Respiratory System

On this page
🫁 The Respiratory Landscape: Your Clinical Foundation
Master the respiratory system's architecture, and you unlock every pattern of gas exchange, ventilation-perfusion matching, and clinical dysfunction. This lesson builds your understanding from anatomical territories through functional integration to clinical mastery. You'll develop systematic frameworks for recognizing respiratory pathology, predicting outcomes from physiological derangements, and integrating multi-system responses. Each concept connects structure to mechanism, mechanism to clinical presentation, and presentation to evidence-based management with quantitative precision.
🗺️ Pulmonary Architecture: The Gas Exchange Territory
The respiratory system operates as a 700 million alveolar interface where atmospheric air meets pulmonary capillary blood across a membrane just 0.5 micrometers thick. Understanding this architectural precision reveals why diffusion capacity, ventilation-perfusion matching, and membrane integrity determine every clinical outcome from exercise tolerance to hypoxemic respiratory failure.
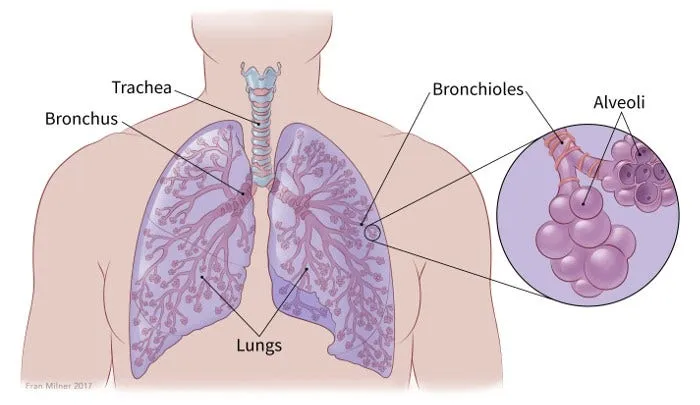
Structural Organization: The Conducting Zone
The airways divide into 23 generations from trachea to alveoli, with the first 16 generations forming the anatomical dead space where no gas exchange occurs. This 150 mL conducting zone volume becomes critical when calculating alveolar ventilation and understanding why rapid shallow breathing reduces effective gas exchange despite normal minute ventilation.
- Upper Airways (Generations 0-4)
- Trachea: 2.5 cm diameter, 11 cm length, reinforced by C-shaped cartilage
- Main bronchi: Right bronchus 25° from vertical (aspiration risk), left 45°
- Lobar bronchi: 3 right divisions, 2 left divisions
- Segmental bronchi: 10 right segments, 8-10 left segments
- Lower Airways (Generations 5-16)
- Bronchioles: <1 mm diameter, smooth muscle control (no cartilage)
- Terminal bronchioles: Final conducting airways, 0.5 mm diameter
- Total dead space: 150 mL (2.2 mL/kg ideal body weight)
- Resistance contribution: 80% from airways >2 mm (proximal dominance)
📌 Remember: "Dead Space = 2.2 × Weight" - For a 70 kg patient, anatomical dead space equals 154 mL. This explains why tidal volumes <350 mL produce minimal alveolar ventilation despite adequate minute ventilation.
Respiratory Zone: The Exchange Interface
Beyond generation 17, the respiratory zone begins with 300 million respiratory bronchioles transitioning to alveolar ducts and sacs. This creates a gas exchange surface area of 50-100 m² (tennis court size) compressed into 5-6 liters of total lung capacity.
- Alveolar Architecture
- Total alveoli: 300-700 million (varies with body size and age)
- Individual alveolus diameter: 200-300 micrometers
- Wall thickness: 0.2-0.5 micrometers (Type I pneumocytes)
- Surface area: 50-100 m² at total lung capacity
- Capillary network: 280 billion capillary segments
- Capillary diameter: 7-10 micrometers (RBC deformation required)
- Blood volume in capillaries: 70-100 mL at rest, ↑200 mL during exercise
- Transit time: 0.75 seconds at rest, ↓0.25 seconds during maximal exercise
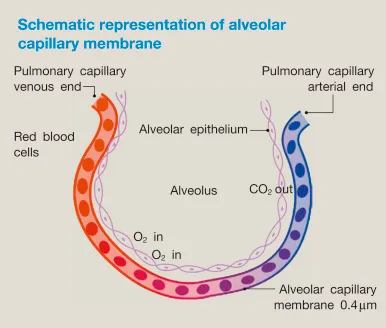
⭐ Clinical Pearl: Alveolar capillary transit time of 0.75 seconds at rest provides 3× safety margin for complete oxygen equilibration (0.25 seconds required). This reserve explains why diffusion limitation only occurs in severe interstitial disease or extreme exercise when transit time drops to <0.25 seconds.
