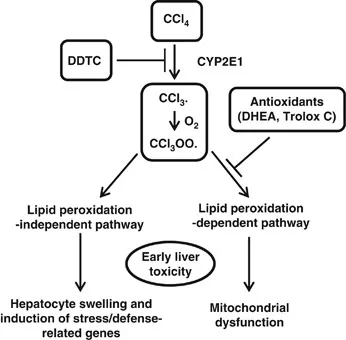
- Chemical & Toxin Injury:
- $CCl_4$ is metabolized by P450 to the $CCl_3\cdot$ radical, causing lipid peroxidation and fatty liver.
- Acetaminophen overdose (see below).
- Inflammation: Inflammatory cells (neutrophils) use NADPH oxidase to create ROS for defense, causing collateral damage.
- Radiation: Ionizing radiation splits water into highly reactive $\cdot OH$ radicals.
- Metal Overload: Iron (hemochromatosis) & copper (Wilson disease) catalyze the Fenton reaction, producing $\cdot OH$.
⭐ Acetaminophen overdose depletes glutathione (GSH), allowing toxic metabolite NAPQI to cause hepatocyte injury. Antidote: N-acetylcysteine (NAC) regenerates GSH.
High‑Yield Points - ⚡ Biggest Takeaways
- Free radicals (O₂⁻, •OH) inflict damage via lipid peroxidation, protein modification, and DNA breaks.
- Key pathologic sources include reperfusion injury, inflammation, and metabolism of drugs like acetaminophen.
- The Fenton reaction uses iron to generate the highly reactive hydroxyl radical (•OH).
- Carbon tetrachloride (CCl₄) causes fatty liver change via conversion to the •CCl₃ radical.
- Cells are protected by enzymes (SOD, catalase, glutathione peroxidase) and antioxidant vitamins (A, C, E).
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

