Substance Use Disorders

On this page
🧠 Substance Use Disorders: The Addiction Landscape
Substance use disorders represent a spectrum of brain diseases affecting 15-20% of psychiatric patients in India. Understanding the shared neurobiology across substances-from alcohol's GABA modulation to opioids' mu-receptor hijacking-transforms your ability to recognize patterns, predict complications, and deploy evidence-based interventions. Master the core principles of tolerance, dependence, and withdrawal, and you'll navigate every substance-specific presentation with confidence. This lesson builds from molecular mechanisms through clinical mastery, equipping you with frameworks that predict outcomes and guide treatment across the entire addiction spectrum.
🗺️ The Addiction Architecture: Foundations of Substance Disorders
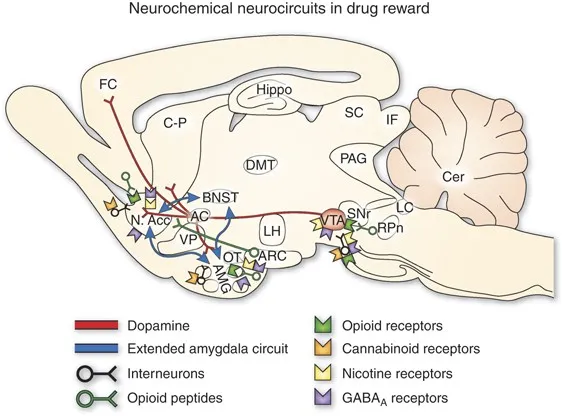
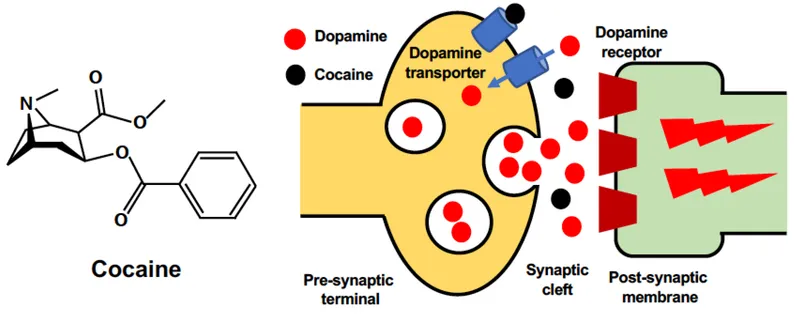
Substance use disorders share a common neurobiological blueprint despite diverse pharmacological profiles. Every addictive substance hijacks the mesolimbic dopamine pathway, creating a 200-400% surge in nucleus accumbens dopamine-far exceeding natural rewards like food (50-100% increase) or sex (100-200%). This fundamental mechanism explains why cocaine, heroin, and alcohol produce similar behavioral patterns despite acting on different primary receptors.
📌 Remember: The 3 D's of Diagnosis - Desire (craving), Difficulty controlling use, Despite harm. All substance use disorders require impaired control + social impairment + risky use + pharmacological criteria (tolerance/withdrawal) per DSM-5.
Core Diagnostic Framework
The severity spectrum operates on a continuum based on criterion count:
- Mild SUD: 2-3 criteria present
- Often missed in clinical settings
- Intervention most effective at this stage (70-80% response to brief counseling)
- No mandatory withdrawal symptoms
- Moderate SUD: 4-5 criteria present
- Structured treatment recommended
- 50-60% achieve sustained remission with comprehensive care
- Tolerance typically evident
- Severe SUD: ≥6 criteria present
- Intensive intervention required
- Only 30-40% maintain 1-year abstinence without medication-assisted treatment
- Withdrawal syndrome common
- Multiple failed quit attempts typical
⭐ Clinical Pearl: The presence of tolerance alone does not constitute a use disorder-patients on prescribed opioids for chronic pain develop tolerance but lack the compulsive use pattern. Conversely, withdrawal alone (physiological dependence) can occur with therapeutic benzodiazepine use without addiction.