The Blood-Gas Barrier: Membrane Components
The alveolar-capillary membrane consists of three layers totaling 0.5 micrometers thickness, creating minimal diffusion distance while maintaining structural integrity against 25 mmHg capillary pressure.
| Layer | Thickness | Key Components | Clinical Significance |
|---|---|---|---|
| Alveolar epithelium | 0.05 μm | Type I pneumocytes (95% surface area) | Damaged in ARDS, increases diffusion distance |
| Interstitium | 0.1-0.2 μm | Basement membranes, elastin, collagen | Thickened in interstitial lung disease (>0.5 μm) |
| Capillary endothelium | 0.05 μm | Continuous endothelium, tight junctions | Disrupted in pulmonary edema (permeability ↑) |
| Type II pneumocytes | Variable | Surfactant production, stem cell function | Deficiency causes neonatal RDS |
| Alveolar macrophages | Mobile | Immune surveillance, particle clearance | First-line defense, activated in inflammation |
💡 Master This: The 0.5 micrometer membrane thickness enables Fick's Law diffusion where gas transfer rate = (Area × Diffusion coefficient × Pressure gradient) / Thickness. Any process that ↑ thickness (edema, fibrosis) or ↓ area (emphysema) reduces diffusion capacity proportionally, manifesting as exertional hypoxemia when transit time limitations emerge.
Pulmonary Surfactant: The Compliance Guardian
Type II pneumocytes (covering 5% of alveolar surface) secrete surfactant, a phospholipid-protein complex that reduces surface tension from 70 dynes/cm to 25 dynes/cm, preventing alveolar collapse and reducing the work of breathing by 50-60%.
- Surfactant Composition
- Dipalmitoylphosphatidylcholine (DPPC): 40% (primary surface tension reducer)
- Other phospholipids: 40% (phosphatidylglycerol, phosphatidylethanolamine)
- Surfactant proteins: 10% (SP-A, SP-B, SP-C, SP-D)
- SP-A and SP-D: Immune function, pathogen binding
- SP-B and SP-C: Essential for surfactant spreading and stability
- Cholesterol: 10% (membrane fluidity)
- Functional Impact
- Surface tension reduction: 70 → 25 dynes/cm at normal lung volumes
- Compliance improvement: ↑2-fold compared to saline-filled lungs
- Prevents atelectasis: Stabilizes small alveoli (Laplace's Law: P = 2T/r)
- Work of breathing: Reduces by 50-60%
📌 Remember: "Surfactant = Surface Tension Slayer" - Without surfactant, surface tension forces would require 3× greater transpulmonary pressure to inflate alveoli. Premature infants born before 32-34 weeks lack adequate surfactant, causing neonatal respiratory distress syndrome with compliance <0.5 mL/cmH₂O/kg (normal: 1-1.5 mL/cmH₂O/kg).
Understanding this architectural foundation connects directly to where these structural elements determine pressure-volume relationships, and where membrane characteristics govern diffusion capacity.
🗺️ Pulmonary Architecture: The Gas Exchange Territory
📚 Ventilation Mechanics: The Pressure-Volume Engine
Breathing operates through cyclical pressure gradients generated by respiratory muscles expanding the thoracic cage. Master these pressure relationships, and you predict every pattern of ventilatory failure, work of breathing, and mechanical ventilation response. The diaphragm generates 70-80% of tidal volume at rest, descending 1-2 cm during quiet breathing and up to 10 cm during maximal inspiration.
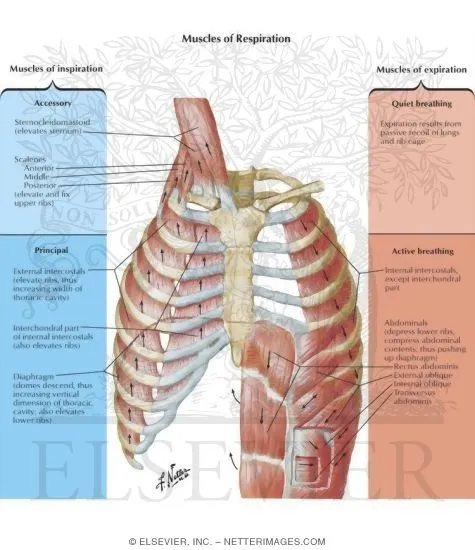
Pressure Gradients: The Driving Forces
Airflow occurs when alveolar pressure differs from atmospheric pressure, following Ohm's Law analogy: Flow = ΔPressure / Resistance. Understanding the four key pressures reveals the mechanics of every breath and explains ventilatory patterns in disease.
