Endocrinology

On this page
🎯 The Endocrine Command Network: Master Control Systems
The endocrine system operates through hierarchical feedback loops connecting hypothalamus, pituitary, and peripheral glands into synchronized networks. This architecture enables precise long-distance signaling through hormones-chemical messengers released into bloodstream reaching target tissues expressing specific receptors. Understanding this master control system predicts every endocrine disorder pattern.

Hierarchical Control Architecture
-
Hypothalamic Command Center
- Synthesizes releasing/inhibiting hormones controlling anterior pituitary
- Produces ADH and oxytocin stored in posterior pituitary
- Integrates neural signals from limbic system, circadian rhythms, stress responses
- Portal circulation delivers hormones directly to pituitary (10-100× higher concentration than systemic)
- CRH → ACTH → cortisol (stress axis)
- TRH → TSH → thyroid hormones (metabolic axis)
- GnRH → LH/FSH → sex steroids (reproductive axis)
- GHRH/somatostatin → GH → IGF-1 (growth axis)
-
Pituitary Integration Hub
- Anterior: synthesizes 6 major hormones under hypothalamic control
- Posterior: stores and releases hypothalamic hormones
- Blood supply vulnerability: Sheehan syndrome affects 30% of women with severe postpartum hemorrhage
- Tumor mass effects: visual field defects occur when adenomas exceed 10 mm (macroadenoma threshold)
-
Peripheral Gland Execution
- Thyroid: T4 half-life 7 days, T3 half-life 1 day (explains delayed symptom onset)
- Adrenal cortex: cortisol peak 6-8 AM (circadian rhythm, 50% higher than evening)
- Gonads: pulsatile GnRH required for normal LH/FSH secretion (continuous exposure suppresses)
- Pancreatic islets: insulin/glucagon ratio determines metabolic state
📌 Remember: FLAT PiG for anterior pituitary hormones FSH, LH, ACTH, TSH, Prolactin, iGF-1 (via GH) These six hormones control reproduction, stress response, metabolism, growth, and lactation-master them to predict all pituitary disorder presentations
Feedback Loop Mastery
- Negative Feedback Principles
- Long loop: peripheral hormone suppresses hypothalamus and pituitary
- Short loop: pituitary hormone suppresses hypothalamus
- Ultra-short loop: hypothalamic hormone self-regulates
- Set-point changes: pregnancy (10-fold hCG elevation), stress (2-5× cortisol), growth (3-fold GH pulses in puberty)
⭐ Clinical Pearl: Primary gland failure shows elevated tropic hormones (e.g., TSH >10 mIU/L in primary hypothyroidism), while secondary failure shows inappropriately normal or low tropic hormones despite low peripheral hormone-this distinction localizes the lesion in >95% of cases
| Axis | Hypothalamic Hormone | Pituitary Hormone | Target Gland | Peripheral Hormone | Feedback Threshold |
|---|---|---|---|---|---|
| Thyroid | TRH | TSH | Thyroid | T4, T3 | TSH >10 mIU/L (primary failure) |
| Adrenal | CRH | ACTH | Adrenal cortex | Cortisol | AM cortisol <5 mcg/dL (insufficiency) |
| Gonadal | GnRH | LH, FSH | Gonads | Estrogen, Testosterone | LH >20 IU/L (primary hypogonadism) |
| Growth | GHRH, Somatostatin | GH | Liver, tissues | IGF-1 | IGF-1 <-2 SD (GH deficiency) |
| Lactation | Dopamine (inhibits) | Prolactin | Breast | None | Prolactin >200 ng/mL (prolactinoma) |
💡 Master This: Every endocrine diagnosis depends on measuring both the peripheral hormone and its controlling tropic hormone simultaneously-this paired measurement localizes pathology to hypothalamus, pituitary, or target gland in a single test, predicting 90% of endocrine disorder classifications
- Hormone Transport Mechanisms
- Peptide hormones: water-soluble, cell surface receptors, rapid onset (minutes)
- Steroid hormones: lipid-soluble, intracellular receptors, delayed onset (hours to days)
- Thyroid hormones: 99.97% protein-bound (TBG, albumin), only free fraction active
- Binding protein changes: pregnancy, liver disease, nephrotic syndrome alter total but not free hormone
⭐ Clinical Pearl: Measuring free hormone levels (free T4, free testosterone) eliminates binding protein artifacts affecting 15-20% of total hormone measurements, particularly in pregnancy (TBG doubles), liver cirrhosis (↓ albumin), and nephrotic syndrome (urinary protein loss)
Hormone Receptor Dynamics
- Receptor Regulation Patterns
- Upregulation: chronic low hormone exposure increases receptor density (2-3× in hypothyroidism)
- Downregulation: chronic high exposure decreases receptors (50-70% reduction in insulin resistance)
- Desensitization: continued stimulation reduces response despite maintained receptors
- Sensitization: intermittent exposure enhances response (explains pulsatile GnRH requirement)
The endocrine command network integrates through receptor-mediated signaling cascades that amplify initial hormone binding into cellular responses. Connect this foundational control architecture through specific gland pathophysiology to understand disease mechanisms.