Neurobiological Substrate Organization
- Primary Reward Circuit
- Ventral tegmental area (VTA) dopamine neurons
- Projects to nucleus accumbens (NAc)
- Also innervates prefrontal cortex (PFC) and amygdala
- Baseline firing rate: 3-5 Hz
- Substance-induced burst firing: 15-25 Hz
- Neurotransmitter Systems Involved
- Dopamine: reward prediction and motivation
- Glutamate: learning and memory consolidation
- GABA: inhibitory control and anxiolysis
- Serotonin: mood and impulse regulation
- Norepinephrine: arousal and stress response
- Brain Regions Showing Chronic Changes
- Prefrontal cortex: 10-15% volume reduction in chronic users
- Hippocampus: 8-12% volume loss affecting memory
- Amygdala: hyperreactivity to drug cues (300% baseline)
- Insula: interoceptive awareness and craving intensity
| Substance Class | Primary Mechanism | Peak Effect Time | Withdrawal Onset | Withdrawal Duration | Overdose Lethality |
|---|---|---|---|---|---|
| Alcohol | GABA-A agonist, NMDA antagonist | 30-90 min | 6-24 hours | 5-7 days | Moderate (respiratory depression) |
| Opioids | Mu-receptor agonist | 5-15 min (IV) | 6-12 hours (short-acting) | 7-10 days | High (respiratory arrest) |
| Stimulants | Dopamine reuptake inhibitor | 3-5 min (smoked) | 24-48 hours | 1-2 weeks | Moderate (cardiovascular) |
| Cannabis | CB1 receptor agonist | 10-30 min | 24-48 hours | 2-4 weeks | Very low |
| Benzodiazepines | GABA-A positive modulator | 15-45 min | 1-7 days (dose-dependent) | 2-8 weeks | Moderate (with alcohol) |
| Nicotine | Nicotinic acetylcholine agonist | 10-20 seconds | 2-4 hours | 2-4 weeks | Low |
💡 Master This: The speed of onset predicts addiction potential more than absolute dopamine surge. Intravenous heroin reaches the brain in 7-8 seconds, creating immediate reinforcement. Oral morphine takes 30-45 minutes, producing lower addiction rates despite similar mu-receptor activation. This principle explains why smoking crack cocaine (6-8 seconds) is more addictive than intranasal powder (3-5 minutes).
Tolerance and Dependence Mechanisms
Chronic substance exposure triggers three distinct neuroadaptive processes:
- Pharmacokinetic Tolerance (metabolic)
- Hepatic enzyme induction (CYP2E1 for alcohol)
- Develops over 2-4 weeks of regular use
- Can increase metabolism 2-3 fold
- Explains why chronic drinkers need more alcohol for effect
- Pharmacodynamic Tolerance (receptor-level)
- Receptor downregulation: 30-50% reduction in receptor density
- Receptor desensitization: decreased signal transduction efficiency
- Develops over 1-2 weeks of continuous exposure
- Accounts for most clinical tolerance
- Behavioral Tolerance (learned adaptation)
- Compensatory behaviors to maintain function
- Context-dependent (environment-specific)
- Can develop within days of repeated use
- Partially explains functional alcoholics
⭐ Clinical Pearl: Cross-tolerance between substances with similar mechanisms allows substitution therapy. Benzodiazepines show complete cross-tolerance with alcohol at GABA-A receptors, enabling safe alcohol withdrawal management. Methadone demonstrates partial cross-tolerance with heroin, providing withdrawal relief at 60-80 mg/day while blocking euphoria from illicit opioid use.
The transition from recreational use to addiction follows predictable stages, each with specific neurobiological markers. Connect these foundational concepts through the shared neurobiology explored in to understand how molecular changes drive clinical presentations.
🗺️ The Addiction Architecture: Foundations of Substance Disorders
📚 The Depressant Triad: Alcohol, Sedatives, and Opioids
Central nervous system depressants share GABA-ergic or opioidergic mechanisms that produce dose-dependent sedation, respiratory depression, and life-threatening withdrawal syndromes. Understanding their distinct pharmacological profiles while recognizing shared patterns enables precise diagnosis and treatment selection.
Alcohol's Dual Receptor Action
Ethanol uniquely modulates multiple neurotransmitter systems simultaneously, creating complex intoxication and withdrawal profiles:
- GABA-A Receptor Enhancement
- Positive allosteric modulation increases chloride influx
- Produces anxiolysis, sedation, and amnesia
- Blood alcohol 80-150 mg/dL: social disinhibition
- Blood alcohol 150-250 mg/dL: ataxia and slurred speech
- Blood alcohol >300 mg/dL: stupor and respiratory depression
- NMDA Receptor Antagonism
- Blocks glutamate-mediated excitation
- Contributes to memory blackouts (>150 mg/dL)
- Chronic blockade triggers compensatory upregulation
- Explains glutamate storm during withdrawal
- Dopamine Release
- VTA activation produces 150-200% dopamine surge
- Reinforces drinking behavior
- Genetic variations in dopamine receptors predict risk
📌 Remember: CAGE Screening - Cut down attempts, Annoyed by criticism, Guilty feelings, Eye-opener drinks. Score ≥2 has 85% sensitivity for alcohol use disorder. Add AUDIT (Alcohol Use Disorders Identification Test) score >8 for comprehensive assessment in primary care.
