Pulmonology (COPD, asthma, interstitial lung disease)

On this page
🗺️ The Pulmonology Power Map: Architecting Your Clinical Foundation
Master the respiratory system through 3 core disease patterns that govern 80% of pulmonology boards. This lesson transforms COPD, asthma, and interstitial lung disease from isolated diagnoses into an integrated clinical framework. Build pattern recognition through quantitative thresholds, evidence-based algorithms, and high-yield discriminators that separate novice from expert-level respiratory medicine. Understanding these foundational concepts unlocks the logic behind every pulmonary function test, treatment escalation, and exacerbation management strategy you'll encounter.
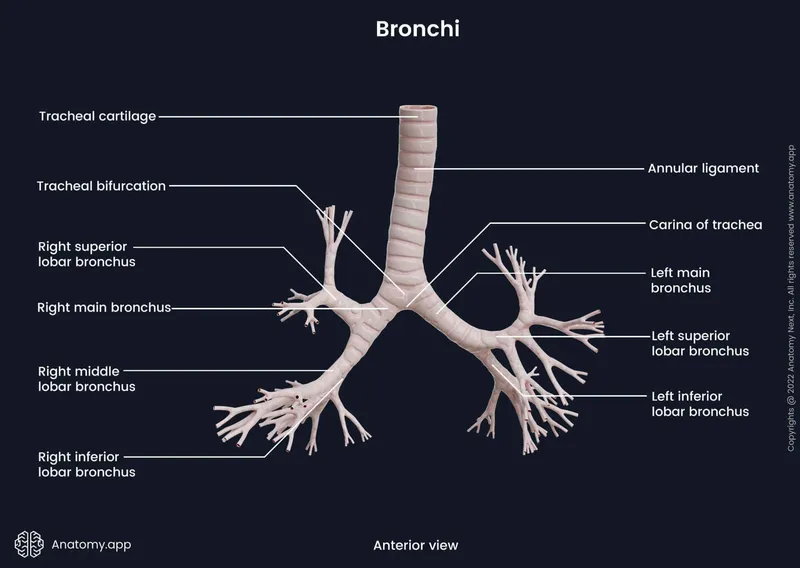
Every pulmonary diagnosis begins with recognizing one fundamental question: Is the problem with airflow (obstructive), lung expansion (restrictive), or gas exchange (diffusion)? This tripartite framework organizes the entire spectrum of respiratory pathology.
-
Obstructive Disease Architecture
- Airway-centered pathology with ↑ resistance to expiratory flow
- FEV₁/FVC ratio <0.70 defines obstruction across all guidelines
- Reversibility testing: ≥12% and ≥200 mL FEV₁ improvement = asthma component
- Bronchodilator response measured 15 minutes post-albuterol
- Irreversible obstruction suggests COPD predominance
- Mixed patterns occur in 40% of COPD patients with asthma overlap
- Air trapping creates hyperinflation: TLC >120% predicted, RV >150% predicted
-
Restrictive Disease Framework
- Parenchymal stiffness or chest wall limitation reduces lung volumes
- TLC <80% predicted with preserved FEV₁/FVC ratio ≥0.70
- DLCO discriminates: <60% predicted suggests ILD, normal suggests extrapulmonary
- Idiopathic pulmonary fibrosis: DLCO typically 40-60% at diagnosis
- Sarcoidosis: DLCO reduction parallels radiographic stage
- Hypersensitivity pneumonitis: acute phase may show <50% DLCO
-
Gas Exchange Disorders
- DLCO isolated reduction with normal spirometry and volumes
- Pulmonary vascular disease: DLCO <80% with normal lung mechanics
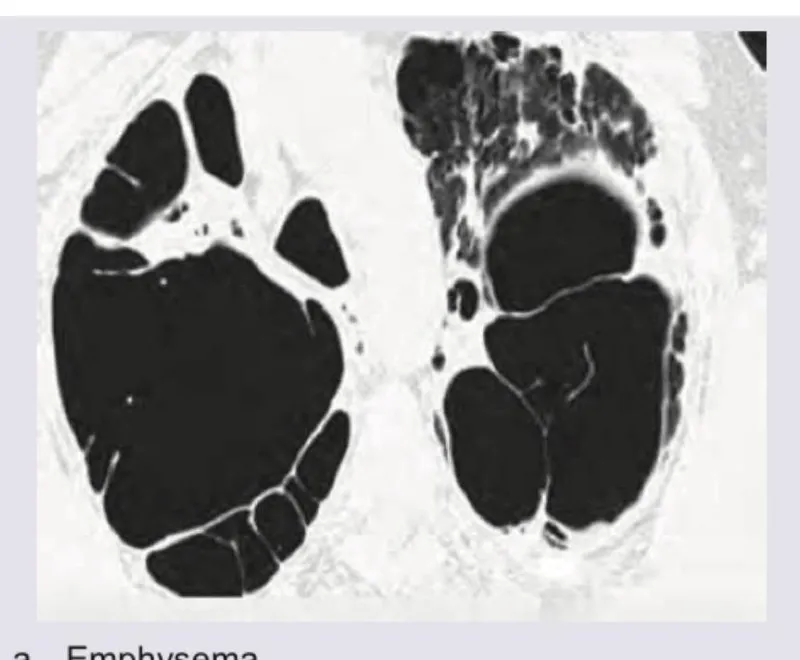
- Emphysema pattern: ↓ DLCO despite obstructive spirometry
- DLCO <40% predicted correlates with severe emphysema on CT
- DLCO/VA (KCO) distinguishes emphysema from anemia
📌 Remember: FORD for Obstructive vs Restrictive - FEV₁/FVC Obstruction Reduced, Diffusion varies
- Obstruction: FEV₁/FVC <0.70, TLC normal or ↑
