Psychopharmacology

On this page
🧬 The Molecular Foundation: Understanding Drug-Brain Interactions
Psychiatric medications exert therapeutic effects by modulating neurotransmitter systems, with specificity determined by receptor binding profiles, affinity constants, and downstream signaling cascades. Understanding these molecular interactions predicts both therapeutic responses and adverse effect profiles across all psychotropic drug classes.
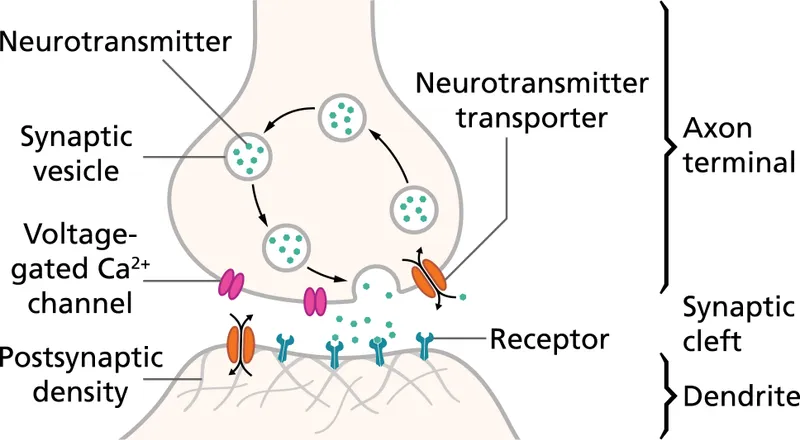
Receptor Pharmacology Fundamentals
The relationship between drug concentration and receptor occupancy follows the Hill-Langmuir equation, where 50% receptor occupancy typically occurs at the dissociation constant (Kd). Therapeutic effects generally require 60-80% receptor occupancy, while adverse effects often emerge beyond 80% occupancy thresholds.
- Receptor Binding Characteristics
- Affinity: Strength of drug-receptor interaction (measured in nanomolar concentrations)
- Efficacy: Ability to activate receptor after binding (full agonist = 100%, partial agonist = 30-70%)
- Selectivity: Preferential binding to specific receptor subtypes
- High selectivity: >100-fold difference in binding affinity
- Moderate selectivity: 10-100-fold difference
- Low selectivity: <10-fold difference (promiscuous binding)
📌 Remember: ABCDE for Receptor Dynamics - Affinity (how tight), Binding (reversible vs irreversible), Competitive (antagonist battles), Desensitization (receptor fatigue), Efficacy (activation power). Affinity determines dose requirements, while efficacy determines maximum achievable response regardless of dose escalation.
Neurotransmitter System Targets
Psychiatric medications primarily target five major neurotransmitter systems, each with distinct receptor subtypes and clinical implications:
| System | Key Receptors | Therapeutic Target | Adverse Effects | Clinical Threshold |
|---|---|---|---|---|
| Dopamine | D2 (65-70% occupancy therapeutic), D1, D3, D4 | Psychosis, motivation | EPS (>80% D2), hyperprolactinemia (>72% D2) | 60-80% D2 occupancy optimal |
| Serotonin | 5-HT1A, 5-HT2A, 5-HT2C, 5-HT3, 5-HT7 | Depression, anxiety, psychosis | Sexual dysfunction, GI upset, weight gain | 80% SERT occupancy for antidepressants |
| Norepinephrine | Alpha-1, Alpha-2, Beta-1 | Depression, ADHD | Orthostasis, tachycardia, hypertension | >50% NET occupancy therapeutic |
| GABA | GABA-A (benzodiazepine site), GABA-B | Anxiety, insomnia, seizures | Sedation, dependence, respiratory depression | 20-30% GABA-A modulation anxiolytic |
| Glutamate | NMDA, AMPA, mGluR | Depression (rapid), cognition | Dissociation, psychotomimetic effects | NMDA antagonism 40-60% for antidepressant effect |
⭐ Clinical Pearl: The 5-HT2A/D2 ratio determines atypical antipsychotic profiles. Ratios >1.5 predict lower EPS risk, with clozapine at >10, risperidone at 3-5, and haloperidol at <0.1. This ratio explains why 12 mg risperidone produces EPS similar to typical antipsychotics despite "atypical" classification.