Sedative-Hypnotic-Anxiolytic Profile
Benzodiazepines and barbiturates produce GABA-A modulation with critical differences in safety and withdrawal severity:
- Benzodiazepine Pharmacology
- Bind alpha-gamma subunit interface
- Increase GABA binding affinity 2-3 fold
- Do not directly open chloride channels (safer than barbiturates)
- Therapeutic index: 10-20 (ratio of toxic to therapeutic dose)
- Withdrawal seizure risk: 20-30% in abrupt cessation
- Short-Acting vs Long-Acting Dynamics
- Short-acting (alprazolam, lorazepam): withdrawal in 1-2 days
- Long-acting (diazepam, clonazepam): withdrawal in 5-7 days
- Half-life predicts withdrawal timeline and severity
- Longer half-life = smoother withdrawal curve
- Barbiturate Mechanisms
- Directly activate GABA-A receptors (higher danger)
- Can open chloride channels without GABA present
- Therapeutic index: 3-5 (narrow safety margin)
- Largely replaced by benzodiazepines except phenobarbital
⭐ Clinical Pearl: The Ashton Protocol for benzodiazepine taper converts short-acting agents to diazepam equivalents, then reduces by 5-10% every 1-2 weeks. A patient on alprazolam 2 mg TID (6 mg/day) converts to diazepam 60 mg/day (1 mg alprazolam = 10 mg diazepam), then tapers over 12-16 weeks to minimize withdrawal severity and seizure risk.
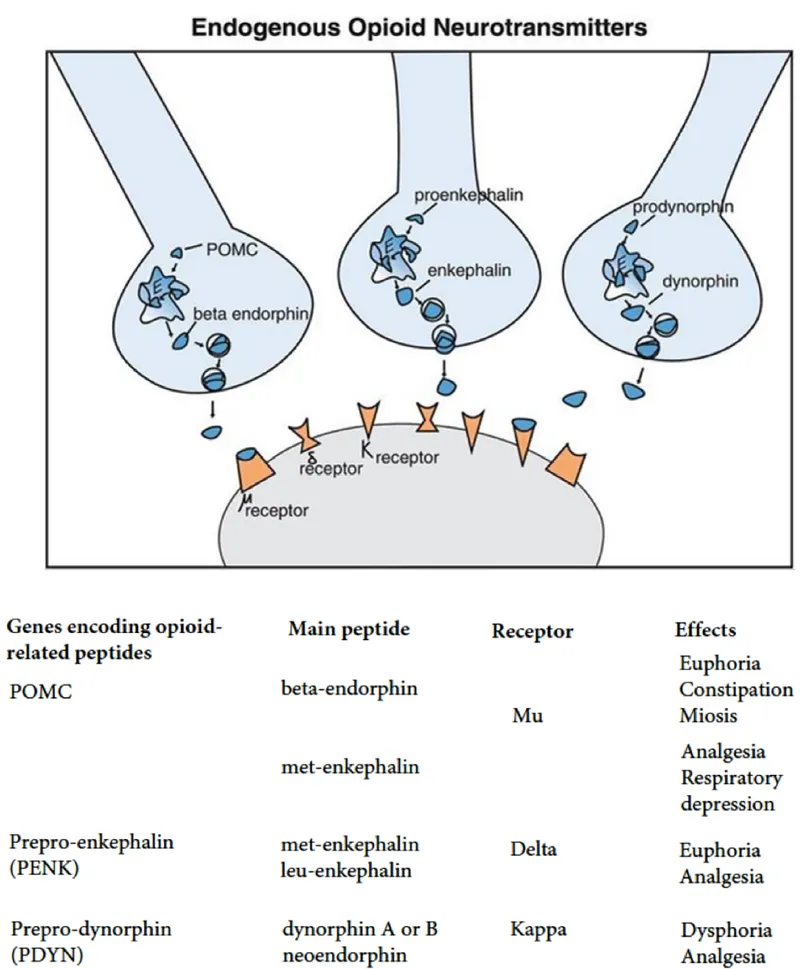
Opioid Receptor Landscape
Opioids produce analgesia, euphoria, and physical dependence through three primary receptor subtypes:
- Mu (μ) Receptor Effects
- Analgesia (supraspinal and spinal)
- Euphoria and reward (primary addiction driver)
- Respiratory depression (dose-limiting toxicity)
- Constipation (peripheral receptors)
- Miosis (pinpoint pupils, <2 mm)
- Kappa (κ) Receptor Effects
- Spinal analgesia
- Dysphoria (aversive, limits abuse potential)
- Sedation without respiratory depression
- Explains why mixed agonists (pentazocine) have lower abuse rates
- Delta (δ) Receptor Effects
- Modulates mu receptor function
- Contributes to tolerance development
- Less clinically relevant for addiction
| Opioid Type | Mu Affinity | Half-Life | Onset (IV) | Withdrawal Onset | Overdose Antidote Dose |
|---|---|---|---|---|---|
| Heroin | High | 3-5 min | 7-8 sec | 6-12 hours | Naloxone 0.4-2 mg |
| Morphine | High | 2-3 hours | 5-10 min | 8-12 hours | Naloxone 0.4-2 mg |
| Fentanyl | Very high | 30-60 min | 1-2 min | 12-24 hours | Naloxone 2-10 mg (repeat) |
| Methadone | Moderate | 24-36 hours | 30-60 min | 24-48 hours | Naloxone (caution) |
| Buprenorphine | Partial agonist | 24-42 hours | 15-30 min | 72-96 hours | Rarely needed |
| Tramadol | Low | 5-6 hours | 30-60 min | 12-24 hours | Naloxone (partial response) |
💡 Master This: Buprenorphine's partial agonist profile creates a ceiling effect for respiratory depression at doses >16-32 mg, making overdose rare. However, its high mu-receptor affinity can precipitate withdrawal if administered to patients with full agonists still present. Wait until COWS score ≥12 (moderate withdrawal) before first buprenorphine dose to avoid precipitated withdrawal.