- Restriction: FEV₁/FVC ≥0.70, TLC <80%
- DLCO adds the third dimension: vascular vs parenchymal vs anemia
| Parameter | Normal Value | Obstructive Pattern | Restrictive Pattern | Mixed Pattern | Clinical Significance |
|---|---|---|---|---|---|
| FEV₁/FVC | ≥0.70 | <0.70 | ≥0.70 | <0.70 | Primary discriminator |
| TLC | 80-120% | ↑ or normal (>100%) | <80% | Variable | Volume assessment |
| RV | 75-125% | ↑↑ (>150%) | ↓ or normal | ↑ if obstructive | Air trapping marker |
| DLCO | 75-125% | ↓ in emphysema (<60%) | ↓↓ in ILD (<60%) | ↓↓ | Gas exchange capacity |
| FEV₁ reversibility | <12% | ≥12% + ≥200 mL = asthma | Minimal | Partial | Bronchodilator response |
| RV/TLC ratio | <0.35 | >0.40 | Normal or ↓ | >0.40 | Air trapping quantification |
⭐ Clinical Pearl: A patient with FEV₁/FVC <0.70 and TLC <80% has mixed obstructive-restrictive disease, seen in 15-20% of advanced COPD with concurrent ILD or after lung resection. These patients show disproportionate dyspnea for their FEV₁ because both mechanics are compromised.
- The Inflammation Spectrum
- Asthma: Type 2 (Th2) inflammation with eosinophils, IgE, IL-4/5/13
- Eosinophilic asthma: peripheral eosinophils ≥300 cells/μL in 50-60%
- Neutrophilic asthma: 20-30% of severe cases, poor steroid response
- Blood eosinophils ≥150 cells/μL predict 70% better ICS response
- COPD: Neutrophilic inflammation with CD8+ T-cells, IL-8, TNF-α
- Emphysema: protease-antiprotease imbalance, elastin destruction
- Chronic bronchitis: mucus hypersecretion, goblet cell hyperplasia
- Eosinophilic COPD phenotype: 20-40% of exacerbations, better steroid response
- ILD: Fibrotic inflammation with TGF-β, myofibroblast proliferation
- IPF: progressive fibrosis despite minimal inflammation
- Hypersensitivity pneumonitis: granulomatous inflammation, antigen-driven
- Sarcoidosis: non-caseating granulomas, Th1/Th17 predominance
- Asthma: Type 2 (Th2) inflammation with eosinophils, IgE, IL-4/5/13
💡 Master This: The eosinophil threshold of ≥300 cells/μL in COPD identifies patients who benefit from ICS therapy with 30% reduction in exacerbations (FLAME, IMPACT trials). Below 100 cells/μL, ICS adds pneumonia risk without exacerbation benefit.
⭐ Clinical Pearl: Methacholine challenge with PC₂₀ <4 mg/mL confirms bronchial hyperreactivity in 95% of asthma cases with normal baseline spirometry. PC₂₀ 4-16 mg/mL = borderline, >16 mg/mL excludes asthma with 95% negative predictive value.