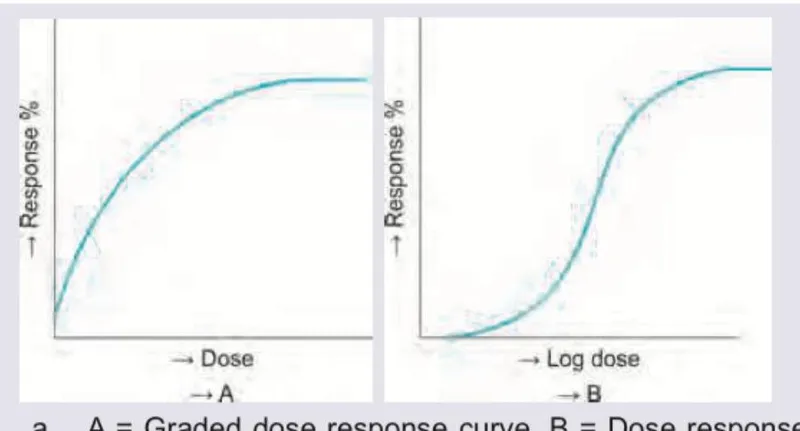
Dose-Response Relationships
The relationship between drug dose and clinical response follows sigmoidal curves, with three critical regions defining therapeutic windows and toxicity thresholds.
- Therapeutic Window Components
- Minimum Effective Concentration (MEC): Plasma level producing 50% of maximum response
- Minimum Toxic Concentration (MTC): Level producing unacceptable adverse effects in >10% of patients
- Therapeutic Index (TI): Ratio of MTC to MEC
- Wide TI (>10): SSRIs, most antipsychotics (safer)
- Narrow TI (<3): Lithium, TCAs, MAOIs (requires monitoring)
- Critical TI (<2): Clozapine for agranulocytosis (mandatory monitoring)
💡 Master This: Lithium's narrow therapeutic index (0.6-1.2 mEq/L therapeutic, >1.5 mEq/L toxic) demands monitoring because the toxic dose is only 25% higher than therapeutic dose. Contrast with sertraline, where 10-fold overdoses rarely cause serious toxicity due to wide therapeutic index (TI >50).
Receptor Adaptation Mechanisms
Chronic drug exposure triggers compensatory changes in receptor density, sensitivity, and downstream signaling pathways, explaining delayed therapeutic responses and withdrawal phenomena.
-
Downregulation Patterns
- Agonist-induced: Chronic stimulation reduces receptor density by 30-50% over 2-4 weeks
- Desensitization: Receptor uncoupling from G-proteins within minutes to hours
- Internalization: Receptor endocytosis removing 40-60% of surface receptors
- Beta-arrestin recruitment triggers receptor sequestration
- Recycling vs degradation determines recovery timeline (hours to days)
-
Upregulation Patterns
- Antagonist-induced: Chronic blockade increases receptor density by 20-40% over 1-3 weeks
- Supersensitivity: Enhanced receptor coupling efficiency
- Rebound phenomena: Explains withdrawal symptoms when discontinuing antagonists
- Antipsychotic discontinuation: Dopamine receptor upregulation causes withdrawal dyskinesia in 30-50%
- Benzodiazepine cessation: GABA-A downregulation produces rebound anxiety in 60-80%
⭐ Clinical Pearl: The 2-4 week delay in antidepressant response correlates with serotonin receptor downregulation and BDNF upregulation timelines, not immediate neurotransmitter changes. 5-HT1A autoreceptor desensitization removes negative feedback, increasing serotonergic tone by 40-60% only after 10-14 days of treatment.
The molecular precision of psychopharmacology determines every clinical outcome, from therapeutic response latency to adverse effect profiles. Understanding receptor occupancy thresholds, dose-response relationships, and adaptation mechanisms provides the foundation for rational prescribing decisions across all psychiatric conditions.
🧬 The Molecular Foundation: Understanding Drug-Brain Interactions
🔄 The ADME Journey: Pharmacokinetic Principles in Psychiatry
Pharmacokinetics governs drug movement through the body via Absorption, Distribution, Metabolism, and Excretion (ADME), determining plasma concentrations, therapeutic onset, and duration of action. Understanding these principles predicts drug interactions, guides dosing adjustments, and explains individual response variability across psychiatric medications.
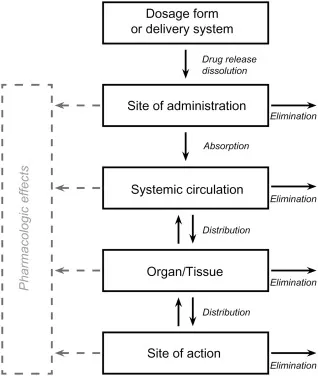
Absorption Dynamics and Bioavailability
Oral bioavailability reflects the fraction of administered drug reaching systemic circulation, influenced by first-pass metabolism, intestinal absorption, and formulation characteristics. Psychiatric medications exhibit bioavailability ranging from <10% (buspirone) to >90% (lithium).