⭐ Clinical Pearl: Fentanyl's lipophilicity (100x morphine) enables rapid CNS penetration but also creates a large volume of distribution. Overdose patients may require 10-20 mg total naloxone and prolonged monitoring (4-6 hours) because fentanyl redistributes from tissues back into circulation, causing recurrent respiratory depression after initial naloxone wears off (30-90 minutes).
The depressant substance class demonstrates how receptor-specific mechanisms produce overlapping clinical syndromes. Understanding these pharmacological nuances predicts complications and guides evidence-based management strategies detailed in , , and .
📚 The Depressant Triad: Alcohol, Sedatives, and Opioids
🔬 Stimulants and Hallucinogens: The Excitatory Spectrum
Substances that enhance monoaminergic neurotransmission produce hyperarousal, perceptual distortions, and cardiovascular complications through distinct but overlapping mechanisms. Recognizing the difference between dopaminergic stimulation and serotonergic modulation enables accurate diagnosis and appropriate crisis management.
Stimulant Pharmacodynamics
Cocaine and amphetamines share dopamine-enhancing properties but differ in mechanism and duration:
- Cocaine's Triple Reuptake Blockade
- Inhibits dopamine transporter (DAT) with Ki = 0.64 μM
- Also blocks norepinephrine and serotonin reuptake
- Produces 300-400% dopamine surge in nucleus accumbens
- Rapid metabolism: half-life 30-90 minutes
- Short high (15-30 minutes) drives binge use patterns
- Crack cocaine: onset 6-8 seconds, duration 5-10 minutes
- Amphetamine's Release Mechanism
- Reverses dopamine transporter function
- Causes vesicular release of dopamine stores
- Longer half-life: 10-12 hours (amphetamine), 12-15 hours (methamphetamine)
- Sustained high (4-8 hours) enables prolonged binges
- Methamphetamine: 70% oral bioavailability vs 30% for cocaine
- Cardiovascular Toxicity Patterns
- Hypertension: systolic >180 mmHg in 40-50% of acute intoxication
- Tachycardia: heart rate >120 bpm typical
- Myocardial infarction: 25-fold increased risk in first hour after cocaine
- Stroke: hemorrhagic and ischemic types, 13-fold increased risk
- Cardiomyopathy: chronic use causes dilated cardiomyopathy in 5-10%
📌 Remember: ACUTE Cocaine Toxicity - Arrhythmias (ventricular tachycardia), Chest pain (MI), Uncontrolled hypertension, Temperature elevation (hyperthermia), Excitation (seizures, agitation). Benzodiazepines are first-line for agitation, hypertension, and seizures-avoid beta-blockers due to unopposed alpha-stimulation risk.
Stimulant Intoxication Recognition
- Acute Presentation Features
- Pupillary dilation (>5 mm)
- Psychomotor agitation or stereotyped behaviors
- Tachycardia and hypertension
- Diaphoresis and hyperthermia (>38.5°C in severe cases)
- Paranoid ideation or hallucinations (tactile "coke bugs")
- Bruxism (teeth grinding) and jaw clenching
- Chronic Use Stigmata
- Weight loss (10-15 kg over months)
- Dental erosion ("meth mouth" in 40% of chronic users)
- Skin excoriations from picking
- Nasal septal perforation (intranasal cocaine)
- Cognitive deficits: attention, memory, executive function
- Stimulant Withdrawal Syndrome
- NOT medically dangerous (unlike alcohol/sedative withdrawal)
- Dysphoria, fatigue, hypersomnia (12-18 hours/day)
- Increased appetite and weight gain
- Psychomotor retardation
- Anhedonia lasting weeks to months
- Intense craving triggered by environmental cues
⭐ Clinical Pearl: The "crash" phase following stimulant binges involves profound dopamine depletion with 50-70% reduction in dopamine synthesis capacity. Patients sleep 12-18 hours/day for 2-5 days, then experience 2-4 weeks of anhedonia, low energy, and intense craving. This protracted withdrawal explains high relapse rates (60-70% within 3 months) without intensive psychosocial support.