- Radiographic Pattern Recognition
- COPD: hyperinflation (flattened diaphragms <1.5 cm excursion), bullae, vascular pruning
- Emphysema distribution: centrilobular (smoking), panlobular (α₁-antitrypsin)
- Chronic bronchitis: bronchial wall thickening, "tram-track" appearance
- Asthma: often normal CXR; hyperinflation during exacerbation
- ABPA complication: central bronchiectasis, mucoid impaction
- ILD patterns guide diagnosis
- UIP pattern (IPF): peripheral, basilar, subpleural reticulation + honeycombing
- NSIP pattern: ground-glass opacity, less honeycombing than UIP
- Sarcoidosis: bilateral hilar lymphadenopathy (Stage I), upper lobe predominance
- COPD: hyperinflation (flattened diaphragms <1.5 cm excursion), bullae, vascular pruning
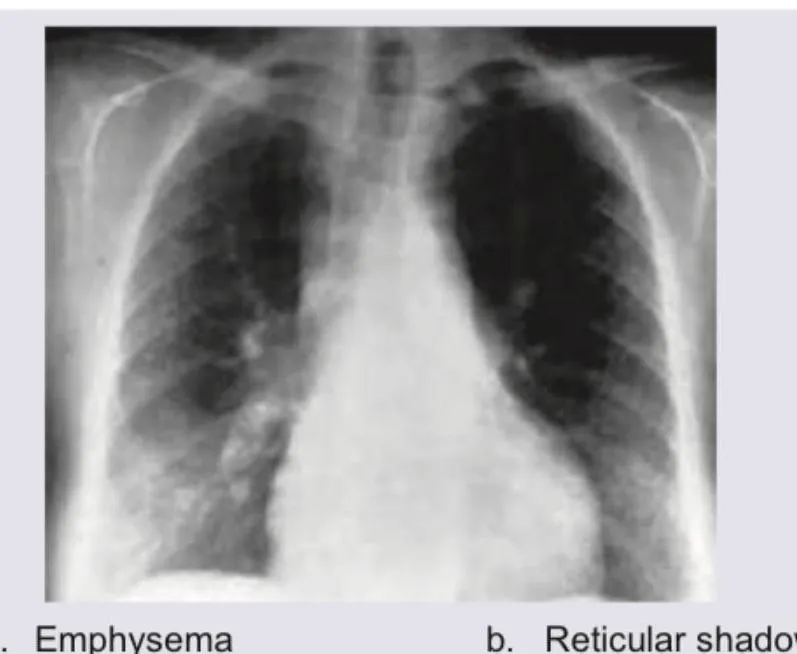
📌 Remember: RIPE for ILD patterns - Reticulation (IPF), Infiltrates (HP), Perilymphatic nodules (sarcoid), Eosinophilic (CEP)
- Reticular: fine lines = early fibrosis, honeycombing = end-stage
- Nodular: perilymphatic (sarcoid), centrilobular (HP), random (miliary)
- Ground-glass: active inflammation vs early fibrosis, needs correlation with clinical timeline
Connect these foundational patterns through to master the quantitative discrimination between obstructive and restrictive disease.
🗺️ The Pulmonology Power Map: Architecting Your Clinical Foundation
📚 The Obstructive Triad: Asthma, COPD, and Overlap Syndrome
Understanding obstructive airway disease requires mastering 3 mechanistic pillars: reversible bronchoconstriction (asthma), irreversible airflow limitation (COPD), and the 15-20% overlap syndrome (ACOS) that shares both features. Each mechanism predicts treatment response, exacerbation patterns, and long-term outcomes.
