Introduction & Hallmarks - Cellular Countdown
- Cellular Aging (Senescence):
- Fundamental biological process; irreversible arrest of cell proliferation.
- Leads to progressive, time-dependent decline in cellular & organismal physiological function.
- Primary Drivers & Triggers:
- Accumulated macromolecular damage (DNA, proteins).
- Genetic factors & epigenetic alterations (e.g., methylation).
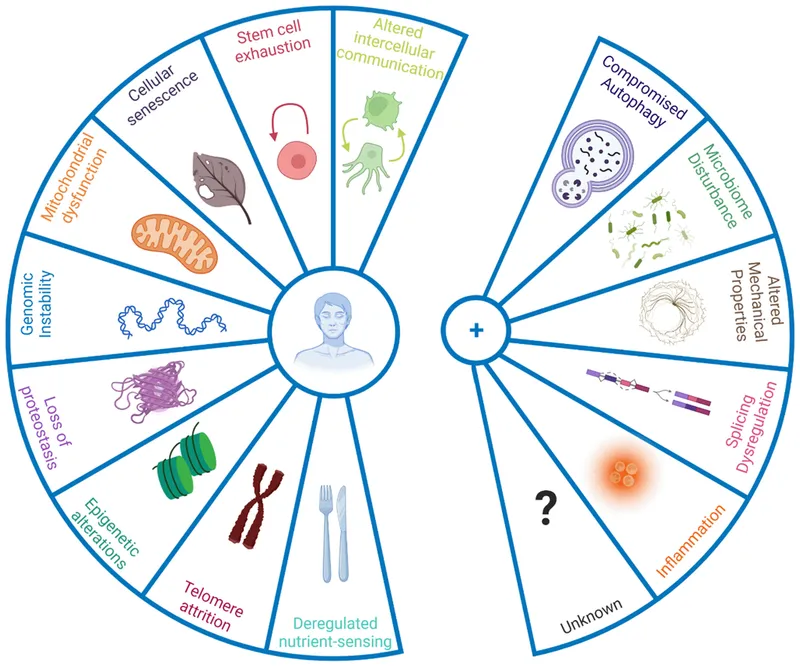
- Key Hallmarks of Aging (Conceptual Framework):
- Genomic instability
- Telomere attrition
- Epigenetic alterations
- Loss of proteostasis
- Deregulated nutrient sensing
- Mitochondrial dysfunction (↓energy, ↑ROS)
- Cellular senescence (SASP secretion)
- Stem cell exhaustion
- Altered intercellular communication (e.g., inflammaging)
⭐ The Hayflick limit describes the finite number of divisions normal human cells can undergo in culture, typically around 40-60 passages.
Mechanisms of Aging - Why Cells Wear Out
📌 GATT LMD CS Hallmarks of Aging:
-
Genomic instability: Accumulated DNA damage (mutations, chromosomal abnormalities).
-
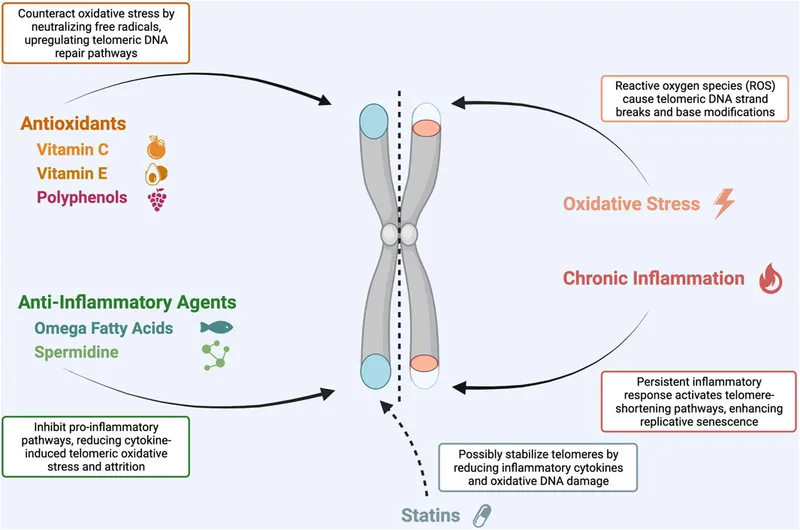
Telomere attrition: Progressive shortening of chromosome ends with cell division; somatic cells have low telomerase.
-
Epigenetic alterations: Changes in DNA methylation, histone modifications affecting gene expression.
-
Loss of proteostasis: Impaired protein folding, ↑misfolded protein aggregation.
-
Deregulated nutrient sensing: Altered pathways (insulin/IGF-1, mTOR, AMPK, sirtuins).
-
Mitochondrial dysfunction: ↓ATP production, ↑Reactive Oxygen Species (ROS).
-
Cellular senescence: Irreversible cell cycle arrest; secretes Senescence-Associated Secretory Phenotype (SASP).
-
Stem cell exhaustion: Depletion and dysfunction, impairing tissue regeneration.