Hallucinogen Serotonergic Modulation
Classic hallucinogens (LSD, psilocybin, mescaline) and dissociatives (PCP, ketamine) produce distinct perceptual changes:
- Serotonergic Hallucinogens
- 5-HT2A receptor agonism (primary mechanism)
- Perceptual distortions: visual geometric patterns, color enhancement
- Synesthesia: 20-30% of users report cross-sensory experiences
- Altered time perception and ego dissolution
- Duration: 6-12 hours (LSD), 4-6 hours (psilocybin)
- Low addiction potential: tolerance develops rapidly (24-48 hours)
- No withdrawal syndrome
- Dissociative Hallucinogens
- NMDA receptor antagonism (PCP, ketamine)
- Produces dissociation, analgesia, and amnesia
- Nystagmus (horizontal, vertical, or rotary)
- Catalepsy and violent behavior (PCP-specific)
- Ketamine: anesthetic at 1-2 mg/kg IV, dissociative at 0.5 mg/kg
- Hallucinogen Persisting Perception Disorder (HPPD)
- Recurrent perceptual disturbances after substance clearance
- Occurs in 4-5% of hallucinogen users
- Visual snow, trails, geometric patterns
- Can persist months to years
- No established treatment; benzodiazepines may help anxiety
💡 Master This: "Bad trips" represent acute anxiety reactions to hallucinogen effects, NOT overdose or toxicity. Management involves calm reassurance in quiet environment ("talk down"), benzodiazepines for severe agitation, and avoiding antipsychotics (may worsen dysphoria). Serotonergic hallucinogens have extremely high therapeutic index-no confirmed LSD overdose deaths despite doses ranging 20-1000 μg.
| Substance | Mechanism | Onset | Duration | Pupil Change | Addiction Risk | Key Danger |
|---|---|---|---|---|---|---|
| Cocaine | DAT inhibitor | 3-5 min | 15-30 min | Dilated | High | Cardiovascular |
| Methamphetamine | DA releaser | 5-10 min | 4-8 hours | Dilated | Very high | Neurotoxicity |
| LSD | 5-HT2A agonist | 30-60 min | 6-12 hours | Dilated | Low | Bad trip/HPPD |
| Psilocybin | 5-HT2A agonist | 20-40 min | 4-6 hours | Dilated | Low | Bad trip |
| PCP | NMDA antagonist | 5-15 min | 4-6 hours | Variable | Moderate | Violence/agitation |
| Ketamine | NMDA antagonist | 2-5 min | 1-2 hours | Dilated | Moderate | Dissociation |
⭐ Clinical Pearl: Cannabis intoxication can mimic stimulant or hallucinogen effects but has distinct features: conjunctival injection (red eyes), increased appetite, dry mouth, and time distortion. Unlike stimulants, cannabis produces tachycardia with orthostatic hypotension rather than hypertension. Synthetic cannabinoids ("K2", "Spice") cause more severe toxicity including seizures (5-10%) and acute kidney injury through unknown mechanisms.
Understanding the excitatory substance spectrum enables rapid pattern recognition in acute intoxication presentations. These concepts integrate with withdrawal management principles in and specific substance chapters including , , and .
🔬 Stimulants and Hallucinogens: The Excitatory Spectrum
🏥 Withdrawal Syndromes: The Neuroadaptation Rebound
Abrupt cessation of chronic substance use unmasks neuroadaptive changes, producing rebound hyperexcitability or neurotransmitter depletion. Recognizing life-threatening withdrawal (alcohol, sedatives) versus uncomfortable but safe withdrawal (opioids, stimulants) guides triage and treatment intensity decisions.
Life-Threatening Withdrawal: The GABA-Glutamate Storm
Alcohol and sedative withdrawal share mechanisms and management because both involve GABA-A receptor neuroadaptation:
- Neurobiological Substrate of Dangerous Withdrawal
- Chronic GABA enhancement causes receptor downregulation
- Chronic NMDA blockade causes receptor upregulation
- Cessation produces glutamate excitotoxicity storm
- Seizure threshold drops 40-60% below baseline