-
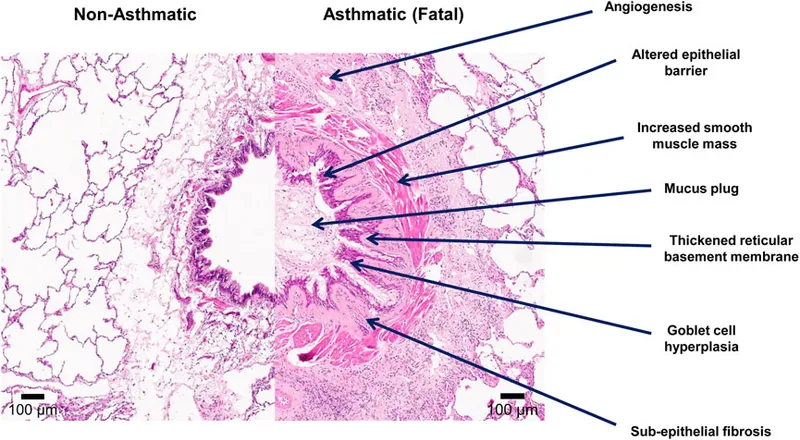
Asthma: The Reversible Airway
- Smooth muscle hypertrophy and hyperreactivity drive acute bronchoconstriction
- Bronchial smooth muscle mass ↑ by 50-300% in severe asthma
- Airway remodeling: subepithelial fibrosis, angiogenesis, mucous gland hyperplasia
- Airway wall thickness correlates with disease severity and FEV₁ decline
- Type 2 inflammation dominates in 60-70% of asthma
- Th2 cells release IL-4 (IgE production), IL-5 (eosinophil survival), IL-13 (mucus)
- Mast cell degranulation: histamine, leukotrienes (LTC₄/D₄/E₄), prostaglandins
- Eosinophils release major basic protein, eosinophil peroxidase → epithelial damage
- Non-Type 2 asthma (30-40%): neutrophilic, paucigranulocytic phenotypes
- Associated with obesity, smoking, older age at onset
- Poor response to ICS; may need macrolide or biologic therapy
- Exhaled nitric oxide (FeNO) typically <25 ppb vs >50 ppb in Type 2
- Smooth muscle hypertrophy and hyperreactivity drive acute bronchoconstriction
-
COPD: The Irreversible Obstruction
- Emphysema destroys alveolar walls through protease-antiprotease imbalance
- Neutrophil elastase, matrix metalloproteinases (MMP-9, MMP-12) degrade elastin
- Loss of elastic recoil → dynamic airway collapse during expiration
- Alveolar surface area loss: normal 70 m² reduced to <30 m² in severe disease
- Chronic bronchitis involves mucus hypersecretion and airway inflammation
- Goblet cell hyperplasia: ↑ from <5% to >20% of epithelial cells
- Mucus plugging, bacterial colonization (H. influenzae, S. pneumoniae)
- Chronic cough with sputum ≥3 months/year for ≥2 consecutive years
- Small airway disease (<2 mm diameter) contributes 80% of airflow obstruction
- Inflammatory infiltration, peribronchiolar fibrosis, luminal occlusion
- Air trapping: RV/TLC ratio >0.40 (normal <0.35)
- Small airway dysfunction precedes spirometric changes by years
- Emphysema destroys alveolar walls through protease-antiprotease imbalance
📌 Remember: ASTHMA for reversible features - Airway hyperreactivity, Smooth muscle spasm, Type 2 inflammation, Histamine release, Mucus production, Albuterol response
- Bronchodilator reversibility: ≥12% and ≥200 mL FEV₁ improvement
- Diurnal variation: >20% peak flow variability over 2 weeks
- FeNO >50 ppb suggests active eosinophilic inflammation
| Feature | Asthma | COPD | ACOS (Overlap) | Discriminating Value |
|---|---|---|---|---|
| Onset age | Usually <40 years | Usually >40 years | Variable | Age >40 favors COPD |
| Smoking history | Not required | ≥10 pack-years in 90% | ≥10 pack-years | Smoking essential for COPD |
| Symptom variability | High (diurnal, seasonal) | Progressive, persistent | Moderate | >20% PEF variability = asthma |
| Bronchodilator response | ≥12% + ≥200 mL | <12% or <200 mL | ≥12% + ≥200 mL | Reversibility favors asthma |
| Blood eosinophils | ≥300 cells/μL in 50% | <100 cells/μL in 60% | ≥300 cells/μL | ≥300 predicts ICS response |
| FeNO | >50 ppb in Type 2 | <25 ppb | Variable | >40 ppb suggests asthma |
| DLCO | Normal or ↑ | ↓↓ in emphysema (<60%) | ↓ | <60% indicates emphysema |
⭐ Clinical Pearl: Asthma-COPD Overlap Syndrome (ACOS) affects 15-20% of obstructive disease patients and carries 50% higher exacerbation rates than COPD alone. Diagnostic criteria require 3+ asthma features (eosinophils ≥300, reversibility ≥400 mL, atopy history) plus 10+ pack-year smoking history.
- Exacerbation Mechanisms Differ by Disease
- Asthma exacerbations: acute bronchospasm triggered by allergens, infections, irritants
- Viral infections (rhinovirus, RSV) cause 80% of pediatric, 50% of adult exacerbations
- Allergen exposure → IgE-mediated mast cell degranulation within minutes
- Exercise-induced: airway cooling/drying → osmotic stress → mediator release
- Peak flow drops >20% from baseline, FEV₁ <60% predicted = severe
- COPD exacerbations: bacterial infection (40-50%), viral (30%), pollution (10%)
- Bacterial: H. influenzae, S. pneumoniae, M. catarrhalis
- Increased sputum purulence (Anthonisen criteria): green/yellow = neutrophil influx
- Systemic inflammation: CRP >10 mg/L, fibrinogen ↑, leukocytosis
- Frequent exacerbators (≥2/year) have 3-fold faster FEV₁ decline
- ACOS exacerbations: mixed triggers, respond to both bronchodilators and steroids
- Higher hospitalization rates: 30-40% vs 20-25% for COPD alone
- Blood eosinophils ≥2% during exacerbation predict steroid response
- Asthma exacerbations: acute bronchospasm triggered by allergens, infections, irritants
💡 Master This: The "2% eosinophil rule" during COPD exacerbations predicts corticosteroid responsiveness. Eosinophils ≥2% (or ≥300 cells/μL) → give systemic steroids for 5 days (40 mg prednisone). Below this threshold, steroids add minimal benefit but increase hyperglycemia and infection risk.