- Absorption Variables
- First-pass metabolism: Hepatic extraction before systemic circulation
- High extraction drugs (>70%): Venlafaxine (70%), nortriptyline (50-70%)
- Low extraction drugs (<30%): Lithium (0%), gabapentin (<10%)
- Food effects: Lipophilic drugs show enhanced absorption with fatty meals
- Lurasidone: 2-fold increase with 350 kcal meal (mandatory food requirement)
- Ziprasidone: 2-fold increase with food (recommend with meals)
- Sertraline: 25% increase with food (take consistently)
- Formulation impact
- Immediate-release: Peak levels in 1-3 hours
- Extended-release: Peak levels in 4-8 hours, reduces peak-trough fluctuation by 40-60%
- Sublingual: Bypasses first-pass, 80-90% bioavailability (asenapine)
- First-pass metabolism: Hepatic extraction before systemic circulation
📌 Remember: FAST Absorption Factors - Formulation (IR vs ER), Acidity (gastric pH affects ionization), Surface area (intestinal), Transporters (P-glycoprotein efflux). P-glycoprotein pumps drugs back into gut lumen, reducing bioavailability by 20-50% for substrates like quetiapine and risperidone.
Distribution Patterns and Volume of Distribution
Volume of distribution (Vd) represents the theoretical volume needed to contain total drug amount at plasma concentration, ranging from 0.6 L/kg (confined to plasma) to >40 L/kg (extensive tissue distribution). Psychiatric drugs typically show high Vd due to lipophilicity and CNS penetration.
| Drug Class | Representative Agent | Vd (L/kg) | Protein Binding | CNS Penetration | Clinical Implication |
|---|---|---|---|---|---|
| Lithium | Lithium carbonate | 0.7-0.9 | 0% | Slow (6-12 hr equilibration) | Loading dose ineffective, steady-state in 5-7 days |
| SSRIs | Fluoxetine | 20-45 | 95% | Rapid (<2 hr) | Long half-life (4-6 days), extensive tissue stores |
| Typical Antipsychotics | Haloperidol | 18-30 | 92% | Moderate (2-4 hr) | Depot formulations viable, 3-4 week duration |
| Atypical Antipsychotics | Olanzapine | 10-15 | 93% | Rapid (<2 hr) | Moderate tissue binding, 30-hour half-life |
| Benzodiazepines | Diazepam | 1-2 | 98% | Very rapid (<30 min) | High lipophilicity, redistribution terminates single-dose effects |
⭐ Clinical Pearl: Protein binding displacement interactions rarely cause clinically significant problems because only unbound drug distributes to tissues. When warfarin (99% bound) is displaced by valproate, free warfarin transiently doubles but then redistributes, maintaining similar free concentrations. However, narrow therapeutic index drugs (lithium, phenytoin) require monitoring during displacement.
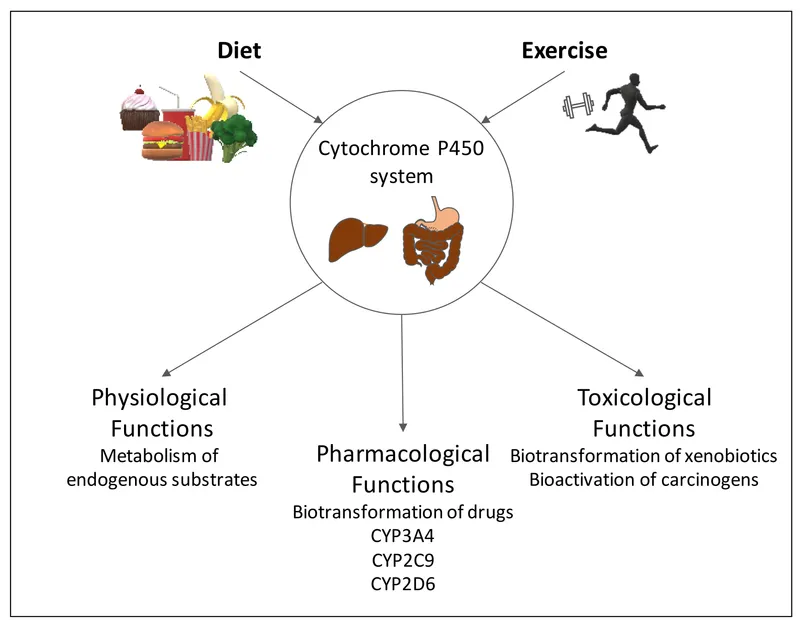
Metabolism: The Cytochrome P450 System
The CYP450 enzyme system metabolizes >80% of psychiatric medications, with genetic polymorphisms, drug interactions, and enzyme induction/inhibition creating 5-50-fold variability in drug exposure across individuals.