- Atmospheric Pressure (Patm)
- Sea level: 760 mmHg (reference point = 0 cmH₂O)
- Driving force for inspiration when alveolar pressure becomes subatmospheric
- Alveolar Pressure (Palv)
- End-expiration: 0 cmH₂O (equal to atmospheric, no flow)
- Mid-inspiration: -1 to -3 cmH₂O (subatmospheric, air flows in)
- Mid-expiration: +1 to +3 cmH₂O (supraatmospheric, air flows out)
- Forced expiration: Can reach +100 cmH₂O or higher
- Intrapleural Pressure (Pip)
- End-expiration at FRC: -5 cmH₂O (always subatmospheric in health)
- Peak inspiration: -8 to -10 cmH₂O during quiet breathing
- Maximal inspiration: Can reach -30 cmH₂O or lower
- Becomes positive during forced expiration or mechanical ventilation
- Transpulmonary Pressure (PL = Palv - Pip)
- Distending pressure across lung parenchyma
- End-expiration: +5 cmH₂O (prevents alveolar collapse)
- Peak inspiration: +8 to +10 cmH₂O during quiet breathing
- Determines lung volume via compliance relationship: V = PL × Compliance
⭐ Clinical Pearl: Intrapleural pressure remains -5 cmH₂O at end-expiration due to opposing elastic recoil forces: lung parenchyma pulling inward (+5 cmH₂O) and chest wall pulling outward (-5 cmH₂O). Pneumothorax eliminates this subatmospheric pressure, causing lung collapse to minimal volume (~10% of normal) as elastic recoil goes unopposed.
The Respiratory Cycle: Pressure-Volume Choreography
Each breath follows a predictable sequence of pressure changes driving volume changes through the compliance relationship. This cycle becomes disrupted in obstructive and restrictive diseases with characteristic patterns.
- Inspiration Phase (Active Process)
- Duration: 1-1.5 seconds during quiet breathing
- Diaphragm descent: 1-2 cm (quiet), 10 cm (maximal)
- External intercostals: Elevate ribs 2-3 cm, expand lateral diameter
- Accessory muscles (exercise/disease): Scalenes, sternocleidomastoids
- Activate when minute ventilation exceeds 40 L/min
- Recruitment indicates respiratory distress (sensitivity 85%)
- Pressure changes: Pip -5 → -8 cmH₂O, Palv 0 → -1 cmH₂O
- Volume change: 500 mL tidal volume at rest
- Expiration Phase (Passive at Rest)
- Duration: 2-3 seconds during quiet breathing (I:E ratio = 1:2)
- Elastic recoil: Lung parenchyma and chest wall return to equilibrium
- Passive pressure changes: Pip -8 → -5 cmH₂O, Palv 0 → +1 cmH₂O
- Active expiration (exercise/disease): Internal intercostals, abdominal muscles
- Abdominal contraction increases Pip to +20 to +40 cmH₂O
- Forced expiratory volume in 1 second (FEV₁) depends on active expiration
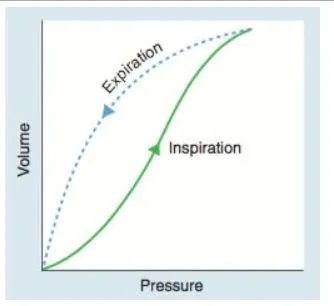
💡 Master This: The I:E ratio of 1:2 during quiet breathing provides adequate time for passive expiration via elastic recoil. In obstructive diseases (asthma, COPD), airway resistance ↑ prolongs expiration time, creating dynamic hyperinflation when expiratory time is insufficient. Patients adopt pursed-lip breathing to maintain positive airway pressure (+5 to +10 cmH₂O), preventing premature airway collapse.
Work of Breathing: Energy Economics
The respiratory muscles consume 2-3% of total body oxygen consumption at rest, increasing to 15-20% during maximal exercise. This work overcomes elastic resistance (65%) and airflow resistance (35%) during normal breathing.