- Phenotype-Driven Treatment Strategies
- Asthma phenotypes guide biologic therapy selection
- Allergic asthma: IgE-mediated, atopy, elevated IgE → omalizumab (anti-IgE)
- Eosinophilic asthma: eosinophils ≥300, FeNO >50 → mepolizumab (anti-IL-5)
- Type 2-high: IL-4/13 pathway → dupilumab (anti-IL-4Rα)
- Severe refractory: oral steroids, frequent exacerbations → biologic escalation
- COPD phenotypes determine inhaler combinations
- Emphysema-predominant: LABA + LAMA (bronchodilation), avoid ICS if eosinophils <100
- Chronic bronchitis: add roflumilast (PDE4 inhibitor) if FEV₁ <50% + frequent exacerbations
- Frequent exacerbator (≥2/year): LABA + LAMA + ICS triple therapy
- Eosinophilic COPD: eosinophils ≥300 → add ICS, reduces exacerbations 30%
- ACOS management: requires both controller therapies
- Start with ICS + LABA (asthma component), add LAMA (COPD component)
- Monitor eosinophils: guide ICS dosing and biologic consideration
- Smoking cessation critical: 50% reduction in FEV₁ decline rate if quit
- Asthma phenotypes guide biologic therapy selection
⭐ Clinical Pearl: α₁-Antitrypsin deficiency causes 1-3% of COPD cases, presenting with early-onset emphysema (<45 years), basilar-predominant disease, and minimal smoking history. Screen with serum AAT level <100 mg/dL (normal 150-350 mg/dL); confirm with phenotype (PiZZ, PiSZ). Augmentation therapy (weekly IV AAT) slows FEV₁ decline in FEV₁ 35-60% predicted.
Connect obstructive disease mechanisms through and to build comprehensive pattern recognition.
📚 The Obstructive Triad: Asthma, COPD, and Overlap Syndrome
🔬 The Restrictive Spectrum: Interstitial Lung Disease Decoded
Restrictive lung disease encompasses >200 distinct entities, but mastering 4 core patterns enables accurate diagnosis in 85% of cases: idiopathic pulmonary fibrosis (IPF), hypersensitivity pneumonitis (HP), sarcoidosis, and connective tissue disease-associated ILD (CTD-ILD). Each pattern has unique radiographic signatures, DLCO profiles, and treatment responses.
-
Idiopathic Pulmonary Fibrosis: The Progressive Fibroser
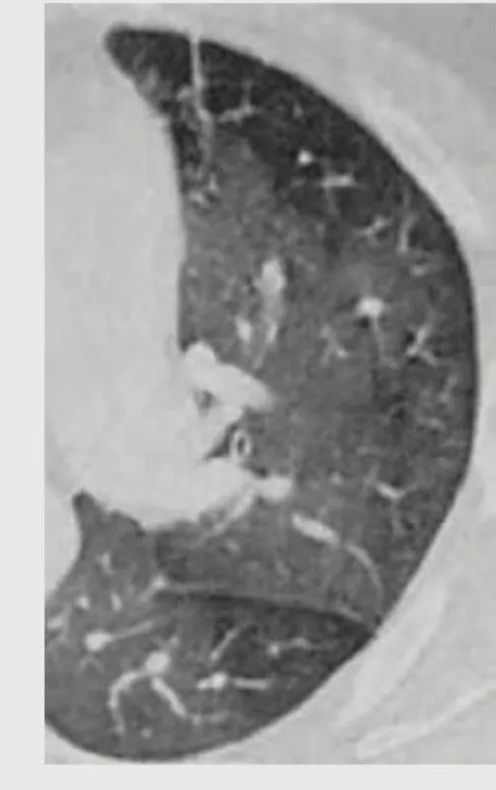
- Usual interstitial pneumonia (UIP) pattern defines IPF radiographically and histologically
- HRCT: peripheral, basilar, subpleural reticulation with honeycombing
- Honeycombing = clustered cystic airspaces 3-10 mm, thick walls, subpleural
- Traction bronchiectasis: dilated airways pulled open by surrounding fibrosis
- Absence of ground-glass opacity, nodules, consolidation (rules out other ILD)
- Progressive fibrosis despite minimal active inflammation
- Median survival 3-5 years from diagnosis without antifibrotic therapy
- FVC decline >10% or DLCO decline >15% annually = disease progression
- Acute exacerbation: sudden worsening, new ground-glass, mortality >50%
- Antifibrotic therapy slows but doesn't reverse fibrosis
- Nintedanib or pirfenidone: 50% reduction in FVC decline rate
