Blood and Immunity

On this page
🗺️ The Blood & Immunity Blueprint: Your Hematological Foundation
Master the blood and immune systems, and you unlock the logic behind every infection, bleeding disorder, transfusion reaction, and immunological disease. This lesson builds your understanding from cellular components through complex immune responses, integrating 10+ quantitative thresholds, pattern recognition frameworks, and clinical correlations that transform laboratory values into diagnostic precision. You'll develop systematic approaches to anemia classification, immune dysfunction, and coagulation disorders-essential tools for clinical mastery.
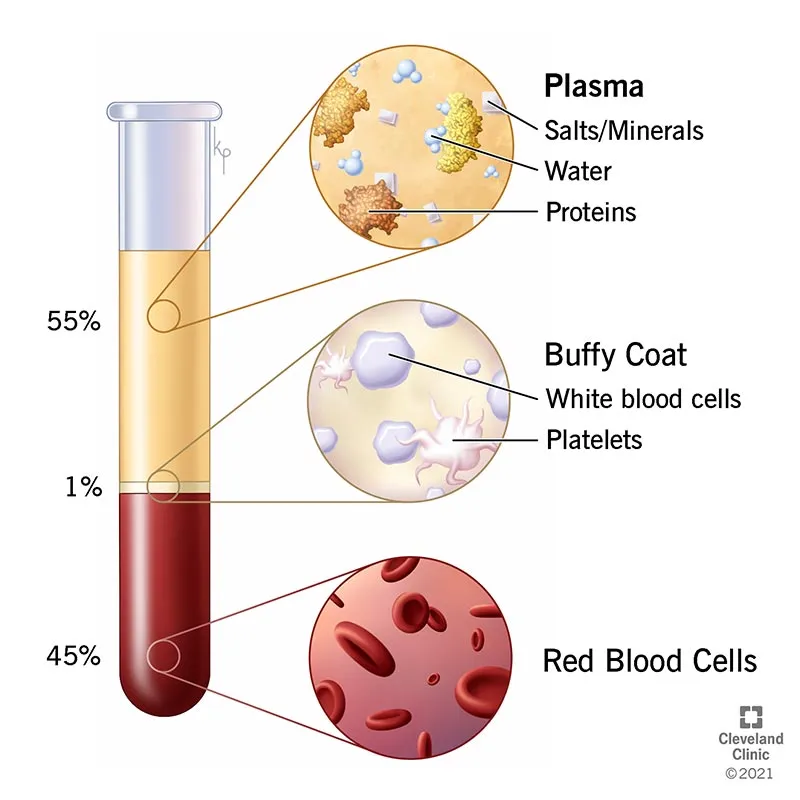
Blood represents 7-8% of total body weight (approximately 5-6 liters in adults), functioning as the body's transport, defense, and homeostatic system. Understanding its composition reveals why specific diseases manifest with characteristic patterns.
- Plasma (55% of blood volume)
- Water: 91-92% of plasma weight
- Proteins: 7-8% (albumin 3.5-5.0 g/dL, globulins 2.0-3.5 g/dL, fibrinogen 200-400 mg/dL)
- Albumin maintains oncotic pressure (25 mmHg), preventing edema
- Globulins include immunoglobulins (IgG 800-1800 mg/dL dominates at 75% of total)
- Fibrinogen converts to fibrin during coagulation
- Electrolytes, nutrients, gases, waste products: 1-2%
- Formed Elements (45% of blood volume = hematocrit)
- Erythrocytes: 4.5-5.5 million/μL (males), 4.0-5.0 million/μL (females)
- Leukocytes: 4,000-11,000/μL total
- Platelets: 150,000-400,000/μL
📌 Remember "PAIN" for Plasma Proteins: Pressure (albumin maintains oncotic), Antibodies (immunoglobulins), Inflammation (acute phase proteins), Network (fibrinogen forms clot mesh). Albumin comprises 60% of total plasma protein and contributes 80% of oncotic pressure despite being only 4% of plasma weight.
The hematocrit (packed cell volume) varies by sex due to testosterone's erythropoietic effects: males 40-54%, females 37-47%. Values below 30% indicate significant anemia requiring investigation, while values above 55% suggest polycythemia with increased thrombotic risk (3-fold elevation).
⭐ Clinical Pearl: The "Rule of Three" validates CBC accuracy-hemoglobin (g/dL) × 3 ≈ hematocrit (%). If Hgb = 12 g/dL, expect Hct ≈ 36%. Discrepancies suggest laboratory error, hemolysis, or abnormal RBC morphology. This rapid check identifies >90% of CBC processing errors.