- Elastic Work (Compliance-related)
- Overcomes lung elastic recoil: 70% of elastic work
- Overcomes chest wall elastic recoil: 30% of elastic work
- Increases with: ↓ Compliance (fibrosis, ARDS, pulmonary edema)
- Normal compliance: 200 mL/cmH₂O (combined lung-thorax system)
- Lung alone: 200 mL/cmH₂O
- Chest wall alone: 200 mL/cmH₂O
- Series combination: 100 mL/cmH₂O total
- Resistive Work (Flow-related)
- Airway resistance: 80% from airways >2 mm diameter
- Tissue resistance: 20% from lung and chest wall deformation
- Normal airway resistance: 0.5-2.5 cmH₂O/L/sec
- Increases with: Bronchospasm, secretions, airway edema
- Flow pattern: Laminar in small airways (Poiseuille's Law: R ∝ 1/r⁴)
- Turbulent in large airways: Resistance ∝ Flow² (explains flow limitation)
| Condition | Elastic Work | Resistive Work | Total Work | Primary Mechanism |
|---|---|---|---|---|
| Normal rest | 0.3-0.5 J/L | 0.2-0.3 J/L | 0.5-0.8 J/L | Balanced efficiency |
| Restrictive disease | ↑↑ 2-4 J/L | Normal | ↑↑ 2-4 J/L | ↓ Compliance requires ↑ pressure |
| Obstructive disease | Normal | ↑↑ 2-4 J/L | ↑↑ 2-4 J/L | ↑ Resistance, prolonged expiration |
| Acute pulmonary edema | ↑↑ 3-5 J/L | ↑ 1-2 J/L | ↑↑↑ 4-7 J/L | ↓ Compliance + ↑ resistance |
| Maximal exercise | ↑ 1-2 J/L | ↑↑ 3-5 J/L | ↑↑↑ 5-8 J/L | High flow rates, turbulence |
📌 Remember: "Work = Pressure × Volume" - Patients minimize work by adopting optimal breathing patterns. Restrictive disease → rapid shallow breathing (↓ elastic work per breath, ↑ rate). Obstructive disease → slow deep breathing (↓ resistive work from lower flow rates, ↓ rate).
Lung Volumes: The Capacity Spectrum
Lung volumes partition total lung capacity into functional compartments, with four primary volumes and four capacities (combinations of volumes) providing diagnostic patterns for restrictive versus obstructive disease.
- Primary Volumes (Non-overlapping)
- Tidal Volume (TV): 500 mL (6-8 mL/kg) - Volume per breath at rest
- Inspiratory Reserve Volume (IRV): 3000 mL - Maximal additional inspiration
- Expiratory Reserve Volume (ERV): 1200 mL - Maximal additional expiration
- Residual Volume (RV): 1200 mL - Volume remaining after maximal expiration
- Cannot be measured by spirometry (requires body plethysmography or dilution)
- ↑ in obstructive disease (air trapping), ↑ with age
- Capacities (Combinations)
- Inspiratory Capacity (IC): TV + IRV = 3500 mL - Maximal inspiration from FRC
- Functional Residual Capacity (FRC): ERV + RV = 2400 mL - Lung volume at rest
- Equilibrium point where lung recoil inward = chest wall recoil outward
- Oxygen reservoir during expiration (prevents desaturation between breaths)
- Vital Capacity (VC): TV + IRV + ERV = 4700 mL - Maximal voluntary ventilation
- Total Lung Capacity (TLC): TV + IRV + ERV + RV = 6000 mL - Maximal lung volume
⭐ Clinical Pearl: FRC of 2400 mL provides an oxygen reservoir containing approximately 500 mL O₂ (at 21% FiO₂), preventing arterial desaturation during the 2-3 seconds of expiration when no fresh gas enters. Rapid sequence intubation with apnea causes desaturation faster in patients with ↓ FRC (obesity, pregnancy, supine position) because this reservoir is depleted.
This mechanical foundation connects directly to where these pressure-volume relationships determine minute ventilation and alveolar ventilation, and for detailed analysis of compliance and resistance patterns in disease states.
📚 Ventilation Mechanics: The Pressure-Volume Engine
🔬 Ventilation-Perfusion Matching: The Efficiency Equation
The lung matches ventilation (V̇) to perfusion (Q̇) across 300 million alveolar units, creating regional V̇/Q̇ ratios from 0 (shunt) to ∞ (dead space). Optimal gas exchange occurs when V̇/Q̇ = 0.8-1.0, but gravitational and physiological factors create V̇/Q̇ heterogeneity even in healthy lungs. Understanding this matching reveals why hypoxemia develops in pulmonary embolism, pneumonia, and ARDS with characteristic patterns.
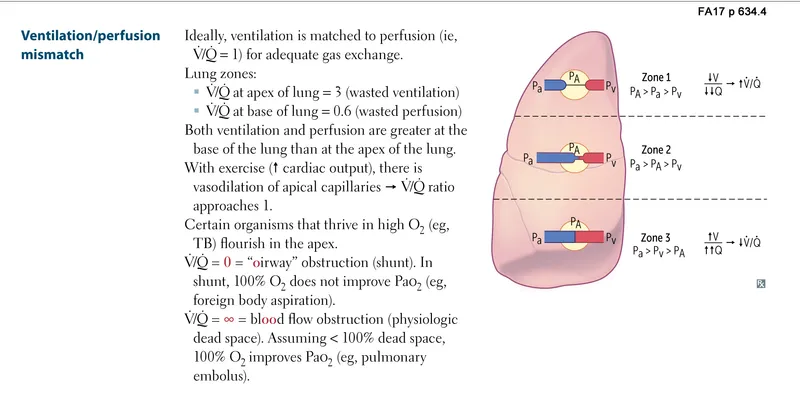
Regional V̇/Q̇ Distribution: Gravity's Influence
In upright lungs, both ventilation and perfusion increase from apex to base, but perfusion increases more steeply, creating V̇/Q̇ gradients. At the apex, V̇/Q̇ = 3.0 (high ventilation, low perfusion), while at the base V̇/Q̇ = 0.6 (high perfusion, relatively lower ventilation).