-
Altered intercellular communication: Chronic, low-grade inflammation ("inflammaging").
⭐ Telomerase, an enzyme adding telomeric repeats to chromosome ends, is highly active in germ cells, stem cells, and most cancer cells, but repressed in normal somatic cells, leading to progressive telomere shortening with each cell division.
Cellular Senescence - The Zombie Cell State
- Irreversible cell cycle arrest despite viability; "zombie cells" resist apoptosis, are metabolically active, and accumulate with age.
- Triggers:
- Replicative senescence (telomere shortening, Hayflick limit).
- Persistent DNA Damage Response (DDR).
- Oncogene activation.
- Oxidative stress, inflammation.
- Key Markers:
- Enlarged, flattened morphology.
- ↑ Senescence-Associated β-galactosidase (SA-β-gal) activity.
- Senescence-Associated Heterochromatin Foci (SAHF).
- Core Pathways to Arrest:
⭐ The Senescence-Associated Secretory Phenotype (SASP) involves secretion of pro-inflammatory cytokines (e.g., IL-6, IL-8), chemokines, growth factors, and proteases. These can have context-dependent detrimental (chronic inflammation, tumor promotion) or beneficial (tumor suppression, wound healing) effects.
Modulators & Clinical Links - Aging's Web
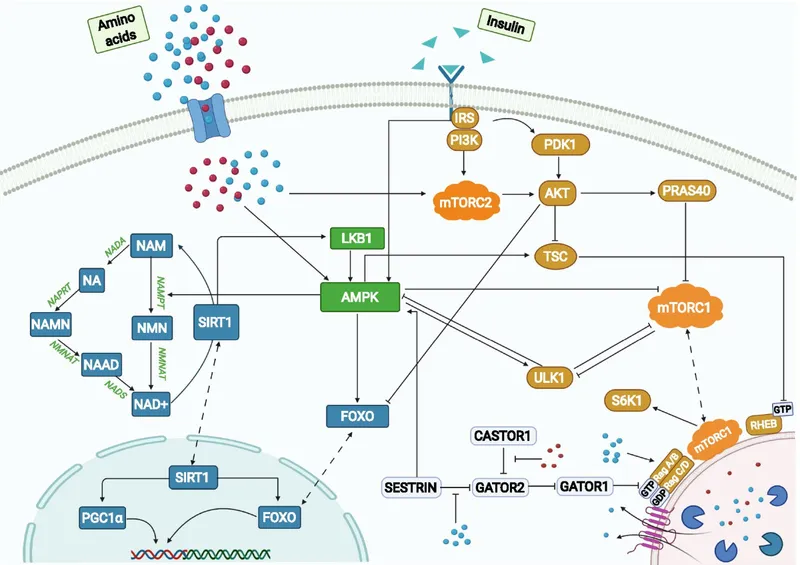
- Nutrient-Sensing Pathways: Key regulators.
- Insulin/IGF-1 Pathway (IIS): Promotes growth, ↓longevity when overactive.
- mTOR (mechanistic Target of Rapamycin): Senses nutrients (amino acids); overactivation → ↓lifespan. Rapamycin inhibits mTOR.
- AMPK (AMP-activated Protein Kinase): Senses low energy (↑AMP/ATP ratio); promotes longevity. Metformin activates AMPK.
- Sirtuins (SIRT1-7): NAD+-dependent deacetylases. Activated by caloric restriction (CR), resveratrol. Promote DNA repair, stress resistance.
⭐ Sirtuins (e.g., SIRT1) are NAD+-dependent deacetylases that are key regulators of cellular health and longevity, often activated by caloric restriction, and they target a wide range of proteins involved in stress resistance, metabolism, and DNA repair.
- Interventions (Experimental/Potential):
- Caloric Restriction (CR): Consistent lifespan extender in many species.
- Pharmacological: Rapamycin, Metformin, Resveratrol.
- Clinical Links: Dysregulation contributes to age-related diseases:
- Neurodegeneration (Alzheimer's, Parkinson's).
- Cardiovascular disease (CVD).
- Cancer.
- Type 2 Diabetes Mellitus (T2DM).
High‑Yield Points - ⚡ Biggest Takeaways
- Telomere attrition with each cell division leads to replicative senescence (Hayflick limit).
- Cellular senescence is characterized by stable cell cycle arrest and a pro-inflammatory Senescence-Associated Secretory Phenotype (SASP).
- Oxidative stress (↑ROS) from sources like mitochondrial dysfunction is a key driver of cellular damage.
- Progressive DNA damage accumulation and genomic instability impair essential cellular functions.
- Loss of proteostasis results in the accumulation of misfolded or aggregated proteins.
- Epigenetic alterations, such as changes in DNA methylation and histone modifications, contribute to aging.
- Progeroid syndromes like Werner syndrome (WRN helicase defect) and Hutchinson-Gilford progeria syndrome (lamin A defect) demonstrate accelerated aging features due to specific genetic defects.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