🎯 The Endocrine Command Network: Master Control Systems
🔥 Metabolic Mayhem: Diabetes and Obesity Mechanisms
Diabetes mellitus represents disordered glucose homeostasis from insulin deficiency (Type 1), insulin resistance (Type 2), or both-affecting 537 million adults globally (10.5% prevalence). Obesity drives metabolic syndrome through adipose tissue dysfunction, creating insulin resistance, dyslipidemia, hypertension, and prothrombotic states. Master these interconnected metabolic disasters to predict complications and guide treatment.

Glucose Homeostasis Breakdown
-
Type 1 Diabetes: Autoimmune Destruction
- Pancreatic β-cell destruction by T-cell mediated autoimmunity
- Genetic susceptibility: HLA-DR3/DR4 confers 40-50% of genetic risk
- Autoantibody markers: GAD65 (70-80%), IA-2 (60-70%), ZnT8 (60-70%), insulin autoantibodies (50-70%)
- Honeymoon phase: residual β-cell function persists 3-12 months, reducing insulin requirements
- C-peptide <0.6 ng/mL confirms absolute insulin deficiency
- Peak incidence ages 4-6 and 10-14 years (bimodal distribution)
- 10-15% present with diabetic ketoacidosis (DKA) at diagnosis
-
Type 2 Diabetes: Resistance and Exhaustion
- Insulin resistance in muscle, liver, adipose tissue precedes β-cell failure
- Compensatory hyperinsulinemia maintains euglycemia initially
- Progressive β-cell dysfunction: 50% function lost at diagnosis
- Relative insulin deficiency develops as demand exceeds secretory capacity
- Fasting glucose 100-125 mg/dL defines prediabetes (impaired fasting glucose)
- HbA1c 5.7-6.4% indicates prediabetes (5-10% annual progression to diabetes)
- 80-90% of Type 2 patients are overweight or obese (BMI >25 kg/m²)
📌 Remember: The 4 T's of Type 1 diabetes presentation Toilet (polyuria), Thirty (polydipsia), Tired (fatigue), Thinner (weight loss) Classic osmotic symptoms from glucosuria when blood glucose exceeds renal threshold (180 mg/dL)-present in 60-80% of new diagnoses
| Feature | Type 1 DM | Type 2 DM | Gestational DM |
|---|---|---|---|
| Pathophysiology | Autoimmune β-cell destruction | Insulin resistance + β-cell dysfunction | Pregnancy hormones → insulin resistance |
| Age of onset | Peak <20 years (bimodal) | Usually >40 years (younger trends) | 24-28 weeks gestation |
| Body habitus | Normal or thin | 80-90% overweight/obese | Variable |
| Autoantibodies | Present (GAD, IA-2, ZnT8) | Absent | Absent |
| C-peptide | Low/undetectable (<0.6 ng/mL) | Normal or elevated initially | Normal or elevated |
| Insulin requirement | Absolute, immediate | Progressive, eventual 50% | Resolves postpartum (90-95%) |
| DKA risk | High (10-15% at diagnosis) | Low (except stress) | Rare |
⭐ Clinical Pearl: Measuring C-peptide distinguishes Type 1 from Type 2 diabetes when phenotype is unclear-levels <0.6 ng/mL indicate absolute insulin deficiency requiring insulin therapy, while >1.0 ng/mL suggests preserved β-cell function responsive to oral agents, guiding treatment in 85-90% of ambiguous cases
Obesity and Metabolic Syndrome Pathways

-
Adipose Tissue Dysfunction
- Visceral fat: metabolically active, secretes inflammatory adipokines
- Adiponectin ↓ (30-50% reduction): protective hormone, enhances insulin sensitivity
- Leptin ↑ (2-3× elevation): satiety hormone, resistance develops in obesity
- TNF-α, IL-6 ↑: pro-inflammatory cytokines drive insulin resistance