- Apical Lung Zones (Zone 1)
- V̇/Q̇ ratio: 3.0 (ventilation exceeds perfusion)
- Alveolar PO₂: 130 mmHg (high, approaching inspired PO₂)
- Alveolar PCO₂: 28 mmHg (low, hyperventilation effect)
- Perfusion: Minimal at rest, 0 during hypotension
- Ventilation: 60% of basal ventilation per unit volume
- Clinical relevance: Preferential tuberculosis localization (high PO₂)
- Mid-Lung Zones (Zone 2)
- V̇/Q̇ ratio: 1.0 (optimal matching)
- Alveolar PO₂: 100 mmHg (arterial equilibrium)
- Alveolar PCO₂: 40 mmHg (arterial equilibrium)
- Perfusion: Intermittent, pulsatile flow
- Represents ideal gas exchange conditions
- Basal Lung Zones (Zone 3)
- V̇/Q̇ ratio: 0.6 (perfusion exceeds ventilation)
- Alveolar PO₂: 89 mmHg (lower than arterial)
- Alveolar PCO₂: 42 mmHg (higher than arterial)
- Perfusion: 130% of apical perfusion per unit volume
- Ventilation: 100% baseline (less than perfusion increase)
- Clinical relevance: Preferential pulmonary edema, atelectasis
💡 Master This: The normal A-a gradient of 10-15 mmHg results primarily from basal V̇/Q̇ mismatch (V̇/Q̇ = 0.6) where mixed venous blood is incompletely oxygenated. This physiological shunt accounts for 2-3% of cardiac output even in healthy lungs, explaining why breathing room air produces PaO₂ of 95-100 mmHg rather than alveolar PO₂ of 100-105 mmHg.
V̇/Q̇ Extremes: Shunt and Dead Space
Perfect gas exchange requires V̇/Q̇ = 1.0, but pathological conditions create extreme ratios approaching 0 (shunt) or ∞ (dead space). These extremes have distinct effects on arterial blood gases and responses to supplemental oxygen.
- Shunt (V̇/Q̇ = 0) - Perfusion Without Ventilation
- Anatomic shunt: 2-3% of cardiac output (bronchial, thebesian veins)
- Pathological causes: Atelectasis, pneumonia, pulmonary edema, ARDS
- Effect on PaO₂: ↓↓ (refractory to supplemental O₂ if shunt fraction >30%)
- Effect on PaCO₂: Usually normal (↑ ventilation compensates via peripheral chemoreceptors)
- A-a gradient: ↑↑ (widened, increases with ↑ FiO₂)
- Clinical calculation: Shunt fraction = (CcO₂ - CaO₂) / (CcO₂ - Cv̄O₂)
- Normal: <5% of cardiac output
- Symptomatic hypoxemia: >15% shunt fraction
- Refractory hypoxemia: >30% shunt fraction
- Dead Space (V̇/Q̇ = ∞) - Ventilation Without Perfusion
- Anatomic dead space: 150 mL conducting airways (2.2 mL/kg)
- Alveolar dead space: Ventilated but unperfused alveoli
- Pathological causes: Pulmonary embolism, low cardiac output, ARDS
- Effect on PaO₂: ↓ (modest, improves with supplemental O₂)
- Effect on PaCO₂: ↑↑ (requires ↑ minute ventilation to maintain normal PaCO₂)
- Dead space fraction: VD/VT = (PaCO₂ - PĒ CO₂) / PaCO₂
- Normal: 0.2-0.3 (20-30% of tidal volume)
- Pulmonary embolism: 0.4-0.6 (40-60%)
- ARDS: 0.5-0.7 (50-70%)
| V̇/Q̇ State | Ratio | Alveolar PO₂ | Alveolar PCO₂ | A-a Gradient | O₂ Response | Clinical Examples |
|---|---|---|---|---|---|---|
| Normal | 0.8-1.0 | 100 mmHg | 40 mmHg | 10-15 mmHg | Excellent | Healthy lung mid-zones |
| High V̇/Q̇ | 2-3 | 130 mmHg | 28 mmHg | Normal | Excellent | Apical lung, pulmonary embolism |
| Low V̇/Q̇ | 0.1-0.6 | 60-89 mmHg | 42-50 mmHg | ↑ 20-40 mmHg | Good | Basal lung, COPD, asthma |
| Shunt | 0 | Variable | Variable | ↑↑ >50 mmHg | Poor | Atelectasis, pneumonia, ARDS |
| Dead space | ∞ | 130 mmHg | 0 mmHg | Normal | Excellent | PE, low cardiac output, PEEP |
📌 Remember: "Shunt = Stubborn, Dead Space = Demands" - Shunt causes refractory hypoxemia (stubborn, doesn't respond to O₂ if shunt >30%). Dead space demands increased minute ventilation to eliminate CO₂ (PaCO₂ rises unless total ventilation increases proportionally).