Blood viscosity increases exponentially with hematocrit elevation. At Hct 45%, viscosity = 3.5-4.0 times water; at Hct 60%, viscosity = 6-8 times water. This explains why polycythemia vera patients develop hyperviscosity syndrome with cerebrovascular accidents when Hct exceeds 55%.
| Parameter | Male Reference | Female Reference | Critical Low | Critical High | Clinical Significance |
|---|---|---|---|---|---|
| Hemoglobin | 13.5-17.5 g/dL | 12.0-16.0 g/dL | <7.0 g/dL | >20 g/dL | <7: transfusion threshold; >20: hyperviscosity |
| Hematocrit | 40-54% | 37-47% | <20% | >60% | <20: severe anemia; >60: stroke risk |
| RBC Count | 4.5-5.5 M/μL | 4.0-5.0 M/μL | <2.0 M/μL | >6.5 M/μL | Guides anemia vs polycythemia diagnosis |
| WBC Count | 4,000-11,000/μL | 4,000-11,000/μL | <2,000/μL | >30,000/μL | <2,000: infection risk; >30,000: leukemia concern |
| Platelet Count | 150,000-400,000/μL | 150,000-400,000/μL | <20,000/μL | >1,000,000/μL | <20,000: spontaneous bleeding; >1M: thrombosis |
💡 Master This: Blood volume regulation depends on albumin's oncotic pressure (25 mmHg) opposing capillary hydrostatic pressure (30 mmHg arterial, 15 mmHg venous). When albumin drops below 2.5 g/dL, net filtration exceeds reabsorption, causing edema. This explains why nephrotic syndrome (albumin <3.0 g/dL) and cirrhosis manifest with peripheral and ascitic fluid accumulation.
Erythrocyte sedimentation rate (ESR) measures RBC aggregation velocity: males 0-15 mm/hr, females 0-20 mm/hr. Elevation indicates ↑ fibrinogen or immunoglobulins promoting rouleaux formation. ESR >100 mm/hr suggests infection, malignancy, or temporal arteritis with 90% specificity, demanding immediate investigation.
🗺️ The Blood & Immunity Blueprint: Your Hematological Foundation
📚 The Erythrocyte Engine: Oxygen Delivery Architecture
Red blood cells represent the body's most specialized transport system, with 2.5 million RBCs produced and destroyed every second to maintain steady-state homeostasis. Understanding erythrocyte structure, hemoglobin biochemistry, and iron metabolism reveals the mechanistic basis for anemia classification and guides targeted treatment.
![]()
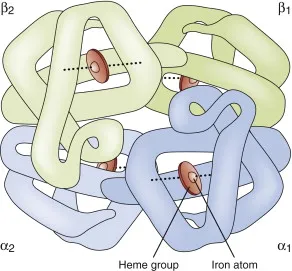
RBC Morphology & Lifespan Determinants
The biconcave disc shape (7-8 μm diameter, 2 μm thick) maximizes surface area-to-volume ratio (140 μm² surface area for 90 fL volume), facilitating oxygen diffusion. This geometry allows RBCs to deform through capillaries as narrow as 3-4 μm without rupture.
- Membrane Structure
- Lipid bilayer with 50% protein, 40% lipid, 10% carbohydrate by weight
- Spectrin-ankyrin cytoskeleton provides deformability
- Hereditary spherocytosis: spectrin deficiency → ↓ deformability → splenic sequestration
- Mean corpuscular hemoglobin concentration (MCHC) 32-36 g/dL in spherocytes vs 32-36 g/dL normal
- Na⁺/K⁺-ATPase maintains osmotic stability (consumes 90% of RBC ATP)
- Metabolic Pathways
- Glycolysis (Embden-Meyerhof): 90% of glucose metabolism → ATP production
- Hexose monophosphate shunt: 10% of glucose → NADPH production
- NADPH maintains glutathione in reduced state
- G6PD deficiency → oxidative hemolysis when NADPH depleted
- Rapoport-Luebering shunt: generates 2,3-DPG
- 2,3-DPG binds hemoglobin β-chains, ↓ oxygen affinity (right-shifts curve)
- ↑ 2,3-DPG in chronic hypoxia, anemia (adaptive response)
📌 Remember "GLAD Pathways" for RBC Metabolism: Glycolysis (ATP for pumps), Luebering (2,3-DPG for O₂ release), Antioxidant (hexose monophosphate shunt for NADPH), Degradation (senescent RBC removal after 120 days). G6PD deficiency affects the "A" pathway, causing hemolysis with oxidant stress (fava beans, sulfa drugs).
Hemoglobin Structure & Oxygen Binding
Hemoglobin comprises 4 globin chains (2α, 2β in adult HbA) and 4 heme groups (protoporphyrin IX + Fe²⁺). Each gram of hemoglobin binds 1.34 mL O₂ when fully saturated, yielding oxygen-carrying capacity = Hgb (g/dL) × 1.34 × 10.
