Pathophysiology - Chronic Inflammation's Iron Fist
- Central Mediator: Chronic inflammation (autoimmune, infectious, malignant) ↑ key cytokines like IL-6.
- Hepcidin Surge: IL-6 triggers the liver to release hepcidin, the master iron regulator.
- Iron Sequestration: Hepcidin acts as an "iron gatekeeper":
- Blocks ferroportin channels in the gut, ↓ dietary iron absorption.
- Traps iron within macrophages of the reticuloendothelial system (RES).
- Result: Functional iron deficiency. Despite adequate body stores (↑ ferritin), iron is unavailable for erythropoiesis, leading to ↓ heme synthesis.
- Other Factors: Blunted erythropoietin (EPO) response.
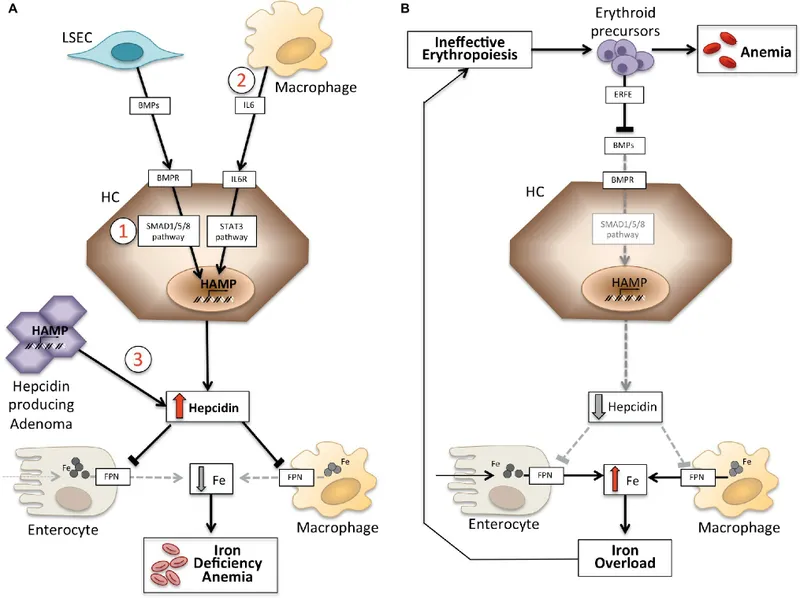
⭐ Despite low serum iron, ferritin levels are paradoxically normal or ↑ because it's an acute phase reactant and reflects trapped iron stores. This is a key diagnostic clue.
Diagnosis & Labs - Cracking the Code
- CBC/Blood Smear: Initially normocytic, normochromic anemia (MCV 80-100 fL); can become microcytic over time.
- Iron Studies Panel: The key to diagnosis.
- ↓ Serum Iron
- ↓ Total Iron-Binding Capacity (TIBC)
- ↑ or Normal Serum Ferritin (acute phase reactant)
- ↓ Transferrin Saturation (<20%)
- Advanced Markers:
- ↑ Hepcidin (confirms pathophysiology)
- Normal soluble transferrin receptor (sTfR)
⭐ Serum ferritin, an acute-phase reactant, is characteristically normal or elevated in ACD, contrasting sharply with its depletion in pure iron deficiency anemia.
Differential Diagnosis - Anemia Imposters
- Iron Deficiency Anemia (IDA): The most critical differential. Overlaps with early ACD (normocytic).
- Thalassemia: Presents with microcytic anemia but with normal or ↑ iron stores.
- Sideroblastic Anemia: Can be microcytic; characterized by ring sideroblasts in bone marrow.
⭐ The soluble transferrin receptor (sTfR) level is normal in ACD but elevated in IDA. The sTfR/log(ferritin) index is highly specific for differentiating IDA from ACD.
Management - Treating the Cause
- Primary Goal: Resolve the underlying chronic disease (e.g., infection, autoimmune condition, malignancy).
- Iron Supplementation: Only if coexistent iron deficiency is confirmed (↓ ferritin). IV iron is often preferred due to poor oral absorption from hepcidin block.
- Erythropoiesis-Stimulating Agents (ESAs): Consider for specific conditions like CKD or myelosuppressive chemotherapy if anemia is symptomatic.
⭐ ESAs (e.g., epoetin alfa) require adequate iron stores to be effective and may increase the risk of thrombosis and hypertension.
High-Yield Points - ⚡ Biggest Takeaways
- ACD is a normocytic, normochromic anemia, but can become microcytic over time.
- It is driven by inflammatory cytokines (e.g., IL-6) in chronic diseases, which stimulate ↑ hepcidin production.
- Hepcidin blocks iron release from macrophages and reduces gut absorption, leading to functional iron deficiency.
- Key lab profile: ↓ serum iron, ↓ TIBC, and normal or ↑ ferritin (an acute-phase reactant).
- Bone marrow shows adequate iron stores, unlike in true iron deficiency.
- Management focuses on treating the underlying condition.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