Hypoxic Pulmonary Vasoconstriction: The Auto-Regulation
When alveolar PO₂ falls below 60-70 mmHg, pulmonary arterioles constrict within 2-10 minutes, diverting blood flow away from poorly ventilated regions toward better-ventilated areas. This mechanism optimizes V̇/Q̇ matching but becomes maladaptive in global hypoxia.
- Mechanism
- Sensor: Pulmonary artery smooth muscle cells detect alveolar PO₂
- Threshold: Activated when alveolar PO₂ <60-70 mmHg
- Time course: Onset 2-10 minutes, maximum effect 30-60 minutes
- Mediators: ↓ K⁺ channel activity → depolarization → Ca²⁺ influx → contraction
- Inhibitors: Nitric oxide, prostacyclin, calcium channel blockers
- Physiological Benefits
- Diverts blood from low V̇/Q̇ regions to high V̇/Q̇ regions
- Reduces shunt fraction by 30-50% in regional disease
- Improves PaO₂ by 10-20 mmHg in unilateral pneumonia
- Essential for fetal circulation (maintains high pulmonary vascular resistance)
- Pathological Consequences
- High altitude: Global alveolar hypoxia → ↑ pulmonary artery pressure → right heart strain
- Chronic hypoxemia: Sustained vasoconstriction → pulmonary hypertension → cor pulmonale
- ARDS: Heterogeneous vasoconstriction worsens V̇/Q̇ mismatch
- Anesthetic inhibition: Volatile anesthetics inhibit HPV by 50-60% (worsen shunt)
⭐ Clinical Pearl: Administering 100% oxygen to patients with regional lung disease (pneumonia, atelectasis) can paradoxically worsen hypoxemia by 5-10 mmHg through absorption atelectasis and inhibition of hypoxic pulmonary vasoconstriction. The high PO₂ abolishes HPV, allowing increased blood flow through collapsed/consolidated regions, increasing shunt fraction from 15% to 25%.
This V̇/Q̇ matching foundation directly connects to where these principles determine diffusion capacity and oxygen uptake, and for compensatory mechanisms in V̇/Q̇ mismatch.
🔬 Ventilation-Perfusion Matching: The Efficiency Equation
🏥 Gas Transport Mastery: The Oxygen-Hemoglobin Alliance
Oxygen delivery to tissues requires hemoglobin-mediated transport carrying 98% of arterial oxygen content, while dissolved oxygen contributes only 2%. The oxygen-hemoglobin dissociation curve's sigmoidal shape provides both efficient loading at pulmonary capillaries (PO₂ 100 mmHg, saturation 97-98%) and efficient unloading at tissue capillaries (PO₂ 40 mmHg, saturation 75%). Understanding curve shifts and their clinical triggers predicts tissue oxygenation in anemia, shock, and metabolic derangements.
Oxygen Content Calculation: The Carrying Capacity
Total arterial oxygen content (CaO₂) combines hemoglobin-bound oxygen (98%) with dissolved oxygen (2%), typically totaling 20 mL O₂/dL blood. This content, multiplied by cardiac output, determines oxygen delivery (DO₂) of 1000 mL O₂/min at rest.
$$\text{CaO}_2 = (1.34 \times \text{Hb} \times \text{SaO}_2) + (0.003 \times \text{PaO}_2)$$
-
Hemoglobin-bound oxygen: 1.34 mL O₂/g Hb × 15 g/dL Hb × 0.98 saturation = 19.7 mL O₂/dL
-
Dissolved oxygen: 0.003 mL O₂/mmHg/dL × 100 mmHg PaO₂ = 0.3 mL O₂/dL
-
Total CaO₂: 20 mL O₂/dL blood (normal range 18-22 mL/dL)
-
Oxygen Delivery (DO₂)
- Formula: DO₂ = CaO₂ × Cardiac Output × 10
- Normal: 20 mL/dL × 5 L/min × 10 = 1000 mL O₂/min
- Critical threshold: <400 mL/min → anaerobic metabolism, lactic acidosis
- Compensatory mechanisms: ↑ Cardiac output (up to 3× baseline), ↑ oxygen extraction
-
Oxygen Extraction Ratio (O₂ER)
- Formula: O₂ER = (CaO₂ - Cv̄O₂) / CaO₂
- Normal: 25% at rest (extracting 5 mL O₂/dL from 20 mL O₂/dL)
- Exercise: ↑ to 75-80% (maximal extraction 15 mL O₂/dL)
- Shock states: ↑ to 50-60% as compensatory mechanism
💡 Master This: In anemia with hemoglobin 7.5 g/dL (50% of normal), CaO₂ falls to 10 mL/dL despite normal PaO₂ and saturation. To maintain oxygen delivery of 1000 mL/min, cardiac output must double to 10 L/min. This explains why anemic patients develop high-output cardiac failure and why increasing PaO₂ from 100 to 400 mmHg (dissolved oxygen 0.3 → 1.2 mL/dL) provides minimal benefit.
