Clinical Suspicion - Red Flag Roundup
- Pattern: Progressive, multi-system disease in a child or young adult.
- Key Features:
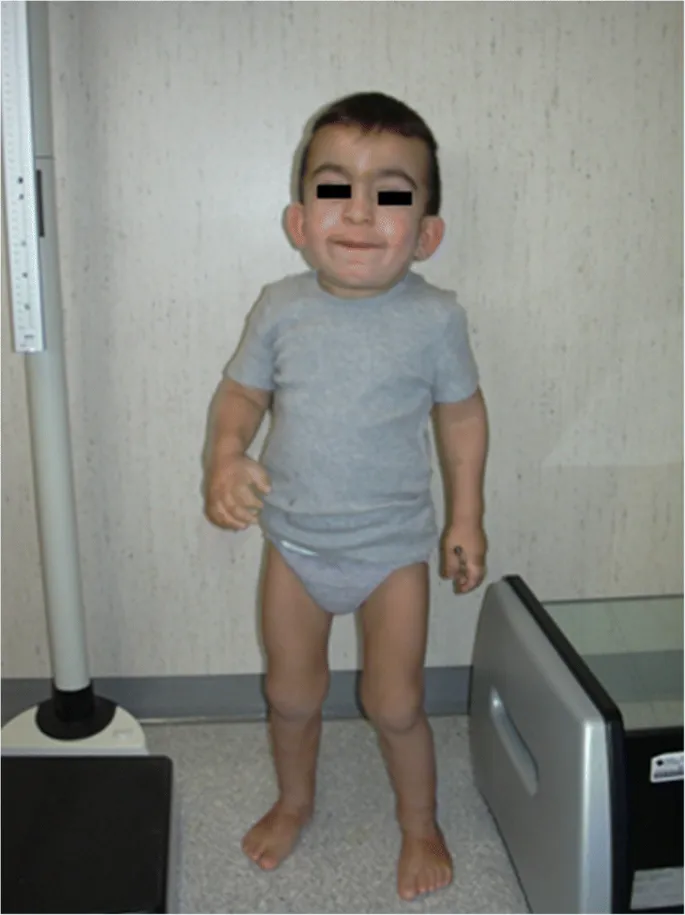
- Coarse Facial Features: Gargoylism (e.g., Hurler, Hunter syndrome).
- Hepatosplenomegaly: Massive enlargement without other clear cause.
- Neurodegeneration: Loss of developmental milestones, seizures, ataxia.
- Skeletal: Dysostosis multiplex (multiple skeletal deformities).
- Ocular: Corneal clouding, optic atrophy.
⭐ A "cherry-red spot" on the macula is a classic finding in Tay-Sachs and Niemann-Pick disease, resulting from lipid accumulation in retinal ganglion cells surrounding the fovea.
Biochemical Analysis - The Core Workup
-
Primary Screening: Measurement of accumulated substrate in urine or blood.
- Urine glycosaminoglycans (GAGs) for Mucopolysaccharidoses (MPS).
- Oligosaccharides for oligosaccharidoses.
-
Definitive Diagnosis: Enzyme activity assays are the gold standard.
- Performed on leukocytes, cultured fibroblasts, plasma, or dried blood spots.
- Confirms specific enzyme deficiency.
-
Confirmatory/Carrier Testing: Molecular analysis (gene sequencing) identifies the causative mutation.
⭐ Pseudodeficiency: Be aware of artificially low enzyme activity on an assay in an asymptomatic individual. This can be due to a benign polymorphism, not a true disease-causing mutation. It is a common pitfall in interpreting results for conditions like Tay-Sachs or Krabbe disease.
Molecular Genetics - Nailing the Diagnosis
- Confirmatory Gold Standard: Molecular genetic testing is the definitive method to confirm an LSD diagnosis after abnormal biochemical findings, directly identifying the causative mutation.
- Common Methodologies:
- Targeted Mutation Analysis: For populations with known common mutations (e.g., Gaucher disease panel).
- Single-Gene Sequencing: Sanger sequencing of the suspected gene (e.g., HEXA for Tay-Sachs).
- Next-Generation Sequencing (NGS): Multi-gene panels efficiently screen numerous LSDs, useful for ambiguous presentations.
- Key Clinical Applications:
- Family Planning: Crucial for carrier screening and prenatal diagnosis (CVS/amniocentesis).
- Prognosis: Genotype can predict disease severity (e.g., neuronopathic vs. non-neuronopathic Gaucher).
⭐ Several LSDs, including Tay-Sachs, Gaucher, and Niemann-Pick disease, have a significantly higher carrier frequency in the Ashkenazi Jewish population, making targeted screening vital.
Early Detection - Prenatal & Newborn Screening
-
Prenatal Screening
- Typically for high-risk pregnancies (e.g., positive family history, prior affected child).
- Methods include amniocentesis or chorionic villus sampling (CVS).
- Analysis focuses on measuring enzyme activity in cultured cells or direct DNA mutation analysis.
-
Newborn Screening (NBS)
- A state-mandated public health initiative for early diagnosis.
- Sample: Dried blood spot (DBS) from a heel prick.
- Method: Tandem mass spectrometry (MS/MS) is used to measure enzyme activity or accumulated substrates.
- Goal: Allows for timely intervention (e.g., enzyme replacement therapy) to prevent irreversible pathology.
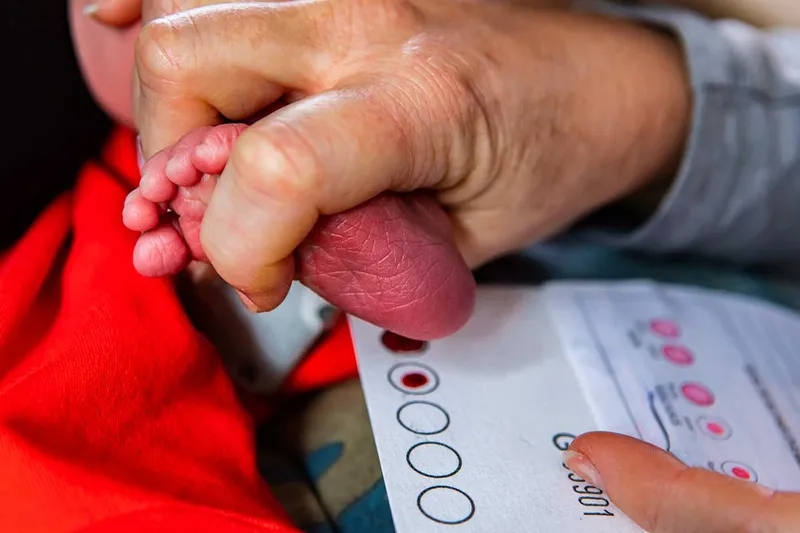
⭐ NBS panels for LSDs vary significantly by state, affecting which conditions are detected at birth.
High‑Yield Points - ⚡ Biggest Takeaways
- Enzyme activity assays in leukocytes or cultured fibroblasts are the gold standard for diagnosis.
- Genetic testing is crucial for confirming the specific mutation and for carrier screening.
- Substrate accumulation (e.g., glycosaminoglycans) can be detected in urine or plasma.
- Tissue biopsy may reveal characteristic storage material, like Zebra bodies or foamy macrophages.
- Fundoscopy can reveal a "cherry-red spot" in diseases like Tay-Sachs and Niemann-Pick type A.
- Newborn screening increasingly uses tandem mass spectrometry.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