- Oxygen-Hemoglobin Dissociation Curve
- P₅₀ (oxygen tension at 50% saturation): 27 mmHg (normal)
- Right shift (↓ affinity, ↑ tissue delivery): ↑ 2,3-DPG, ↑ H⁺ (↓ pH), ↑ CO₂, ↑ temperature
- Chronic hypoxia → ↑ 2,3-DPG → P₅₀ 30-32 mmHg (adaptive)
- Exercise → ↑ temperature + ↓ pH → enhanced O₂ unloading
- Left shift (↑ affinity, ↓ tissue delivery): ↓ 2,3-DPG, ↓ H⁺ (↑ pH), ↓ CO₂, ↓ temperature, HbF, CO poisoning
- Stored blood (2,3-DPG depletes after 7 days) → P₅₀ 19 mmHg → tissue hypoxia despite normal Hgb
- Carbon monoxide affinity 240-fold > O₂ → COHb >20% causes symptoms
⭐ Clinical Pearl: The Bohr effect explains why exercising muscle receives more oxygen despite unchanged arterial PO₂. Local ↑ CO₂ and ↓ pH shift the curve rightward in capillaries, increasing O₂ extraction from 25% (resting) to 75% (maximal exercise). Conversely, hyperventilation-induced alkalosis (pH 7.55) left-shifts the curve, reducing tissue O₂ delivery and causing paresthesias despite normal SaO₂.
| Hemoglobin Type | Chain Structure | Normal Adult % | Oxygen Affinity | Clinical Context |
|---|---|---|---|---|
| HbA (Adult) | α₂β₂ | 95-98% | P₅₀ = 27 mmHg | Predominant form; β-thalassemia ↓ HbA |
| HbA₂ | α₂δ₂ | 2-3% | Similar to HbA | ↑ to 4-8% in β-thalassemia trait (diagnostic) |
| HbF (Fetal) | α₂γ₂ | <1% adult | P₅₀ = 19 mmHg | ↑ in β-thalassemia, sickle cell (protective) |
| HbS (Sickle) | α₂β₂ˢ | 0% normal | Polymerizes when deoxygenated | Glu→Val at β6; sickling at O₂ sat <85% |
| Methemoglobin | Fe³⁺ form | <1% | Cannot bind O₂ | >15%: cyanosis; >70%: lethal hypoxia |
Iron Metabolism: The Heme Engine
Total body iron: 3-4 g (males), 2-3 g (females). Distribution: 65% hemoglobin, 10% myoglobin/enzymes, 25% storage (ferritin/hemosiderin). Daily requirement: 1-2 mg (males), 2-3 mg (menstruating females).
- Absorption (Duodenum & Proximal Jejunum)
- Dietary iron: heme (10-20% absorbed) vs non-heme (1-5% absorbed)
- DMT1 (divalent metal transporter 1) imports Fe²⁺ into enterocytes
- Ferroportin exports iron to plasma (inhibited by hepcidin)
- Transferrin binds plasma iron (normal saturation 20-45%)
- Regulation by Hepcidin
- Liver produces hepcidin in response to ↑ iron stores, inflammation
- Hepcidin binds ferroportin → internalization/degradation → ↓ iron absorption and ↓ macrophage iron release
- Anemia of chronic disease: ↑ hepcidin → ↓ iron availability despite adequate stores
- Serum iron ↓, TIBC ↓, ferritin ↑ (>100 ng/mL), transferrin saturation <20%
💡 Master This: Iron deficiency anemia evolves through three stages: (1) ↓ storage iron (ferritin <30 ng/mL, normal Hgb), (2) ↓ transport iron (transferrin saturation <15%, normal Hgb), (3) ↓ functional iron (Hgb <12 g/dL, MCV <80 fL). Treating stage 3 requires 3-6 months of supplementation to replenish stores, not just normalize hemoglobin. Premature discontinuation causes relapse in 60% of patients.
RBC Indices: The Anemia Classification Framework
- Mean Corpuscular Volume (MCV): RBC volume
- Normal: 80-100 fL
- Microcytic (<80 fL): iron deficiency, thalassemia, anemia of chronic disease, sideroblastic anemia
- Macrocytic (>100 fL): B₁₂/folate deficiency, alcoholism, hypothyroidism, liver disease
- Mean Corpuscular Hemoglobin (MCH): Hgb per RBC
- Normal: 27-33 pg
- Parallels MCV (microcytic = hypochromic)
- Mean Corpuscular Hemoglobin Concentration (MCHC): Hgb concentration
- Normal: 32-36 g/dL
- ↓ in iron deficiency, ↑ in spherocytosis (dehydrated cells)
- Red Cell Distribution Width (RDW): Variation in RBC size
- Normal: 11.5-14.5%
- ↑ RDW + microcytosis: iron deficiency (mixed RBC populations)
- Normal RDW + microcytosis: thalassemia trait (uniform small cells)
📌 Remember "MR. MCHC" for Microcytic Anemia: Microcytic (MCV <80), Reticulocyte response (↓ in production defects), Marrow iron (absent in IDA, present in ACD), Chronic disease (hepcidin ↑), Heme synthesis defect (sideroblastic), Chronic blood loss (GI, menstrual). RDW >15% favors iron deficiency over thalassemia trait with 85% sensitivity.