- Start when FVC >50% predicted for maximal benefit
- Side effects: nintedanib (diarrhea 60%), pirfenidone (photosensitivity, GI upset)
- Usual interstitial pneumonia (UIP) pattern defines IPF radiographically and histologically
-
Hypersensitivity Pneumonitis: The Antigen-Driven Infiltrator
- Antigen exposure triggers granulomatous inflammation
- Acute HP: 4-6 hours post-exposure, fever, dyspnea, crackles, ground-glass on CT
- Chronic HP: insidious, progressive fibrosis, may mimic IPF if exposure remote
- Common antigens: bird proteins (pigeon breeder's lung), mold (farmer's lung), hot tubs
- Diagnostic triad: exposure history + compatible imaging + lymphocytic BAL
- BAL lymphocytosis >30% (normal <15%), CD4/CD8 ratio <1 in acute HP
- Serum precipitins (IgG antibodies) confirm exposure but not disease
- Transbronchial biopsy: poorly formed non-caseating granulomas, chronic inflammation
- Antigen avoidance is primary therapy
- Acute HP: complete resolution if exposure stopped early
- Chronic HP: fibrosis may be irreversible despite avoidance
- Corticosteroids for symptomatic acute HP: prednisone 0.5 mg/kg × 2 weeks, taper
- Antigen exposure triggers granulomatous inflammation
📌 Remember: FIBROSIS for ILD patterns - Fine reticulation (early), Interlobular septal thickening, Bronchiectasis (traction), Reticular (late), Opacity (ground-glass), Subpleural (peripheral), Irregular interfaces, Septal lines
- UIP pattern: peripheral + basilar reticulation + honeycombing, no ground-glass
- NSIP pattern: ground-glass + fine reticulation, less honeycombing than UIP
- HP pattern: centrilobular nodules + ground-glass + mosaic attenuation
| ILD Type | HRCT Pattern | DLCO | BAL Findings | Biopsy Pattern | Treatment Response |
|---|---|---|---|---|---|
| IPF (UIP) | Peripheral, basilar reticulation + honeycombing | 40-60% predicted | Normal or ↑ neutrophils | UIP: fibroblastic foci | Antifibrotics 50% slow decline |
| NSIP | Ground-glass + reticulation, less honeycombing | 50-70% predicted | Lymphocytosis 20-40% | Uniform temporal fibrosis | Steroids 60-70% response |
| HP (chronic) | Centrilobular nodules + ground-glass + fibrosis | <50% in fibrotic | Lymphocytosis >30%, CD4/CD8 <1 | Poorly formed granulomas | Avoidance + steroids variable |
| Sarcoidosis | Perilymphatic nodules, upper lobe | Variable, 60-80% | Lymphocytosis >15%, CD4/CD8 >3.5 | Well-formed non-caseating granulomas | Steroids 70-80% response |
| CTD-ILD | Variable (NSIP most common) | 50-70% predicted | Variable | NSIP or UIP pattern | Immunosuppression 50-60% |
⭐ Clinical Pearl: Honeycombing vs traction bronchiectasis distinguishes irreversible fibrosis from potentially reversible inflammation. Honeycombing = end-stage fibrosis with <10% reversibility. Traction bronchiectasis without honeycombing may improve with treatment in 30-40% of NSIP or HP cases.