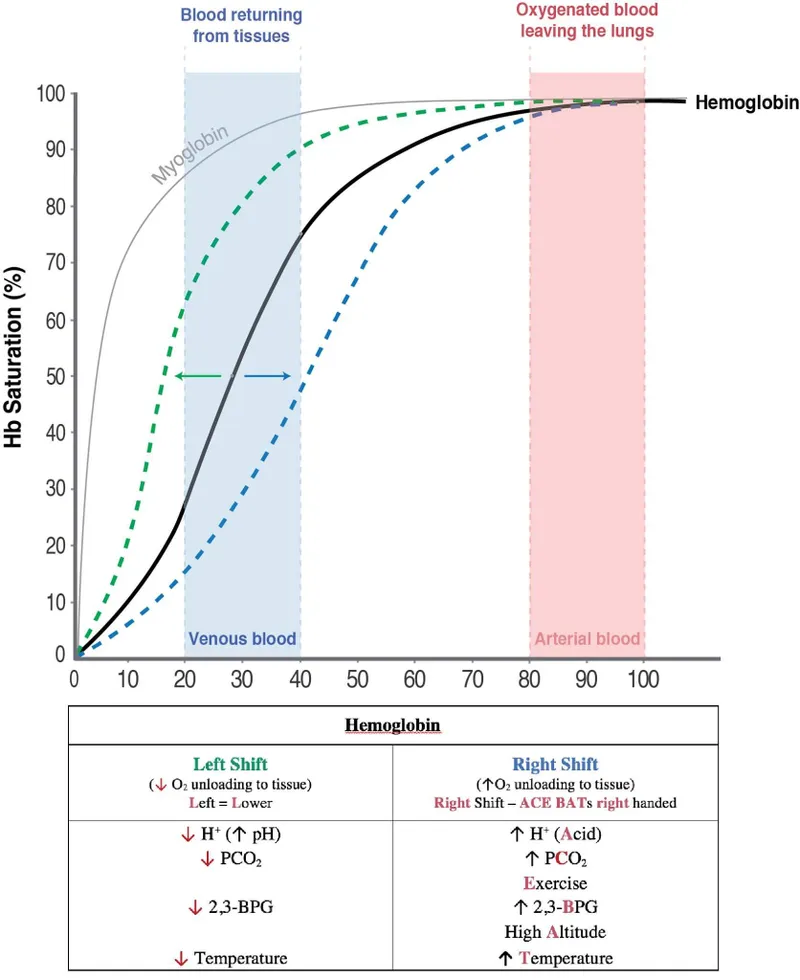
The Oxyhemoglobin Dissociation Curve: Loading and Unloading
The sigmoidal curve shape reflects hemoglobin's cooperative binding where each oxygen molecule bound increases affinity for subsequent molecules. The P₅₀ (PO₂ at 50% saturation) of 27 mmHg defines the curve position, with rightward shifts (↑ P₅₀) facilitating oxygen unloading to tissues.
- Plateau Region (PO₂ 60-100 mmHg)
- Saturation: 90-98% (flat portion provides safety margin)
- Clinical significance: PO₂ can fall from 100 to 60 mmHg with only 8% ↓ saturation
- Pulmonary capillary loading: Occurs in plateau region (PO₂ 100 mmHg)
- Supplemental oxygen effect: Minimal saturation change above PO₂ 60 mmHg
- Steep Region (PO₂ 20-60 mmHg)
- Saturation: 35-90% (steep slope enables efficient unloading)
- Tissue capillary unloading: Occurs in steep region (PO₂ 40 mmHg)
- Clinical significance: Small PO₂ changes produce large saturation changes
- Tissue extraction: PO₂ 100 → 40 mmHg releases 23% of oxygen content
- P₅₀ Value (Curve Position)
- Normal: 27 mmHg (PO₂ producing 50% saturation)
- Rightward shift (↑ P₅₀ to 30-35 mmHg): ↓ Affinity, ↑ tissue unloading
- Leftward shift (↓ P₅₀ to 20-25 mmHg): ↑ Affinity, ↓ tissue unloading
📌 Remember: "Right = Release" - Rightward curve shifts (↑ P₅₀) enhance oxygen release to tissues. Mnemonic: "CADET, face Right!" = CO₂, Acid, DPG (2,3-DPG), Exercise, Temperature all shift curve right, facilitating oxygen delivery during metabolic stress.