📚 The Erythrocyte Engine: Oxygen Delivery Architecture
🔬 The Leukocyte Legion: Cellular Defense Stratification
White blood cells orchestrate immune surveillance, pathogen elimination, and tissue repair through specialized subpopulations with distinct functions. Understanding leukocyte kinetics, activation mechanisms, and dysfunction patterns enables systematic approach to infections, immunodeficiencies, and hematologic malignancies.
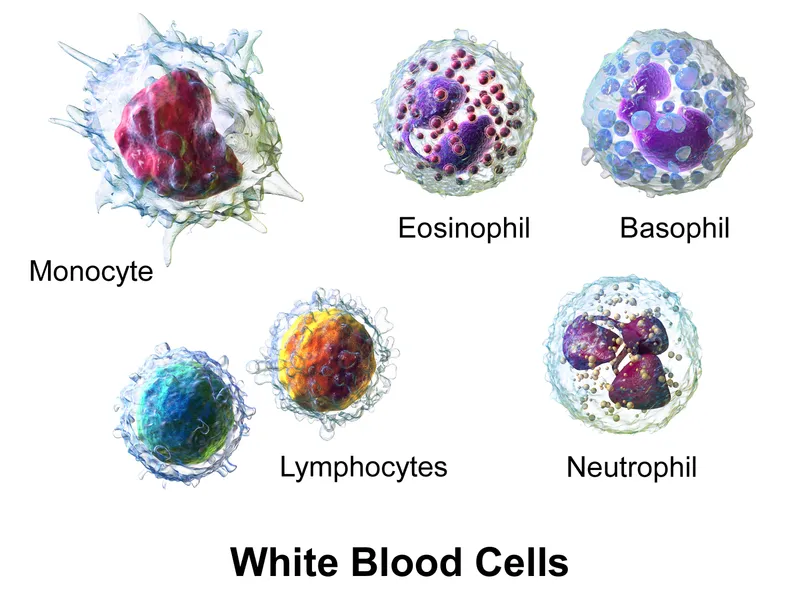
Leukocyte Differential: The Cellular Census
Total WBC count 4,000-11,000/μL comprises five major populations with characteristic distributions and half-lives.
- Neutrophils (40-70%, absolute count 2,000-7,500/μL)
- Half-life: 6-8 hours in circulation, 1-2 days in tissues
- Marginated pool (adhered to vessel walls) equals circulating pool
- Stress demargination → rapid doubling of circulating count without production ↑
- Functions: phagocytosis, degranulation, NET formation
- Primary granules: myeloperoxidase, elastase, defensins
- Secondary granules: lactoferrin, collagenase, gelatinase
- Tertiary granules: gelatinase, cathepsins
- Left shift: ↑ bands (>10%) indicates acute bacterial infection
- Toxic granulation, Döhle bodies, cytoplasmic vacuoles suggest sepsis
- Lymphocytes (20-40%, absolute count 1,000-4,800/μL)
- T cells: 60-80% of lymphocytes (CD4 500-1,500/μL, CD8 300-1,000/μL)
- B cells: 10-20% of lymphocytes
- NK cells: 5-15% of lymphocytes
- Lifespan: hours (activated) to years (memory cells)
- Monocytes (2-10%, absolute count 200-1,000/μL)
- Largest leukocyte (12-20 μm diameter)
- Circulate 1-3 days, then migrate to tissues → macrophages (lifespan months)
- Functions: phagocytosis, antigen presentation, cytokine production (TNF-α, IL-1, IL-6)
- Eosinophils (1-4%, absolute count 50-500/μL)
- Half-life: 8-12 hours in circulation, 8-12 days in tissues
- Functions: parasitic defense, allergic inflammation modulation
- Eosinophilia (>500/μL): parasites, allergies, drugs, malignancy, autoimmune
- Basophils (0-1%, absolute count 0-100/μL)
- Rarest leukocyte; tissue counterpart = mast cells
- Granules contain histamine, heparin, leukotrienes
- Basophilia (>100/μL): rare, seen in myeloproliferative disorders
📌 Remember "Neutrophils Like Making Everything Better" for WBC Differential: Neutrophils (60%), Lymphocytes (30%), Monocytes (6%), Eosinophils (3%), Basophils (1%). This "60-30-6-3-1 rule" provides rapid reference for identifying abnormal differentials. Reversal (lymphocyte predominance) occurs in viral infections, pertussis, and chronic lymphocytic leukemia.
Neutrophil Function: The Phagocytic Powerhouse
Neutrophils provide first-line defense against bacterial and fungal pathogens through coordinated steps requiring 5-10 minutes from chemotaxis to pathogen killing.
