Respiratory Microbiome - The Breathing Buddies
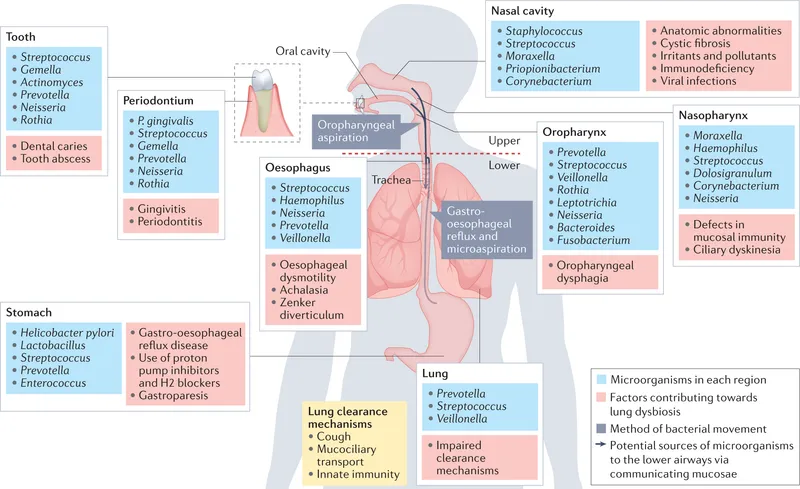
- Upper Respiratory Tract (URT): Dense, diverse community.
- Nose/Pharynx: Staphylococcus, Streptococcus, Corynebacterium, Neisseria.
- Lower Respiratory Tract (LRT): Previously considered sterile. Now known to have low bacterial biomass, primarily from URT microaspiration.
- Common genera: Prevotella, Veillonella, Streptococcus.
- Function: Crucial for immune system education and colonization resistance against pathogens.
- Dysbiosis: Imbalance linked to asthma, COPD exacerbations, and ↑ pneumonia risk.
⭐ The trio of Haemophilus influenzae, Streptococcus pneumoniae, and Moraxella catarrhalis are common URT colonizers that can become pathogenic, causing otitis media, sinusitis, and pneumonia, especially post-viral infection.
Tract Topography - Upstairs vs Downstairs
-
Upper Respiratory Tract (URT): Nasopharynx, Oropharynx
- Densely populated, similar to the oral cavity.
- Common colonizers: Staphylococcus epidermidis, Viridans streptococci, Haemophilus influenzae, Neisseria spp., and Corynebacterium spp. (diphtheroids).
- Acts as a reservoir for potential pathogens.
-
Lower Respiratory Tract (LRT): Trachea, Bronchi, Lungs
- Traditionally considered sterile; now known to have a low-biomass microbiome.
- Populated by transient microaspiration from the URT.
- Predominant genera: Prevotella, Veillonella, and Streptococcus.
⭐ The sterility of the LRT is actively maintained by the mucociliary escalator, which traps and expels microbes, and by alveolar macrophages that clear any organisms reaching the alveoli.
Clinical Correlations - Balance & Battles
- Dysbiosis: The disruption of the stable microbial community, creating a window for opportunistic pathogens (“pathobionts”) to overgrow and cause disease.
- Common Triggers: Viral infections (e.g., Influenza), smoking, broad-spectrum antibiotics, and immunosuppression.
- Key Pathobionts:
- Streptococcus pneumoniae
- Haemophilus influenzae
- Moraxella catarrhalis
- 📌 Mnemonic: SHiM
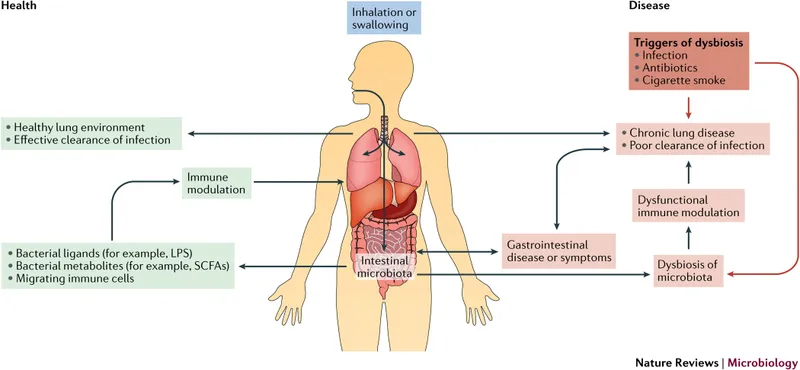
⭐ Viral URIs, particularly influenza, damage the respiratory epithelium and impair ciliary function. This paves the way for secondary bacterial pneumonia, most frequently caused by S. pneumoniae.
- The upper respiratory tract (URT) is heavily colonized; the lower respiratory tract (LRT) is kept sterile by the mucociliary escalator.
- Common URT flora include S. pneumoniae, H. influenzae, and N. meningitidis, which can be opportunistic pathogens.
- Staphylococcus aureus colonization in the anterior nares is a major risk factor for endogenous and nosocomial infections.
- Aspiration of this oral/nasal flora is a primary mechanism for developing pneumonia.
- Viral infections disrupt this balance, predisposing to secondary bacterial infections.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