- Major CYP450 Isoforms in Psychiatry
- CYP2D6 (7% poor metabolizers, 5-10% ultrarapid)
- Substrates: Venlafaxine, duloxetine, risperidone, aripiprazole, TCAs
- Inhibitors: Fluoxetine (potent), paroxetine (potent), bupropion (moderate)
- Clinical impact: 5-10-fold concentration differences between phenotypes
- CYP3A4 (most abundant, 40% of drug metabolism)
- Substrates: Alprazolam, midazolam, quetiapine, lurasidone, pimavanserin
- Inhibitors: Fluvoxamine (potent), nefazodone (potent), ketoconazole (potent)
- Inducers: Carbamazepine (potent, 70-90% reduction), St. John's Wort (moderate)
- CYP1A2 (smoking induces by 40-100%)
- Substrates: Clozapine, olanzapine, duloxetine, caffeine
- Inhibitors: Fluvoxamine (potent, 5-10-fold increase)
- Inducers: Tobacco smoke (requires 50% higher clozapine doses in smokers)
- CYP2D6 (7% poor metabolizers, 5-10% ultrarapid)
💡 Master This: Fluvoxamine is the most promiscuous CYP inhibitor, affecting CYP1A2 (potent), CYP3A4 (potent), and CYP2C19 (moderate). Combining fluvoxamine with clozapine increases clozapine levels 5-10-fold, while adding to alprazolam doubles concentrations. Always check multi-enzyme inhibition profiles before combining medications.
Excretion and Half-Life Considerations
Drug elimination follows first-order kinetics for most psychiatric medications, where a constant fraction (not amount) is eliminated per unit time. Half-life determines dosing frequency and time to steady-state (4-5 half-lives).
-
Renal Excretion Patterns
- Unchanged renal excretion: Lithium (95%), gabapentin (100%), memantine (>75%)
- Dose adjustment required when CrCl <50 mL/min
- Lithium clearance parallels sodium clearance (20-40 mL/min)
- Metabolite excretion: Most antidepressants and antipsychotics
- Parent drug hepatically metabolized, then renally excreted
- Renal impairment less critical unless active metabolites accumulate
- Unchanged renal excretion: Lithium (95%), gabapentin (100%), memantine (>75%)
-
Half-Life Clinical Implications
- Short half-life (<12 hours): Venlafaxine IR (5 hours), quetiapine (6 hours)
- Requires 2-3 daily doses for stable levels
- Withdrawal symptoms within 1-2 days of discontinuation
- Extended-release formulations improve compliance
- Intermediate half-life (12-36 hours): Sertraline (26 hours), olanzapine (30 hours)
- Once-daily dosing achievable
- Steady-state in 5-7 days
- Long half-life (>48 hours): Fluoxetine (4-6 days), aripiprazole (75 hours)
- Steady-state in 4-6 weeks
- Natural taper during discontinuation (4-6 week washout)
- Missed doses less clinically significant
- Short half-life (<12 hours): Venlafaxine IR (5 hours), quetiapine (6 hours)
⭐ Clinical Pearl: Fluoxetine's active metabolite norfluoxetine has a 7-15 day half-life, creating a 4-6 week washout requirement before starting MAOIs. This protects against serotonin syndrome but also means therapeutic effects persist 2-3 weeks after discontinuation. Contrast with paroxetine (21-hour half-life), requiring only 2-week MAOI washout but causing withdrawal in 60-80% within 2-3 days.
Special Population Pharmacokinetics
Age, hepatic function, and renal function alter pharmacokinetic parameters, requiring dose adjustments to maintain therapeutic concentrations without toxicity.