- Sarcoidosis: The Granulomatous Enigma
- Non-caseating granulomas in multiple organs, 90% involve lungs
- Perilymphatic distribution: along bronchovascular bundles, interlobular septa, pleura
- Upper lobe predominance distinguishes from IPF (lower lobe)
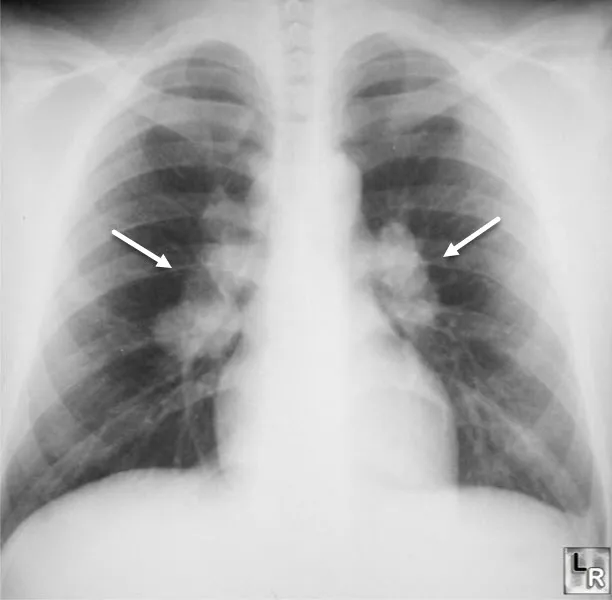
- Bilateral hilar lymphadenopathy (Stage I) in 50% at presentation
- Scadding staging predicts prognosis
- Stage 0: normal CXR (rare at diagnosis, <5%)
- Stage I: hilar lymphadenopathy alone, 60-80% spontaneous remission
- Stage II: lymphadenopathy + parenchymal infiltrates, 50-60% remission
- Stage III: parenchymal infiltrates without lymphadenopathy, 30% remission
- Stage IV: pulmonary fibrosis, <10% remission, chronic disease
- Extrapulmonary manifestations guide treatment decisions
- Cardiac sarcoid: conduction abnormalities, ventricular arrhythmias, mortality 25-50%
- Neurosarcoid: cranial neuropathies (CN VII 50%), aseptic meningitis
- Ocular: uveitis (25-30%), requires ophthalmology co-management
- Hypercalcemia: granuloma 1α-hydroxylase converts vitamin D, 10-20% of cases
- Non-caseating granulomas in multiple organs, 90% involve lungs
💡 Master This: CD4/CD8 ratio >3.5 on BAL has 94% specificity for sarcoidosis (normal 1.5-2.0). Combine with compatible imaging and clinical context to avoid biopsy in 60% of cases. However, biopsy remains gold standard to exclude malignancy and infection.
- Connective Tissue Disease-Associated ILD
- Prevalence varies by CTD type
- Systemic sclerosis: ILD in 70-80%, leading cause of death
- Rheumatoid arthritis: ILD in 10-20%, UIP pattern has worse prognosis
- Polymyositis/dermatomyositis: ILD in 40-50%, NSIP most common
- Sjögren's syndrome: ILD in 10-20%, lymphocytic interstitial pneumonia
- Screening recommendations
- Baseline HRCT and PFTs at CTD diagnosis, repeat annually if high risk
- High risk: anti-Scl-70 (systemic sclerosis), anti-Jo-1 (myositis), anti-CCP (RA)
- DLCO <70% or FVC <80% warrants HRCT even if asymptomatic
- Treatment combines immunosuppression and antifibrotics
- Mycophenolate or cyclophosphamide for progressive CTD-ILD
- Nintedanib approved for systemic sclerosis-ILD: slows FVC decline 44%
- Rituximab for refractory cases: 60-70% stabilization in systemic sclerosis
- Prevalence varies by CTD type
⭐ Clinical Pearl: Anti-synthetase syndrome (anti-Jo-1 antibody) presents with the classic triad: ILD (90%), inflammatory arthritis (70%), and myositis (60%). Additional features include mechanic's hands, Raynaud's phenomenon, and fever. ILD often precedes myositis by months to years, making antibody testing critical in cryptogenic ILD workup.
Connect ILD classification through and specific entities through , , and for comprehensive pattern mastery.
🔬 The Restrictive Spectrum: Interstitial Lung Disease Decoded
🏥 Exacerbation Management: The Acute Crisis Algorithm
Acute exacerbations of obstructive and restrictive lung disease represent 30-40% of respiratory hospitalizations. Mastering the severity stratification, treatment escalation, and disposition criteria transforms outcomes. Mortality ranges from <1% (mild asthma) to >20% (severe COPD with respiratory failure).