- Margination & Rolling
- Selectins (E-selectin, P-selectin) mediate initial tethering to endothelium
- Inflammatory signals (IL-1, TNF-α) upregulate selectin expression within 2-4 hours
- Tight Adhesion
- Integrins (LFA-1, Mac-1) bind ICAM-1 on endothelium
- Leukocyte adhesion deficiency (LAD): integrin defects → recurrent infections, WBC >20,000/μL without pus
- Diapedesis
- Neutrophils squeeze between endothelial cells via PECAM-1 interactions
- Transmigration time: 2-5 minutes per cell
- Chemotaxis
- Gradient sensing toward C5a, LTB₄, IL-8, bacterial products
- Migration speed: 10-20 μm/min in tissues
- Phagocytosis & Killing
- Opsonization (IgG, C3b) enhances recognition 1,000-fold
- Phagosome-lysosome fusion occurs within 1-2 minutes
- Oxidative burst: NADPH oxidase generates superoxide (O₂⁻) → H₂O₂ → HOCl (bleach)
- Chronic granulomatous disease (CGD): NADPH oxidase defect → catalase-positive organism infections (Staph aureus, Aspergillus, Burkholderia)
- Nitroblue tetrazolium (NBT) test: <5% positive in CGD vs >95% normal
- Non-oxidative killing: defensins, lysozyme, lactoferrin
⭐ Clinical Pearl: Neutropenia severity predicts infection risk: mild (1,000-1,500/μL, minimal risk), moderate (500-1,000/μL, 10% infection rate), severe (<500/μL, 50% infection rate), profound (<100/μL, >90% infection rate within weeks). Fever in neutropenic patient (<500/μL) constitutes medical emergency requiring empiric broad-spectrum antibiotics within 1 hour (every hour delay increases mortality 10-15%).
| WBC Disorder | Absolute Count | Mechanism | Clinical Clues | High-Yield Association |
|---|---|---|---|---|
| Leukocytosis | >11,000/μL | Infection, inflammation, stress, malignancy | Left shift, toxic granulation | Leukemoid reaction: >50,000/μL mimics leukemia |
| Neutrophilia | >7,500/μL | Bacterial infection, corticosteroids, stress | Bands >10%, Döhle bodies | Corticosteroids demarginate within 4-6 hours |
| Lymphocytosis | >4,800/μL | Viral infection, pertussis, CLL | Atypical lymphocytes (EBV) | Absolute count >5,000 in adults suggests CLL |
| Monocytosis | >1,000/μL | Chronic infection (TB), recovery, malignancy | Large cells with folded nuclei | Monocyte count >1,000 with anemia suggests MDS |
| Eosinophilia | >500/μL | Parasites, allergies, drugs, malignancy | Absolute count matters, not % | >1,500/μL: organ damage risk (cardiac, pulmonary) |
Lymphocyte Subsets: Adaptive Immunity Architects
- T Lymphocytes (Cell-Mediated Immunity)
- CD4+ Helper T cells: 500-1,500/μL (normal)
- Th1: IFN-γ production → macrophage activation (intracellular pathogens)
- Th2: IL-4, IL-5, IL-13 → eosinophil activation, IgE production (helminths, allergies)
- Th17: IL-17 production → neutrophil recruitment (extracellular bacteria, fungi)
- Treg: IL-10, TGF-β → immune suppression (prevent autoimmunity)
- CD8+ Cytotoxic T cells: 300-1,000/μL (normal)
- Perforin/granzyme-mediated killing of virus-infected, tumor cells
- Require MHC class I presentation (all nucleated cells)
- HIV targets CD4+ cells: AIDS defined as CD4 <200/μL or opportunistic infection
- CD4 <200: Pneumocystis pneumonia, toxoplasmosis risk
- CD4 <50: CMV retinitis, MAC infection risk
- CD4+ Helper T cells: 500-1,500/μL (normal)
- B Lymphocytes (Humoral Immunity)
- Mature in bone marrow → migrate to secondary lymphoid organs
- Antibody production: 2-3 g/day total immunoglobulins
- Memory B cells provide rapid anamnestic response (2-3 days vs 7-10 days primary)
- Natural Killer Cells
- Kill cells lacking MHC class I (tumor cells, virus-infected cells evading T cell recognition)
- Antibody-dependent cell-mediated cytotoxicity (ADCC)
- Produce IFN-γ early in infection (innate immunity bridge)
💡 Master This: The CD4:CD8 ratio (normally 1.5-2.5) provides diagnostic insight: ratio <1.0 suggests HIV, viral infection, or immunosuppression; ratio >3.0 suggests sarcoidosis, autoimmune disease, or Th2-predominant states. In HIV, ratio inverts as CD4 cells decline, with ratio <0.5 indicating advanced disease. Monitoring absolute CD4 count guides prophylaxis timing better than ratio alone.
🔬 The Leukocyte Legion: Cellular Defense Stratification
🏥 Hemostasis & Coagulation: The Clotting Command Center
Hemostasis maintains blood fluidity while enabling rapid clot formation at injury sites-a balance achieved through coordinated platelet activation, coagulation cascade amplification, and fibrinolytic regulation. Understanding this system's three phases (primary hemostasis, secondary hemostasis, fibrinolysis) reveals mechanisms underlying bleeding disorders and thrombotic diseases.