- Autonomic instability from noradrenergic hyperactivity
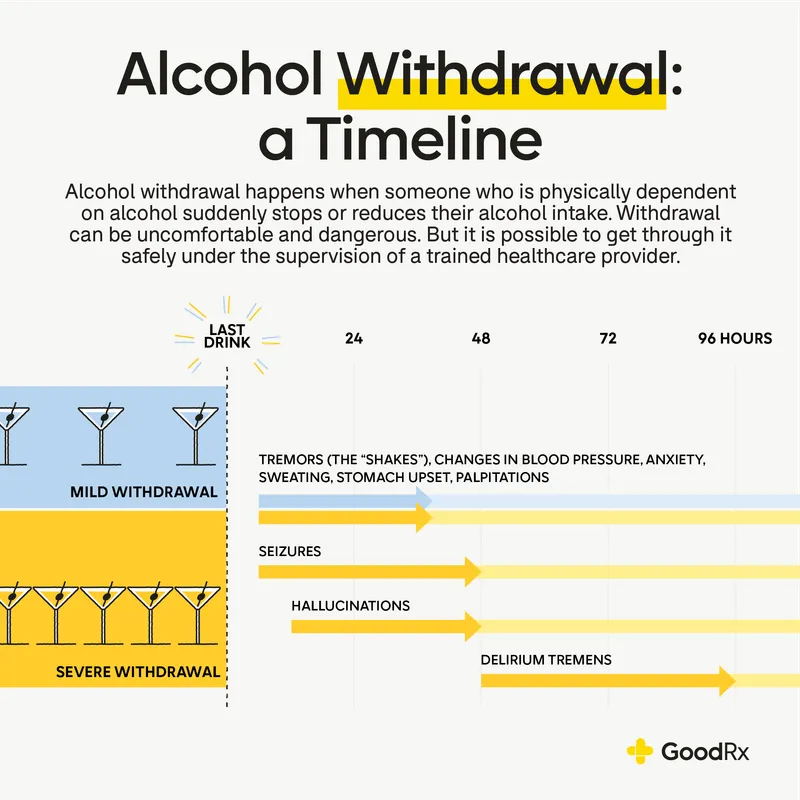
- Alcohol Withdrawal Timeline
- 6-12 hours: tremor, anxiety, tachycardia, hypertension
- 12-24 hours: hallucinations (visual, tactile, auditory)
- 24-48 hours: seizures (peak risk at 24-36 hours)
- 48-96 hours: delirium tremens (DTs) in 5% of hospitalized patients
- Mortality of untreated DTs: 15-20%
- Mortality with treatment: 1-5%
- Clinical Institute Withdrawal Assessment (CIWA-Ar) Scoring
- 10 symptom categories, maximum score 67
- Score <8: minimal withdrawal, supportive care
- Score 8-15: moderate withdrawal, scheduled benzodiazepines
- Score >15: severe withdrawal, ICU consideration
- Recheck every 1-4 hours depending on severity
📌 Remember: TREMOR for Delirium Tremens - Tremor (coarse, generalized), Restlessness and agitation, Elevated vitals (HR >100, BP >140/90, Temp >37.5°C), Mental status changes (confusion, disorientation), Onset 48-96 hours post-cessation, Risk factors (previous DTs, seizure history, concurrent illness, age >40).
Alcohol Withdrawal Management Protocol
- Benzodiazepine Selection Principles
- Long-acting preferred: diazepam, chlordiazepoxide
- Self-tapering effect reduces breakthrough symptoms
- Diazepam: 10-20 mg PO/IV every 1-2 hours until CIWA <8
- Total first-day doses: 40-200 mg depending on severity
- Lorazepam preferred if hepatic impairment (no active metabolites)
- Adjunctive Medications
- Thiamine 100 mg IV/IM daily x3 days (prevents Wernicke encephalopathy)
- Folate 1 mg daily (corrects deficiency)
- Magnesium sulfate 2 g IV if seizures or arrhythmias
- Beta-blockers (atenolol 50-100 mg daily) for refractory tachycardia
- Phenobarbital (loading dose 10 mg/kg) for benzodiazepine-refractory cases
⭐ Clinical Pearl: The phenobarbital protocol for severe alcohol withdrawal involves loading dose 15-20 mg/kg IV (typically 1000-1500 mg over 2-4 hours), then 30-60 mg q6h maintenance. Phenobarbital's long half-life (80-120 hours) provides smooth coverage and reduces benzodiazepine requirements by 50-70%. Monitor for respiratory depression (risk increased with concurrent benzodiazepines).
Opioid Withdrawal: Miserable but Not Lethal
Opioid withdrawal produces intense discomfort but lacks seizure or delirium risk (except neonates):
- Clinical Opioid Withdrawal Scale (COWS) Assessment
- 11 symptom categories, maximum score 48
- Score 5-12: mild withdrawal
- Score 13-24: moderate withdrawal
- Score 25-36: moderately severe withdrawal
- Score >36: severe withdrawal
- Guides medication dosing and treatment setting
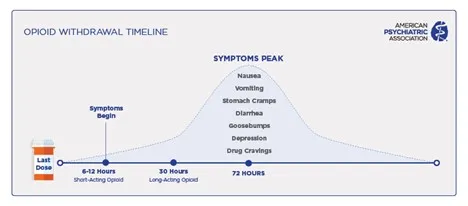
- Opioid Withdrawal Timeline
- Short-acting (heroin, oxycodone): onset 6-12 hours, peak 36-72 hours, duration 5-7 days
- Long-acting (methadone): onset 24-48 hours, peak 3-8 days, duration 2-3 weeks
- Protracted withdrawal (dysphoria, insomnia) lasts weeks to months
- Withdrawal Symptom Clusters
- Autonomic: lacrimation, rhinorrhea, diaphoresis, piloerection
- GI: nausea, vomiting, diarrhea, abdominal cramping
- Musculoskeletal: myalgias, arthralgias, restless legs
- Psychological: anxiety, dysphoria, insomnia, craving
- Vital signs: tachycardia, hypertension, fever (low-grade)
- Pupillary dilation (opposite of intoxication miosis)
💡 Master This: Clonidine (alpha-2 agonist) reduces autonomic symptoms by 60-70% but doesn't address craving or relapse risk. Dose 0.1-0.2 mg PO q4-6h (max 1.2 mg/day), monitoring for hypotension (hold if SBP <90 mmHg). Combine with symptomatic agents: loperamide for diarrhea, NSAIDs for myalgias, hydroxyzine for anxiety. This supportive approach has 80-90% completion rates but 70-80% relapse within 6 months without maintenance therapy.