- Free fatty acids ↑: impair insulin signaling in muscle and liver
- Ectopic fat deposition: liver (NAFLD in 70-90% of obese), pancreas, muscle
-
Metabolic Syndrome Criteria (ATP III)
- Requires ≥3 of 5 criteria for diagnosis
- Central obesity: waist circumference >102 cm (men), >88 cm (women)
- Triglycerides ≥150 mg/dL or treatment
- HDL <40 mg/dL (men), <50 mg/dL (women) or treatment
- Blood pressure ≥130/85 mmHg or treatment
- Fasting glucose ≥100 mg/dL or diabetes diagnosis
- Prevalence: 35% of US adults, 50% over age 60
- CVD risk: 2-fold increase, diabetes risk: 5-fold increase
💡 Master This: Metabolic syndrome represents a 5-fold increased risk for Type 2 diabetes and 2-fold risk for cardiovascular disease-aggressive lifestyle intervention (7% weight loss, 150 min/week exercise) reduces diabetes progression by 58% compared to 31% with metformin, making lifestyle the most effective prevention strategy
- Insulin Resistance Mechanisms
- Post-receptor signaling defects: IRS-1 phosphorylation impairment
- GLUT4 translocation failure: glucose uptake ↓ 50-70% in muscle
- Hepatic gluconeogenesis: inappropriately elevated despite hyperinsulinemia
- Pancreatic compensation: β-cell mass ↑ 50%, insulin secretion ↑ 2-3×
- Failure threshold: when secretion cannot overcome resistance
- Glucotoxicity: chronic hyperglycemia impairs β-cell function further
- Lipotoxicity: elevated free fatty acids accelerate β-cell apoptosis
⭐ Clinical Pearl: Acanthosis nigricans-velvety hyperpigmentation of neck, axillae, groin-indicates severe insulin resistance present in 75% of obese adolescents with Type 2 diabetes and 90% with metabolic syndrome, serving as a visible clinical marker for underlying metabolic dysfunction requiring screening
Diagnostic Thresholds and Testing
-
Diabetes Diagnostic Criteria (ADA)
- Fasting plasma glucose ≥126 mg/dL (after 8-hour fast) on 2 occasions
- Random glucose ≥200 mg/dL with classic symptoms
- 2-hour OGTT glucose ≥200 mg/dL (75 g glucose load)
- HbA1c ≥6.5% (reflects 3-month average glucose)
- Prediabetes: FPG 100-125 mg/dL, HbA1c 5.7-6.4%, 2-hr OGTT 140-199 mg/dL
- HbA1c limitations: hemoglobinopathies, anemia, hemolysis cause inaccuracy
-
Screening Recommendations
- Universal screening starting age 35 (previously 45, updated 2023)
- Earlier screening if overweight/obese (BMI ≥25 kg/m²) plus risk factor
- Risk factors: family history, high-risk ethnicity, gestational diabetes history, hypertension, dyslipidemia, PCOS, physical inactivity
- Repeat screening every 3 years if normal, 1 year if prediabetes
Metabolic dysfunction from diabetes and obesity creates cascading complications affecting every organ system. Connect these pathophysiologic mechanisms through specific complication patterns to build comprehensive management strategies.
🔥 Metabolic Mayhem: Diabetes and Obesity Mechanisms
🦴 Calcium Command: Parathyroid and Bone Metabolism
Calcium homeostasis maintains serum levels within narrow range (8.5-10.5 mg/dL) through parathyroid hormone (PTH), vitamin D, and calcitonin-essential for neuromuscular function, cardiac rhythm, bone mineralization, and cellular signaling. Disorders manifest as hypercalcemia (>10.5 mg/dL) or hypocalcemia (<8.5 mg/dL), creating neuromuscular, cardiac, and skeletal emergencies. Master the PTH-vitamin D axis to predict every calcium and bone disorder pattern.