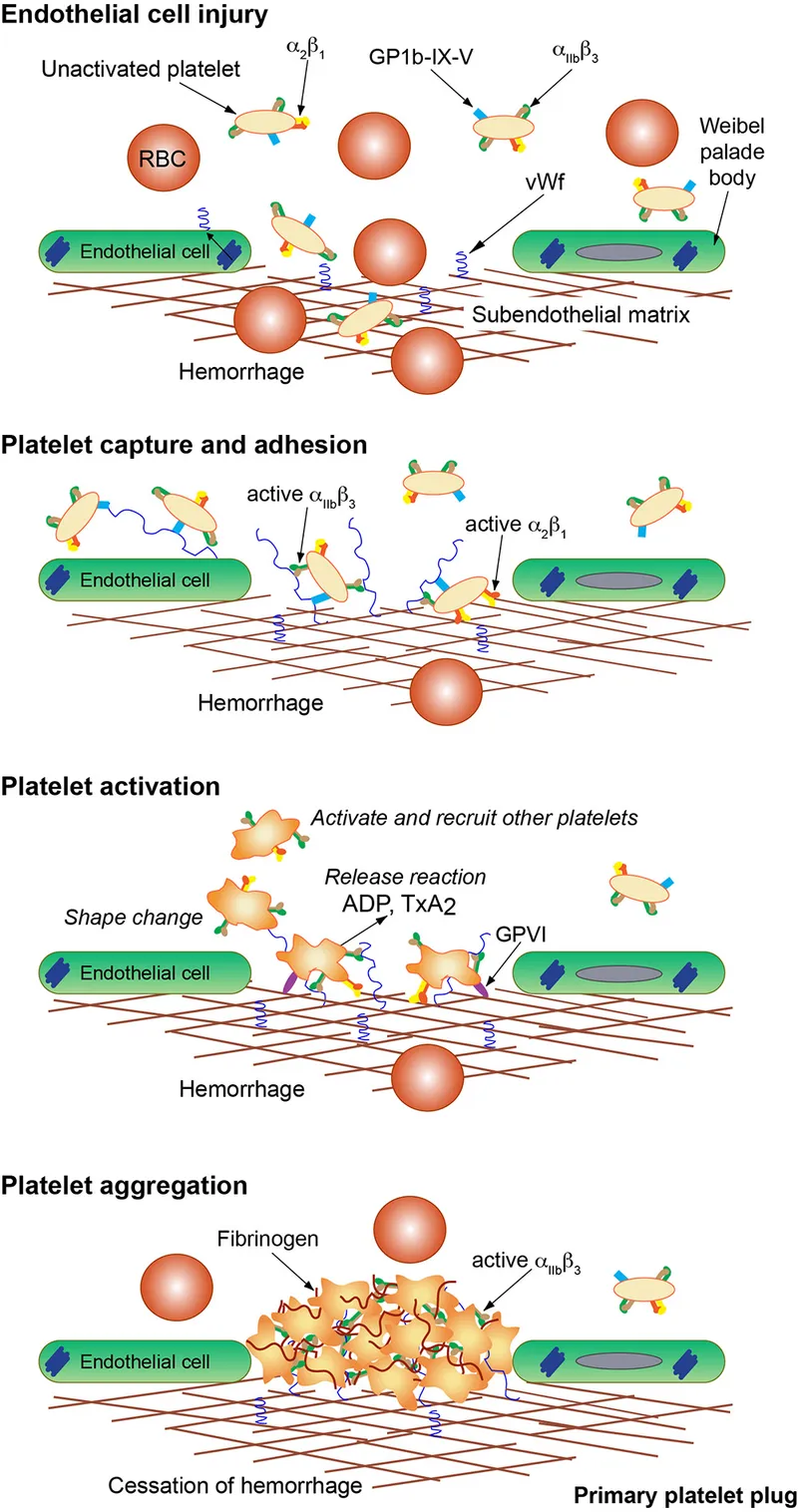
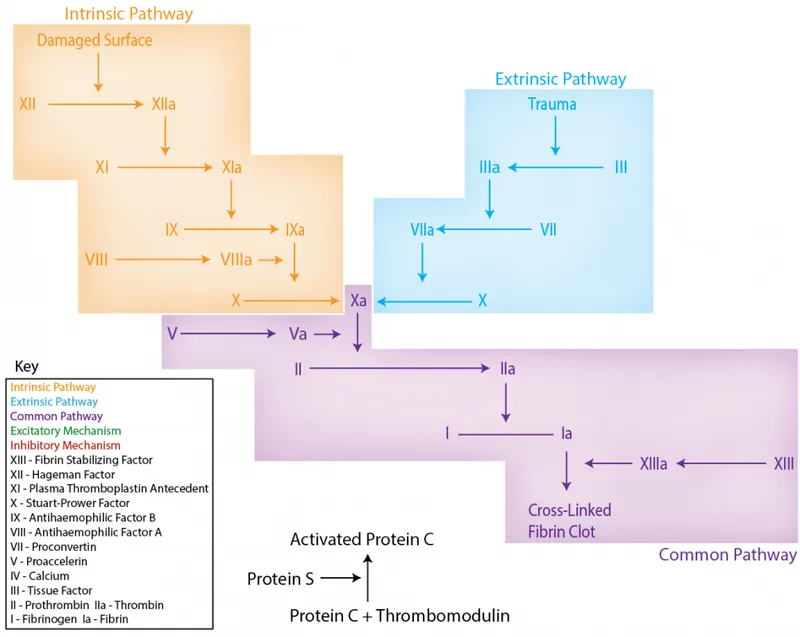
Primary Hemostasis: The Platelet Plug (0-5 Minutes)
Platelets (150,000-400,000/μL) are anucleate cell fragments (2-4 μm diameter) derived from megakaryocytes with lifespan 7-10 days. Approximately one-third of platelets sequester in spleen; splenectomy increases count 50-100%.