- Asthma Exacerbation Severity Stratification
- Mild-moderate: speaks in phrases, RR 20-30, HR <120, O₂ sat >90%, PEF >40%
- Albuterol 2.5-5 mg nebulized or 4-8 puffs MDI every 20 minutes × 3 doses
- Ipratropium 0.5 mg added to first 3 albuterol treatments (↑ FEV₁ 20% more)
- Systemic corticosteroids: prednisone 40-60 mg PO or methylprednisolone 60-80 mg IV
- Reassess after 1 hour: if PEF >70% and sustained 60 minutes, discharge
- Severe: speaks in words, RR >30, HR >120, O₂ sat <90%, PEF <40%
- Continuous albuterol nebulization: 10-15 mg/hour
- Ipratropium 0.5 mg every 20 minutes × 3, then every 4 hours
- Methylprednisolone 125 mg IV every 6 hours (higher dose than oral)
- Magnesium sulfate 2 g IV over 20 minutes if severe (↓ admission 30%)
- Admit if no improvement after 4-6 hours intensive treatment
- Life-threatening: altered mental status, silent chest, bradycardia, PEF <25%
- ICU admission, continuous monitoring, prepare for intubation
- Heliox (70% helium/30% O₂) may buy time: ↓ work of breathing
- Ketamine 1-2 mg/kg IV for intubation (bronchodilator properties)
- Avoid aggressive bag-valve-mask: causes breath stacking, pneumothorax risk
- Mild-moderate: speaks in phrases, RR 20-30, HR <120, O₂ sat >90%, PEF >40%
📌 Remember: ASTHMA SEVERITY mnemonic - Altered mental status, Silent chest, Tachycardia >120, Hypoxia O₂ sat <90%, Maximal accessory muscle use, Acidosis pH <7.35
- Mild: PEF >70%, talks in sentences
- Moderate: PEF 40-70%, talks in phrases
- Severe: PEF <40%, talks in words
- Life-threatening: PEF <25%, cannot speak
| Severity | PEF | O₂ Sat | Speech | Albuterol Dosing | Steroid Dose | Disposition |
|---|---|---|---|---|---|---|
| Mild | >70% | >95% | Sentences | 4-8 puffs MDI q20min × 3 | Prednisone 40 mg PO | Discharge if sustained |
| Moderate | 40-70% | 90-95% | Phrases | 2.5-5 mg neb q20min × 3 | Prednisone 60 mg PO | Observe 4-6 hours |
| Severe | <40% | <90% | Words | Continuous 10-15 mg/hr | Methylpred 125 mg IV q6h | Admit ward |
| Life-threatening | <25% | <90% | Cannot speak | Continuous + MgSO₄ 2 g IV | Methylpred 125 mg IV q6h | ICU admission |
| Near-fatal | Unmeasurable | <85% | Unresponsive | Prepare intubation | Methylpred 125 mg IV q6h | ICU + intubation |
⭐ Clinical Pearl: Magnesium sulfate 2 g IV reduces hospital admissions by 30% in severe asthma exacerbations (PEF <40% or FEV₁ <40% predicted). Mechanism: inhibits calcium-mediated smooth muscle contraction. Give over 20 minutes to avoid hypotension. No benefit in mild-moderate exacerbations.
- COPD Exacerbation Management
- Bronchodilator therapy forms the foundation
- Albuterol 2.5-5 mg + ipratropium 0.5 mg nebulized every 4-6 hours
- LABA + LAMA if patient already on maintenance therapy: continue at home doses
- Avoid excessive β-agonist: tachycardia, arrhythmias in elderly with cardiac disease
- Systemic corticosteroids for 5 days (not longer)
- Prednisone 40 mg PO daily × 5 days = methylprednisolone 125 mg IV daily
- Longer courses (>5 days) add no benefit, ↑ hyperglycemia and infection risk
- Eosinophils ≥2% or ≥300 cells/μL predict better steroid response
- Antibiotics if ≥2 Anthonisen criteria present
- Increased dyspnea + increased sputum volume + increased sputum purulence
- First-line: amoxicillin-clavulanate 875/125 mg BID × 5 days
- Alternatives: doxycycline 100 mg BID, azithromycin 500 mg daily
- Fluoroquinolone (levofloxacin) if recent antibiotics or local resistance
- Non-invasive ventilation (NIV) for acute hypercapnic respiratory failure
- Indications: pH <7.35, PaCO₂ >45 mmHg, RR >24, accessory muscle use
- BiPAP: IPAP 12-20 cmH₂O, EPAP 4-8 cmH₂O
- ↓ intubation rate by 65%, ↓ mortality by 55%, ↓ LOS by 3 days
- Contraindications: altered mental status, hemodynamic instability, facial trauma
- Bronchodilator therapy forms the foundation
💡 Master This: The "5-5-5 rule" for COPD exacerbations - prednisone 40 mg for 5 days, antibiotics for 5 days if indicated, reassess at 5 days. This evidence-based approach from REDUCE trial shows equal efficacy to longer steroid courses with fewer adverse effects.




