| Withdrawal Type | Onset | Peak Severity | Duration | Life-Threatening? | First-Line Treatment | Mortality Risk |
|---|---|---|---|---|---|---|
| Alcohol | 6-24 hours | 24-72 hours | 5-7 days | Yes (seizures, DTs) | Benzodiazepines | 1-5% (treated) |
| Benzodiazepines | 1-7 days | 5-14 days | 2-8 weeks | Yes (seizures) | Taper + substitution | 1-2% (treated) |
| Opioids | 6-48 hours | 36-72 hours | 5-14 days | No (except neonates) | Clonidine or buprenorphine | <0.1% |
| Stimulants | 24-48 hours | 3-5 days | 1-2 weeks | No | Supportive care | <0.1% |
| Cannabis | 24-48 hours | 2-6 days | 2-4 weeks | No | Symptomatic treatment | 0% |
| Nicotine | 2-4 hours | 24-48 hours | 2-4 weeks | No | NRT or pharmacotherapy | 0% |
⭐ Clinical Pearl: Buprenorphine for opioid withdrawal provides superior outcomes compared to clonidine alone: 70-80% treatment retention vs 40-50%. Start when COWS ≥12 (moderate withdrawal) with 2-4 mg SL, titrate to 12-16 mg/day over 2-3 days. Transition to maintenance therapy (not detoxification alone) reduces 1-year relapse from 80-90% to 40-50%.
Sedative-Hypnotic Withdrawal Considerations
Benzodiazepine withdrawal mirrors alcohol withdrawal but with prolonged timeline:
- High-Risk Features
- Daily use >4 months at therapeutic doses
- Daily use >2 months at supratherapeutic doses
- History of withdrawal seizures
- Concurrent alcohol use disorder
- Short-acting agent dependence (alprazolam, lorazepam)
- Substitution Taper Strategy
- Convert to diazepam equivalent (long half-life)
- Reduce by 10% every 1-2 weeks (slow taper)
- Faster taper (25% weekly) increases seizure risk to 20-30%
- Phenobarbital alternative: 30 mg substitutes for 10 mg diazepam
- Protracted Withdrawal Syndrome
- Anxiety, insomnia, perceptual disturbances
- Can persist 6-12 months after cessation
- Affects 10-15% of long-term users
- Requires psychological support and CBT
Understanding withdrawal syndrome severity and timeline enables appropriate triage decisions and evidence-based management. These principles integrate with pharmacotherapy strategies in and specific withdrawal protocols in .
🏥 Withdrawal Syndromes: The Neuroadaptation Rebound
🔍 Treatment Frameworks: From Detoxification to Recovery
Effective substance use disorder treatment requires multi-modal approaches combining pharmacotherapy, psychosocial interventions, and long-term recovery support. Medication-assisted treatment (MAT) demonstrates superior outcomes compared to abstinence-only approaches for opioid and alcohol use disorders.