-
Geriatric Changes (>65 years)
- Absorption: Decreased gastric acid, slowed motility (10-20% reduction)
- Distribution: Decreased lean mass, increased fat (Vd increases 20-40% for lipophilic drugs)
- Metabolism: 30-40% reduction in hepatic blood flow and CYP activity
- Excretion: GFR declines 1 mL/min/year after age 40
- Clinical approach: Start at 25-50% standard dose, titrate slowly
-
Hepatic Impairment
- Child-Pugh A (mild): 25% dose reduction for high-extraction drugs
- Child-Pugh B (moderate): 50% dose reduction, avoid hepatotoxic agents
- Child-Pugh C (severe): Avoid hepatically metabolized drugs, consider renal-excreted alternatives
-
Renal Impairment
- CrCl 30-50 mL/min: 50% lithium dose, monitor levels weekly initially
- CrCl <30 mL/min: Consider alternative to lithium, reduce gabapentin by 50-75%
- Dialysis: Lithium removed (30-50% per session), supplement post-dialysis
Understanding pharmacokinetic principles transforms prescribing from empiric guesswork into precision medicine, predicting drug interactions, individualizing dosing, and optimizing therapeutic outcomes while minimizing adverse effects.
🔄 The ADME Journey: Pharmacokinetic Principles in Psychiatry
💊 The Antidepressant Arsenal: Mechanism-Based Selection Strategies
Antidepressants represent the most prescribed psychiatric medication class, with 12-15% of adults receiving treatment annually. Understanding mechanism-specific therapeutic profiles, onset timelines, and adverse effect patterns enables rational selection matching patient characteristics to optimal pharmacological interventions.
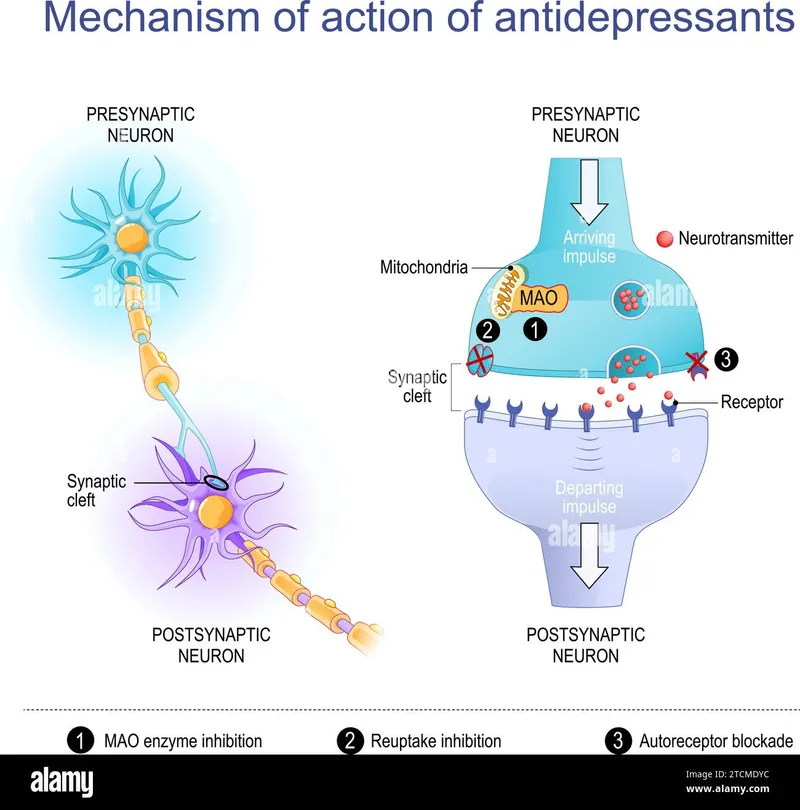
SSRI Pharmacology and Clinical Profiles
Selective Serotonin Reuptake Inhibitors (SSRIs) achieve 80-85% serotonin transporter (SERT) occupancy at therapeutic doses, increasing synaptic serotonin by 3-5-fold within hours, though clinical response requires 2-4 weeks for downstream neuroplastic changes.
- SSRI Differentiation Matrix
- Escitalopram: Highest SERT selectivity (>1000-fold over NET), minimal CYP interactions
- Dose: 10-20 mg/day (FDA maximum 20 mg, QTc concerns at higher doses)
- Half-life: 27-32 hours, steady-state in 5-7 days
- Advantages: Fewest drug interactions, well-tolerated, efficacy in 50-60% of MDD
- Sertraline: Mild dopamine reuptake inhibition (DAT affinity 10% of SERT)
- Dose: 50-200 mg/day, dose-response relationship up to 150 mg
- Half-life: 26 hours, weak CYP2D6 inhibition at >150 mg
- Advantages: Approved for 6 indications (most of any SSRI), pregnancy category with most data
- Fluoxetine: Long half-life (4-6 days), potent CYP2D6 inhibitor
- Dose: 20-80 mg/day, active metabolite norfluoxetine (7-15 days half-life)
- Advantages: Natural taper, weekly formulation available, lowest discontinuation syndrome risk
- Disadvantages: Drug interactions via CYP2D6/3A4, 4-6 week washout for MAOIs
- Escitalopram: Highest SERT selectivity (>1000-fold over NET), minimal CYP interactions
📌 Remember: SSRI Selection Mnemonic - SAFE PETS - Sertraline (most indications), Activating (fluoxetine for atypical depression), Fewer interactions (escitalopram, citalopram), Elderly (sertraline, escitalopram preferred), Pregnancy (sertraline most data), Easy taper (fluoxetine long half-life), Taper difficult (paroxetine short half-life), Sedating (paroxetine, fluvoxamine). Match patient profile to SSRI characteristics for optimal outcomes.