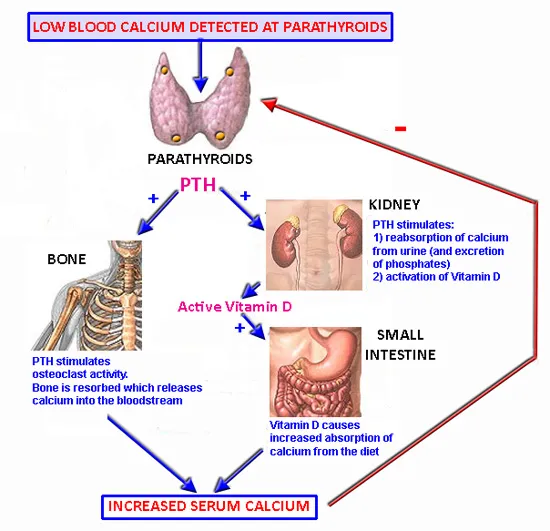
PTH-Vitamin D Axis Orchestration
-
Parathyroid Hormone: Calcium Defender
- Synthesized by chief cells in 4 parathyroid glands (total weight 120-140 mg)
- Calcium-sensing receptor (CaSR) detects ionized calcium changes of 0.1 mg/dL
- Secretion inversely proportional to serum calcium (↓ Ca²⁺ → ↑ PTH)
- Half-life 2-4 minutes enables rapid calcium adjustments
- Bone: stimulates osteoclast activity, releases Ca²⁺ and PO₄³⁻ (minutes to hours)
- Kidney: ↑ Ca²⁺ reabsorption (distal tubule), ↓ PO₄³⁻ reabsorption (proximal tubule)
- Kidney: activates 1α-hydroxylase converting 25-OH-D → 1,25-(OH)₂-D (calcitriol)
- Net effect: ↑ serum Ca²⁺, ↓ serum PO₄³⁻
-
Vitamin D: Calcium Amplifier
- Synthesized in skin (UV exposure) or dietary intake (D2, D3)
- Liver: 25-hydroxylation → 25-OH-D (calcidiol, storage form, half-life 2-3 weeks)
- Kidney: 1α-hydroxylation → 1,25-(OH)₂-D (calcitriol, active form, half-life 4-6 hours)
- Actions: ↑ intestinal Ca²⁺ absorption (2-3×), ↑ bone mineralization, ↑ renal Ca²⁺ reabsorption
- Vitamin D deficiency: 25-OH-D <20 ng/mL (affects 40-50% of adults)
- Insufficiency: 25-OH-D 20-30 ng/mL
- Optimal: 25-OH-D 30-50 ng/mL (>100 ng/mL indicates toxicity)
📌 Remember: PTH Does 3 Things to raise calcium Phosphaturia (renal PO₄ wasting), Tubular calcium reabsorption, Hydroxylation of vitamin D (1α-hydroxylase activation) These three actions coordinate to increase serum calcium while decreasing phosphate-inverse Ca-PO₄ relationship distinguishes hyperparathyroidism from other hypercalcemia causes
| Hormone | Source | Half-life | Primary Actions | Serum Ca Effect | Serum PO4 Effect |
|---|---|---|---|---|---|
| PTH | Parathyroid chief cells | 2-4 minutes | Bone resorption, renal Ca reabsorption, 1α-hydroxylase activation | ↑↑ | ↓ (phosphaturia) |
| Calcitriol (1,25-D) | Kidney (1α-hydroxylase) | 4-6 hours | Intestinal Ca/PO4 absorption, bone mineralization | ↑↑ | ↑ |
| Calcitonin | Thyroid C-cells | 10 minutes | Inhibits osteoclasts (weak effect) | ↓ (minimal) | ↓ (minimal) |
| FGF23 | Osteocytes | 45-60 minutes | Renal PO4 wasting, inhibits 1α-hydroxylase | ↓ (indirect) | ↓↓ |
⭐ Clinical Pearl: Primary hyperparathyroidism shows elevated or inappropriately normal PTH despite hypercalcemia (Ca >10.5 mg/dL)-this combination occurs in 85% of cases caused by parathyroid adenoma, distinguishing it from malignancy-related hypercalcemia where PTH is appropriately suppressed (<10 pg/mL)
Hypercalcemia: Mechanisms and Patterns

-
Primary Hyperparathyroidism (Most Common Outpatient)
- Parathyroid adenoma (80-85%), hyperplasia (15-20%), carcinoma (<1%)
- Prevalence: 1 in 500 adults, 3:1 female predominance, peak age 50-60 years
- Calcium: 10.5-12.0 mg/dL (mild elevation), PTH: elevated or inappropriately normal
- Phosphate: low-normal to low (<2.5 mg/dL in 50%)