Pharmacotherapy Principles Across Substances
Medications for addiction treatment operate through three mechanisms: agonist substitution, antagonist blockade, or aversive conditioning:
- Agonist/Partial Agonist Therapy
- Reduces craving and withdrawal
- Enables functional recovery while dependent
- Examples: methadone, buprenorphine (opioids); nicotine replacement (tobacco)
- Methadone: 60-120 mg/day reduces illicit opioid use by 60-80%
- Buprenorphine: 12-16 mg/day comparable efficacy, lower overdose risk
- Treatment retention: 50-60% at 1 year (vs 10-20% abstinence-only)
- Antagonist Blockade Strategy
- Blocks euphoric effects if relapse occurs
- Requires complete detoxification first
- Examples: naltrexone (opioids, alcohol); flumazenil (benzodiazepines-rarely used)
- Naltrexone 50 mg daily or 380 mg IM monthly
- Reduces heavy drinking days by 25-30% in alcohol use disorder
- Opioid relapse prevention: 50-60% abstinence vs 20-30% placebo
- Aversive/Deterrent Medications
- Creates unpleasant reaction with substance use
- Requires high motivation and monitoring
- Examples: disulfiram (alcohol)
- Disulfiram 250-500 mg daily inhibits aldehyde dehydrogenase
- Alcohol consumption produces flushing, nausea, hypotension within 10-30 minutes
- Effectiveness depends on supervised administration
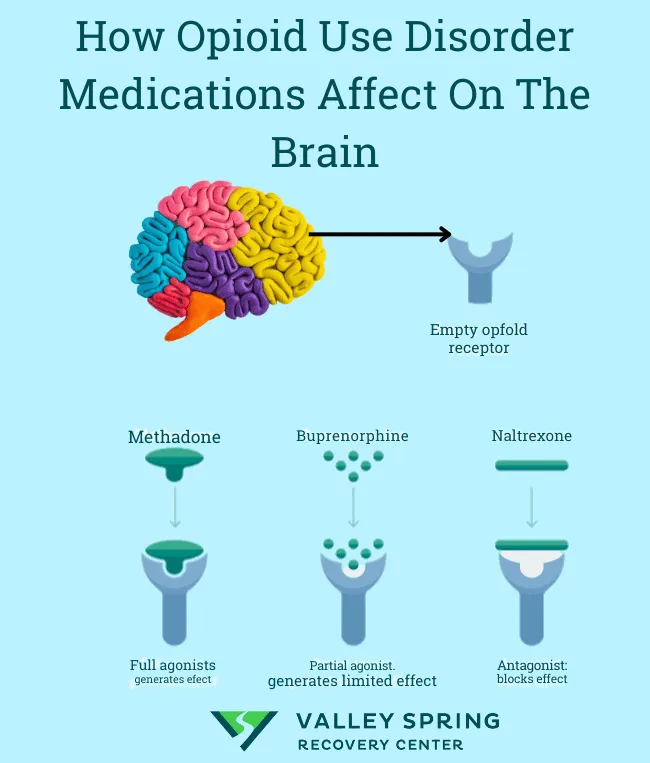
📌 Remember: MAT Success Factors - Medication adherence (supervised dosing increases compliance), Addressing co-occurring disorders (depression, anxiety), Therapy integration (CBT, contingency management). Combining pharmacotherapy with psychosocial treatment improves outcomes by 30-40% compared to medication alone.
Alcohol Use Disorder Pharmacotherapy
- FDA-Approved Medications
- Naltrexone: reduces heavy drinking days by 25-30%, craving reduction
- Acamprosate: 666 mg TID, modulates glutamate, reduces relapse by 15-20%
- Disulfiram: 250-500 mg daily, requires motivation and supervision
- Naltrexone Dosing Strategies
- Oral: 50 mg daily, start after 7-10 days abstinence
- Injectable: 380 mg IM monthly, better adherence (60% vs 40% oral)
- Hepatotoxicity risk: monitor LFTs (contraindicated if AST/ALT >3x normal)
- Opioid blockade: warn patients about pain management challenges
- Off-Label Effective Agents
- Topiramate: 200-300 mg/day, reduces heavy drinking by 30-35%
- Gabapentin: 900-1800 mg/day, reduces craving and improves sleep
- Baclofen: 30-80 mg/day, GABA-B agonist, useful in liver disease
- Ondansetron: 4-16 mg/day, particularly effective in early-onset alcoholism
⭐ Clinical Pearl: Combination therapy with naltrexone + acamprosate shows additive benefits in some studies: 50-55% abstinence at 6 months vs 35-40% with either alone. However, adherence challenges with acamprosate TID dosing often limit real-world effectiveness. Consider naltrexone + gabapentin as alternative combination with simpler dosing.
Opioid Use Disorder Medication-Assisted Treatment
- Methadone Maintenance Therapy
- Requires specialized opioid treatment program (OTP) licensure
- Initial dose: 20-30 mg daily, titrate by 5-10 mg every 3-5 days
- Target dose: 60-120 mg/day (higher doses more effective)
- Daily observed dosing initially, earn take-homes with stability
- QTc prolongation risk: baseline ECG if cardiac risk factors
- Reduces mortality by 50-70% compared to no treatment
- Buprenorphine/Naloxone Maintenance
- Office-based treatment with DATA 2000 waiver (X-waiver)
- Induction when COWS ≥12: day 1 = 4-8 mg, day 2 = 8-16 mg
- Maintenance: 12-16 mg/day (higher doses up to 24-32 mg for some)
- Naloxone component (4:1 ratio) deters IV misuse
- Monthly prescriptions after stabilization
- Ceiling effect for respiratory depression improves safety profile
- Extended-Release Naltrexone
- Requires 7-10 days opioid-free (longer for methadone: 10-14 days)
- 380 mg IM gluteal injection monthly
- Blocks opioid effects for
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more
Have doubts about this lesson?
Ask Rezzy, your AI Study Mate, to explain anything you didn't understand
Everything you need for NEET-PG prep
Get full Oncourse access with lessons, practice questions, flashcards and AI study tools.
Scan to download app





