SNRI and TCA Mechanisms
Dual serotonin-norepinephrine reuptake inhibition provides additional efficacy in treatment-resistant depression, with 60-65% response rates compared to 50-55% for SSRIs in head-to-head trials.
| Agent | SERT:NET Ratio | Therapeutic Dose | Half-Life | Unique Features | Adverse Effect Profile |
|---|---|---|---|---|---|
| Venlafaxine | 30:1 at low dose, 10:1 at high dose | 75-225 mg/day (ER formulation) | 5 hours (IR), 11 hours (ER) | Dose-dependent NET inhibition, >150 mg for dual action | HTN in 10-15% at >225 mg, withdrawal 60% if abrupt stop |
| Duloxetine | 10:1 (balanced at all doses) | 40-120 mg/day | 12 hours | FDA-approved for pain (7 indications), avoid in hepatic impairment | Nausea 25-30% (dose-related), urinary retention 5-8% |
| Desvenlafaxine | 10:1 (balanced) | 50 mg/day (no titration needed) | 11 hours | Minimal CYP metabolism (45% unchanged renal excretion) | Similar to venlafaxine, less HTN risk |
| Amitriptyline | 1:1 (balanced), plus anticholinergic | 75-150 mg/day | 10-28 hours | Pain syndromes, migraine prophylaxis | Anticholinergic burden, orthostasis, weight gain >5 kg in 30% |
| Nortriptyline | 1:3 (NET > SERT) | 50-150 mg/day | 18-44 hours | Less anticholinergic than amitriptyline, therapeutic window 50-150 ng/mL | Orthostasis 20-30%, cardiac conduction delays |
⭐ Clinical Pearl: Venlafaxine's dose-dependent mechanism means <75 mg acts like an SSRI (SERT occupancy >80%, NET <30%), while >150 mg achieves dual inhibition (NET occupancy >50%). Patients failing SSRI trials should receive ≥150 mg venlafaxine to access noradrenergic benefits. The 10-15% hypertension risk at >225 mg requires BP monitoring at higher doses.
Atypical Antidepressants and Novel Mechanisms
Medications targeting non-monoaminergic systems or unique receptor profiles provide alternatives for patients with inadequate SSRI/SNRI responses or specific adverse effect intolerances.
-
Bupropion (Norepinephrine-Dopamine Reuptake Inhibitor)
- Mechanism: NET and DAT inhibition (ratio 1:2), no serotonergic activity
- Dose: 300-450 mg/day (XL formulation preferred)
- Advantages: No sexual dysfunction, weight neutral to loss (−2 to −3 kg), smoking cessation
- Disadvantages: Seizure risk 0.4% at ≥450 mg (contraindicated if seizure history)
- Clinical niche: SSRI-induced sexual dysfunction (add-on restores function in 60-70%)
-
Mirtazapine (Alpha-2 Antagonist)
- Mechanism: Blocks presynaptic alpha-2 autoreceptors, increasing NE and 5-HT release
- Dose: 15-45 mg/day (paradoxically more sedating at 15 mg than 45 mg)
- Advantages: Rapid sleep improvement, appetite stimulation, low nausea (<5%)
- Disadvantages: Weight gain >7% baseline in 20-30%, sedation >50% at low doses
- Clinical niche: Depression with insomnia, poor appetite, or SSRI-induced nausea
-
Vilazodone/Vortioxetine (Serotonin Modulators)
- Mechanism: SERT inhibition plus 5-HT1A partial agonism (vilazodone) or multimodal 5-HT receptor activity (vortioxetine)
- Dose: Vilazodone 20-40 mg/day, vortioxetine 10-20 mg/day
- Advantages: Potentially lower sexual dysfunction (20-30% vs 40-60% with SSRIs)
- Disadvantages: Limited long-term data, higher cost, nausea 25-30%
💡 Master This: Sexual dysfunction hierarchy across antidepressants: Paroxetine (70-80%) > sertraline (60-70%) > venlafaxine (60-65%) > escitalopram (50-60%) > duloxetine (40-50%) > vortioxetine (30-40%) > bupropion (10-15%) > mirtazapine (20-25%). When sexual side effects emerge, switch to bupropion or mirtazapine, or add bupropion 150-300 mg to existing SSRI (60-70% improvement rate).