- Platelet Adhesion
- Endothelial injury exposes subendothelial collagen and von Willebrand factor (vWF)
- Platelet GP Ib-IX-V receptor binds vWF → initial tethering (shear-dependent)
- Platelet GP Ia-IIa receptor binds collagen → firm adhesion
- Von Willebrand disease: vWF deficiency/dysfunction → mucocutaneous bleeding
- Type 1 (70% of cases): partial quantitative deficiency (vWF 20-50% normal)
- Type 2: qualitative defects (vWF:RCo/vWF:Ag ratio <0.6)
- Type 3 (rare): complete deficiency (vWF <5%), severe bleeding
- Platelet Activation
- Collagen, thrombin, ADP, thromboxane A₂ (TXA₂) trigger activation
- Shape change: discoid → pseudopod formation (cytoskeletal reorganization)
- Granule release:
- Dense granules: ADP, ATP, Ca²⁺, serotonin
- α-granules: fibrinogen, vWF, factor V, platelet factor 4 (PF4)
- TXA₂ synthesis via cyclooxygenase-1 (COX-1) → vasoconstriction, platelet recruitment
- Aspirin irreversibly acetylates COX-1 → ↓ TXA₂ for platelet lifespan
- Clinical effect: 75-100 mg/day reduces cardiovascular events 20-25%
- Platelet Aggregation
- GP IIb-IIIa receptor activation (requires Ca²⁺, inside-out signaling)
- Fibrinogen bridges adjacent platelets via GP IIb-IIIa
- Platelet plug forms within 1-3 minutes at injury site
- Glanzmann thrombasthenia: GP IIb-IIIa deficiency → severe mucocutaneous bleeding despite normal platelet count
📌 Remember "VWAP" for Primary Hemostasis: Von Willebrand factor (adhesion), Wound exposure (collagen), Activation (granule release), Plug formation (aggregation). Defects at each step cause characteristic bleeding: vWF deficiency → mucosal bleeding (epistaxis, menorrhagia), thrombocytopenia → petechiae/purpura, GP IIb-IIIa defects → severe bleeding despite normal count.
Secondary Hemostasis: The Coagulation Cascade (5-10 Minutes)
The coagulation cascade amplifies thrombin generation through sequential protease activation, culminating in fibrin mesh formation that stabilizes the platelet plug.
- Intrinsic Pathway (Contact Activation)
- Factor XII → XIIa (contact with collagen, activated platelets)
- XIIa → XI → XIa
- XIa → IX → IXa (requires Ca²⁺)
- IXa + VIIIa (tenase complex) → X → Xa
- Monitored by aPTT (normal 25-35 seconds)
- Hemophilia A (factor VIII deficiency): aPTT ↑, PT normal
- Hemophilia B (factor IX deficiency): aPTT ↑, PT normal
- Extrinsic Pathway (Tissue Factor)
- Tissue factor (TF) exposed at injury site binds factor VII → VIIa
- TF-VIIa complex → X → Xa (fastest pathway, initiates coagulation)
- Monitored by PT/INR (normal PT 11-13 seconds, INR 0.9-1.1)
- Warfarin inhibits vitamin K-dependent factors (II, VII, IX, X) → ↑ PT/INR
- Target INR 2.0-3.0 for most indications, 2.5-3.5 for mechanical valves
- Common Pathway
- Xa + Va (prothrombinase complex) → prothrombin (II) → thrombin (IIa)
- Thrombin cleaves fibrinogen → fibrin monomers → fibrin polymer
- Factor XIIIa (activated by thrombin) cross-links fibrin → stable clot
- Thrombin amplification: 1 molecule factor Xa generates 1,000 molecules thrombin
⭐ Clinical Pearl: The PT/aPTT pattern localizes coagulation defects: ↑ PT + normal aPTT = factor VII deficiency or early warfarin effect (VII has shortest half-life, 4-6 hours); ↑ aPTT + normal PT = hemophilia A/B or heparin; ↑ both = common pathway defect (factors X, V, II, fibrinogen), liver disease, or DIC. Mixing studies differentiate factor deficiency (corrects) from inhibitor (doesn't correct).
