2x2 Contingency Table - The Grid Game
- Organizes diagnostic test results against the true disease status (gold standard).
- Rows: Test Result (Positive / Negative)
- Columns: Disease Status (Present / Absent)
- TP (True Positive): Sick people correctly identified as sick.
- FP (False Positive): Healthy people incorrectly identified as sick.
- FN (False Negative): Sick people incorrectly identified as healthy.
- TN (True Negative): Healthy people correctly identified as healthy.
⭐ Columns represent the truth (actual disease status), while rows represent the test's conclusion.
Sensitivity & Specificity - Trusting Your Test
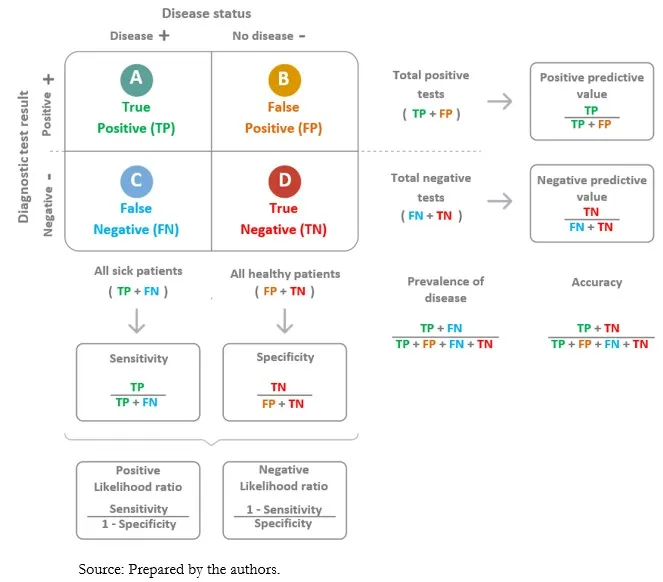
- Sensitivity: Measures a test's ability to correctly identify patients with a disease. A highly sensitive test will capture most true cases.
- Formula: $Sensitivity = TP / (TP + FN)$
- 📌 SNOUT: High Sensitivity, when Negative, rules OUT disease. Ideal for screening.
- Specificity: Measures a test's ability to correctly identify people without a disease. A highly specific test will have few false positives.
- Formula: $Specificity = TN / (TN + FP)$
- 📌 SPIN: High Specificity, when Positive, rules IN disease. Ideal for confirmation.
⭐ These are fixed, intrinsic properties of a test, calculated vertically from the 2x2 table. They do not change with disease prevalence.
PPV & NPV - Patient Prediction Power
-
Positive Predictive Value (PPV): Probability that a + test result means the patient truly has the disease.
- Calculated from rows: $PPV = \frac{TP}{TP + FP}$
- Directly varies with prevalence: ↑ prevalence → ↑ PPV.
-
Negative Predictive Value (NPV): Probability that a - test result means the patient is truly disease-free.
- Calculated from rows: $NPV = \frac{TN}{TN + FN}$
- Inversely varies with prevalence: ↑ prevalence → ↓ NPV.
⭐ Unlike sensitivity/specificity, PPV & NPV are not intrinsic to the test. They are heavily influenced by the pre-test probability (prevalence) of the disease in the specific population being tested.
Prevalence & Predictive Values - The Prevalence Effect
- Prevalence: Proportion of a population with a disease at a given time.
- While Sensitivity & Specificity are fixed test characteristics, PPV & NPV are heavily influenced by prevalence.
- The Prevalence Effect:
- Prevalence ↑ → PPV ↑, NPV ↓
- Prevalence ↓ → PPV ↓, NPV ↑
- A test has a higher PPV in a high-prevalence population vs. a low-prevalence one.
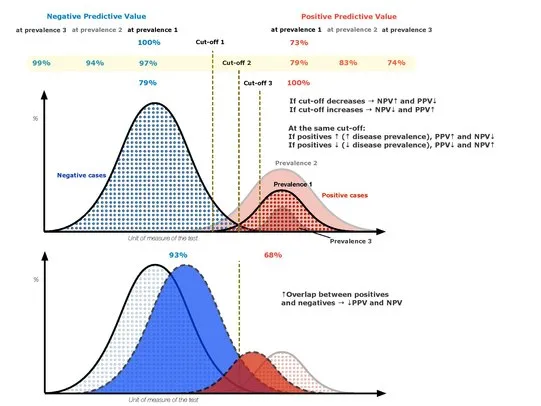
⭐ In low-prevalence (e.g., general screening) settings, positive results have a lower PPV. In high-prevalence (e.g., symptomatic patient) settings, positive results have a much higher PPV.
Likelihood Ratios - Odds & Ends
- A measure of a test's diagnostic power, combining sensitivity and specificity. They are independent of prevalence.
- Positive Likelihood Ratio (LR+): How much the odds of disease increase with a positive test.
- $LR+ = sensitivity / (1 - specificity)$
- Negative Likelihood Ratio (LR-): How much the odds of disease decrease with a negative test.
- $LR- = (1 - sensitivity) / specificity$
- Interpretation:
- LR+ > 10 is strong evidence to rule IN.
- LR- < 0.1 is strong evidence to rule OUT.
⭐ Post-test odds can be calculated from pre-test odds and the likelihood ratio: Post-test odds = Pre-test odds × LR.
High‑Yield Points - ⚡ Biggest Takeaways
- Sensitivity is the True Positive Rate (TP / [TP+FN]); a high sensitivity test, when negative, rules out disease (SNOUT).
- Specificity is the True Negative Rate (TN / [TN+FP]); a high specificity test, when positive, rules in disease (SPIN).
- Screening tests for dangerous diseases require high sensitivity to avoid missing cases.
- Confirmatory tests need high specificity to ensure a positive result is truly a positive.
- Prevalence impacts PPV and NPV but not sensitivity or specificity.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