MAOI Pharmacology and Practical Prescribing
Monoamine Oxidase Inhibitors (MAOIs) irreversibly inhibit MAO-A and MAO-B enzymes, preventing monoamine degradation and increasing synaptic availability by 5-10-fold. Despite superior efficacy in atypical depression (70-75% response), dietary restrictions and drug interactions limit use.
- MAOI Mechanisms and Selectivity
- Phenelzine (non-selective, irreversible): Inhibits MAO-A and MAO-B
- Dose: 45-90 mg/day in divided doses
- Onset: 2-4 weeks, full effect 6-8 weeks
- Tranylcypromine (non-selective, irreversible): Amphetamine-like structure, more activating
- Dose: 30-60 mg/day
- Onset: Faster than phenelzine (1-2 weeks)
- Selegiline transdermal (MAO-B selective at low dose, non-selective at high dose)
- Dose: 6-12 mg/24hr patch
- Advantage: Bypasses GI tract, no dietary restrictions at 6 mg dose
- Disadvantage: Application site reactions 30-40%
- Phenelzine (non-selective, irreversible): Inhibits MAO-A and MAO-B
⚠️ Warning: Tyramine-induced hypertensive crisis occurs when MAO-A inhibition prevents tyramine metabolism, causing norepinephrine release. Foods with >6 mg tyramine/serving (aged cheese, cured meats, draft beer, fermented foods) risk crisis. Symptoms include severe headache, BP >180/120 mmHg, chest pain, stroke risk. Emergency treatment: Nifedipine 10 mg SL or phentolamine 5 mg IV. Dietary restrictions continue 2 weeks post-discontinuation (enzyme regeneration time).
Antidepressant Selection Algorithm
- Patient-Specific Selection Factors
- Insomnia prominent: Mirtazapine 15-30 mg qhs, trazodone 50-200 mg qhs adjunct
- Fatigue/hypersomnia: Bupropion 300-450 mg, fluoxetine 20-40 mg (activating)
- Pain comorbidity: Duloxetine 60-120 mg (FDA-approved for 7 pain conditions)
- Anxiety comorbidity: Escitalopram 10-20 mg, sertraline 50-200 mg (both FDA-approved for anxiety disorders)
- Sexual dysfunction concern: Bupropion 300-450 mg, mirtazapine 15-45 mg (lowest rates)
- Weight gain concern: Bupropion (weight loss −2 to −3 kg), fluoxetine (weight neutral)
- Cardiac disease: Sertraline, citalopram (avoid TCAs, QTc prolongation risk)
⭐ Clinical Pearl: Treatment-resistant depression (failure of ≥2 adequate trials) affects 30-40% of MDD patients. Evidence-based augmentation strategies include: aripiprazole 2-15 mg (NNT 9), quetiapine XR 150-300 mg (NNT 9), lithium 300-900 mg targeting 0.6-1.0 mEq/L (NNT 5-8), or liothyronine 25-50 mcg (NNT 5-10). Combining antidepressants (e.g., SSRI + bupropion) shows 60-65% response rates in SSRI non-responders.
Understanding antidepressant pharmacology enables mechanism-based selection matching patient symptom profiles, comorbidities, and tolerability requirements to optimal therapeutic agents, maximizing response rates while minimizing adverse effects and treatment discontinuation.
💊 The Antidepressant Arsenal: Mechanism-Based Selection Strategies
🧠 The Antipsychotic Spectrum: Receptor Profiles and Clinical Selection
Antipsychotic medications treat psychotic symptoms through dopamine D2 receptor blockade, with 65-70% D2 occupancy producing antipsychotic effects and >80% occupancy causing extrapyramidal symptoms (EPS). Understanding receptor binding profiles predicts therapeutic efficacy, adverse effect profiles, and optimal patient-medication matching across first-generation (typical) and second-generation (atypical) agents.