| Coagulation Factor | Pathway | Half-Life | Vitamin K-Dependent? | Deficiency Disorder | Lab Abnormality |
|---|---|---|---|---|---|
| Factor VIII | Intrinsic | 8-12 hours | No | Hemophilia A (most common) | aPTT ↑, PT normal |
| Factor IX | Intrinsic | 18-24 hours | Yes | Hemophilia B (Christmas disease) | aPTT ↑, PT normal |
| Factor XI | Intrinsic | 40-80 hours | No | Factor XI deficiency (Ashkenazi Jews) | aPTT ↑, mild bleeding |
| Factor VII | Extrinsic | 4-6 hours | Yes | Factor VII deficiency (rare) | PT ↑, aPTT normal |
| Factor X | Common | 24-48 hours | Yes | Factor X deficiency (rare) | PT ↑, aPTT ↑ |
| Prothrombin (II) | Common | 48-72 hours | Yes | Prothrombin deficiency (rare) | PT ↑, aPTT ↑ |
| Fibrinogen (I) | Common | 72-120 hours | No | Afibrinogenemia, DIC | PT ↑, aPTT ↑, TT ↑ |
Fibrinolysis & Anticoagulation: Clot Control Mechanisms
Endogenous anticoagulants and fibrinolytic system prevent excessive clotting and restore vessel patency after healing.
- Natural Anticoagulants
- Antithrombin (AT): inhibits thrombin (IIa), factors Xa, IXa, XIa
- Heparin potentiates AT activity 1,000-fold (immediate effect)
- AT deficiency: thrombosis risk 50-fold ↑, often <50 years
- Protein C + Protein S: inactivate factors Va, VIIIa (require vitamin K)
- Activated by thrombin-thrombomodulin complex
- Protein C/S deficiency: venous thrombosis, warfarin-induced skin necrosis
- Factor V Leiden (most common thrombophilia, 5% Caucasians): factor Va resistant to protein C → 7-fold ↑ VTE risk
- Tissue factor pathway inhibitor (TFPI): inhibits TF-VIIa complex
- Antithrombin (AT): inhibits thrombin (IIa), factors Xa, IXa, XIa
- Fibrinolytic System
- Tissue plasminogen activator (tPA) converts plasminogen → plasmin
- Plasmin cleaves fibrin → D-dimers (fibrin degradation products)
- D-dimer <500 ng/mL: excludes VTE with >95% negative predictive value
- D-dimer ↑: VTE, DIC, recent surgery, pregnancy, malignancy (low specificity)
- Plasminogen activator inhibitor-1 (PAI-1) inhibits tPA
- α₂-antiplasmin inhibits plasmin (prevents systemic fibrinolysis)
💡 Master This: Disseminated intravascular coagulation (DIC) represents consumption coagulopathy where excessive thrombin generation depletes platelets and clotting factors while activating fibrinolysis. Laboratory hallmarks: platelets <100,000/μL (often <50,000), PT/aPTT ↑, fibrinogen <150 mg/dL, D-dimer >10,000 ng/mL. Mortality 30-50% in severe cases. Treatment targets underlying cause (sepsis, malignancy, trauma); supportive care replaces consumed factors/platelets only if active bleeding.
Anticoagulant Mechanisms & Monitoring
- Heparin (Unfractionated)
- Mechanism: potentiates antithrombin → inhibits thrombin, factor Xa
- Monitoring: aPTT (target 60-80 seconds, 1.5-2.5× control)
- Half-life: 60-90 minutes IV
- Reversal: protamine sulfate (1 mg per 100 units heparin)
- Complication: HIT (heparin-induced thrombocytopenia) in 1-5%, platelets drop >50% after 5-10 days
- Low Molecular Weight Heparin (LMWH)
- Mechanism: preferentially inhibits factor Xa (less thrombin inhibition)
- Monitoring: usually not required; anti-Xa level if needed (target 0.6-1.0 IU/mL)
- Half-life: 3-6 hours SC
- Advantages: predictable dose-response, less HIT risk (<1%)
- Warfarin
- Mechanism: inhibits vitamin K epoxide reductase → ↓ factors II, VII, IX, X, protein C/S
- Monitoring: INR (target 2.0-3.0 most indications)
- Half-life: 36-42 hours (but factor VII depletes first, 4-6 hours)
- Reversal: vitamin K (10 mg PO/IV, effect in 12-24 hours), FFP, PCC (immediate)
- Direct Oral Anticoagulants (DOACs)
- Dabigatran: direct thrombin inhibitor (reversal: idarucizumab)
- Rivaroxaban, apixaban, edoxaban: direct factor Xa inhibitors (reversal: andexanet alfa)
- Advantages: fixed dosing, no monitoring, fewer drug interactions
- Half-lives: 12-17 hours (avoid in severe renal impairment)
[[Chapter: Coagulation an
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more
Have doubts about this lesson?
Ask Rezzy, your AI Study Mate, to explain anything you didn't understand
Everything you need for NEET-PG prep
Get full Oncourse access with lessons, practice questions, flashcards and AI study tools.
Scan to download app
