Curve Shifts: Physiological and Pathological Modulators
Four primary factors shift the oxyhemoglobin dissociation curve, with rightward shifts enhancing tissue oxygen delivery during exercise, fever, and metabolic acidosis. Understanding these shifts explains tissue hypoxia despite adequate arterial saturation.
| Factor | Rightward Shift (↑ P₅₀) | Leftward Shift (↓ P₅₀) | Mechanism | Clinical Impact |
|---|---|---|---|---|
| pH | ↓ pH (acidosis) | ↑ pH (alkalosis) | Bohr effect: H⁺ ↓ O₂ affinity | Acidosis: P₅₀ 27→32 mmHg, ↑ tissue O₂ delivery 15% |
| PCO₂ | ↑ PCO₂ (hypercapnia) | ↓ PCO₂ (hypocapnia) | CO₂ + H₂O → H⁺ (carbonic anhydrase) | Hypercapnia: P₅₀ 27→30 mmHg, ↑ O₂ release |
| Temperature | ↑ Temperature (fever) | ↓ Temperature (hypothermia) | Thermal effect on Hb conformation | Fever 40°C: P₅₀ 27→35 mmHg, ↑ tissue extraction 20% |
| 2,3-DPG | ↑ 2,3-DPG | ↓ 2,3-DPG | Binds β chains, ↓ O₂ affinity | High altitude: ↑ 2,3-DPG, P₅₀ 27→31 mmHg after 24-48 hrs |
- Bohr Effect (pH and PCO₂)
- Mechanism: ↑ H⁺ or CO₂ → protonation of hemoglobin → conformational change → ↓ O₂ affinity
- Magnitude: pH 7.4 → 7.2 shifts P₅₀ from 27 → 32 mmHg (↑ 18%)
- Physiological benefit: Metabolically active tissues (↑ CO₂, ↓ pH) extract more oxygen
- Clinical example: Septic shock with lactic acidosis (pH 7.1) → P₅₀ 35 mmHg, ↑ tissue O₂ delivery despite ↓ cardiac output
- 2,3-Diphosphoglycerate (2,3-DPG)
- Production: Glycolysis byproduct in RBCs (Rapoport-Luebering shunt)
- Normal concentration: 4-5 mmol/L in RBCs
- ↑ 2,3-DPG conditions: Chronic hypoxemia (high altitude, anemia, COPD), alkalosis
- High altitude: ↑ 30-40% within 24-48 hours
- Chronic anemia: ↑ 50-60% over weeks
- ↓ 2,3-DPG conditions: Stored blood (depleted after 7-10 days), acidosis, hypophosphatemia
- Clinical impact: Transfusion of stored blood → leftward shift → impaired tissue O₂ delivery for 12-24 hours until 2,3-DPG regenerates
⭐ Clinical Pearl: Massive transfusion with stored blood (2,3-DPG depleted) causes leftward curve shift (P₅₀ 27 → 20 mmHg), reducing tissue oxygen extraction by 25-30% despite normal PaO₂ and saturation. This explains post-transfusion tissue hypoxia and elevated lactate in trauma patients receiving >10 units of packed RBCs. Fresh blood (<7 days old) maintains 2,3-DPG levels and normal P₅₀.
Carbon Dioxide Transport: The Bicarbonate Buffer
Carbon dioxide transport occurs through three mechanisms: dissolved CO₂ (7%), carbamino compounds (23%), and bicarbonate (70%). The bicarbonate system provides the primary buffering mechanism maintaining pH homeostasis with a capacity 20× greater than non-bicarbonate buffers.
- Dissolved CO₂ (7% of total)
- Solubility: 0.03 mmol/L/mmHg (20× more soluble than oxygen)
- Arterial: PCO₂ 40 mmHg = 1.2 mmol/L dissolved
- Venous: PCO₂ 46 mmHg = 1.4 mmol/L dissolved
- Contributes to PCO₂ measurement (not to CO₂
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more
Have doubts about this lesson?
Ask Rezzy, your AI Study Mate, to explain anything you didn't understand
Everything you need for NEET-PG prep
Get full Oncourse access with lessons, practice questions, flashcards and AI study tools.
Scan to download app