Dopamine Receptor Occupancy and Clinical Outcomes
The relationship between D2 receptor occupancy and clinical effects follows predictable thresholds, with PET imaging studies establishing occupancy-response curves across antipsychotic doses.
-
D2 Occupancy Thresholds
- <65%: Subtherapeutic, inadequate antipsychotic effect
- 65-80%: Therapeutic window, optimal antipsychotic efficacy with minimal EPS
- >80%: EPS threshold, akathisia in 30-40%, parkinsonism in 20-30%
- >90%: High EPS risk (>60%), tardive dyskinesia risk increases with duration
- >95%: Hyperprolactinemia threshold, galactorrhea in 10-20%, amenorrhea in 30-50%
-
Typical Antipsychotic D2 Binding
- Haloperidol: 70-80% occupancy at 2-5 mg/day, >90% at >10 mg/day
- High-potency, minimal anticholinergic/antihistaminic effects
- EPS in 50-75% at doses >5 mg/day
- Prolactin elevation >100 ng/mL (normal <20 ng/mL) in >80%
- Chlorpromazine: 65-75% occupancy at 300-600 mg/day
- Low-potency, significant anticholinergic/antihistaminic/alpha-1 effects
- Sedation >70%, orthostasis 30-40%, weight gain >7% in 40-50%
- Fluphenazine decanoate: Depot formulation, 12.5-25 mg IM q2-4 weeks
- Steady D2 occupancy 70-80% with monthly dosing
- Improves adherence in 60-70% compared to oral formulations
- Haloperidol: 70-80% occupancy at 2-5 mg/day, >90% at >10 mg/day
📌 Remember: HELP for High-Potency Typicals - Haloperidol, EPS high risk, Low anticholinergic, Prolactin elevation severe. High-potency agents bind D2 selectively with minimal other receptor effects, while low-potency agents (chlorpromazine, thioridazine) have promiscuous binding causing sedation, orthostasis, and anticholinergic burden but lower EPS rates.
Atypical Antipsychotic Differentiation
Second-generation antipsychotics (SGAs) achieve lower EPS rates through 5-HT2A/D2 receptor binding ratios >1, rapid D2 dissociation, or partial D2 agonism, while introducing metabolic and sedation risks varying by agent.
| Agent | D2 Occupancy at Therapeutic Dose | 5-HT2A:D2 Ratio | Unique Mechanisms | Metabolic Risk | EPS Risk | Sedation |
|---|---|---|---|---|---|---|
| Risperidone | 70-80% at 4-6 mg | 3-5:1 | None | Moderate (20-30% weight gain >7%) | Dose-dependent (<6 mg low, >6 mg high) | Moderate |
| Olanzapine | 65-75% at 10-20 mg | >10:1 | M3 muscarinic antagonism | Highest (60-80% weight gain >7%) | Lowest (<10%) | High |
| Quetiapine | 30-50% at 300-800 mg | >10:1 | Rapid D2 dissociation, H1 antagonism | Moderate-High (30-40% weight gain) | Very low (<5%) | Highest |
| Aripiprazole | >90% (partial agonist) | 10:1 | D2/5-HT1A partial agonism | Lowest (<10% weight gain) | Low (10-15% akathisia) | Lowest |
| Clozapine | 40-60% at 300-600 mg | >10:1 | Multiple receptors, rapid D2 dissociation | Highest (80% weight gain >7%) | Lowest (<5%) | Very high |
| Lurasidone | 70-80% at 40-120 mg | >20:1 | 5-HT7 antagonism | Low (<15% weight gain) | Low (10-15%) | Low-Moderate |
⭐ Clinical Pearl: Clozapine remains the gold-standard for treatment-resistant schizophrenia, with 50-60% response rates in patients failing ≥2 adequate antipsychotic trials (vs <20% with other agents). Despite superior efficacy, agranulocytosis risk (0.8% cumulative, 80% occurs in first 18 weeks) mandates weekly CBC monitoring for 6 months, then biweekly for 6 months, then monthly. Absolute neutrophil count (ANC) thresholds: <1500 discontinue, <1000 contraindicated permanently.
Long-Acting Injectable (LAI) Antipsychotics
Depot formulations eliminate daily adherence requirements, maintaining therapeutic D2 occupancy for 2-12 weeks per injection and reducing relapse rates by 30-40% compared to oral
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more
Have doubts about this lesson?
Ask Rezzy, your AI Study Mate, to explain anything you didn't understand
Everything you need for NEET-PG prep
Get full Oncourse access with lessons, practice questions, flashcards and AI study tools.
Scan to download app

