- Complications: kidney stones (20%), osteoporosis (↓ BMD 10-15%), neurocognitive symptoms
- "Stones, bones, abdominal groans, psychiatric moans" (classic mnemonic)
- Surgical cure: parathyroidectomy indicated if Ca >11.0 mg/dL, age <50, osteoporosis, renal impairment
-
Malignancy-Associated Hypercalcemia (Most Common Inpatient)
- Humoral hypercalcemia: PTHrP secretion by solid tumors (80% of cancer hypercalcemia)
- Osteolytic metastases: local bone destruction (breast, lung, multiple myeloma)
- Calcium: >12.0 mg/dL (often >14 mg/dL, severe), PTH: suppressed (<10 pg/mL)
- PTHrP: elevated (>2.0 pmol/L), 1,25-D: low-normal
- Prognosis: median survival 30-60 days after hypercalcemia diagnosis
- Treatment urgency: Ca >14 mg/dL requires aggressive IV hydration, bisphosphonates
💡 Master This: Simultaneous measurement of calcium, PTH, and phosphate localizes hypercalcemia etiology in >90% of cases-high PTH indicates parathyroid pathology (adenoma, hyperplasia), while low PTH with high calcium points to malignancy, granulomatous disease, or vitamin D toxicity, guiding immediate diagnostic workup
- Vitamin D Toxicity and Granulomatous Disease
- Vitamin D toxicity: 25-OH-D >100 ng/mL from excessive supplementation
- Granulomatous disease: sarcoidosis, tuberculosis produce extra-renal 1α-hydroxylase
- Calcium: 11-13 mg/dL, PTH: suppressed, 1,25-D: elevated (>80 pg/mL)
- Mechanism: unregulated calcitriol production → ↑ intestinal Ca absorption
- Sarcoidosis: 10% develop hypercalcemia, 50% have elevated 1,25-D
- Treatment: glucocorticoids suppress extra-renal 1α-hydroxylase activity
Hypocalcemia: Deficiency and Resistance
-
Hypoparathyroidism: PTH Deficiency
- Surgical: post-thyroidectomy/parathyroidectomy (most common, 1-2% of thyroid surgeries)
- Autoimmune: isolated or part of APS-1 (with Addison's, mucocutaneous candidiasis)
- Genetic: DiGeorge syndrome (22q11 deletion), activating CaSR mutations
- Calcium: <8.5 mg/dL, PTH: low or undetectable, PO₄: elevated (>4.5 mg/dL)
- Symptoms: perioral paresthesias, carpopedal spasm (Trousseau sign), facial twitching (Chvostek sign)
- QT prolongation: corrected QT >460 ms in 60-70%, arrhythmia risk
- Treatment: calcitriol 0.25-2.0 mcg/day, calcium carbonate 1-3 g elemental Ca/day
-
Vitamin D Deficiency: Calcium Malabsorption
- 25-OH-D <20 ng/mL: insufficient intestinal calcium absorption
- Secondary hyperparathyroidism: PTH elevated (>65 pg/mL) compensating for low calcium
- Calcium: low-normal to low, PO₄: low (PTH-mediated phosphaturia)
- Alkaline phosphatase: elevated in severe deficiency (↑ bone turnover)
- Risk factors: limited sun exposure, dark skin, malabsorption, CKD
- Rickets/osteomalacia: inadequate bone mineralization, growth plate abnormalities
- Treatment: vitamin D3 50,000 IU weekly × 8 weeks, then maintenance 1000-2000 IU daily
⭐ Clinical Pearl: Chvostek sign (facial twitching with facial nerve tap) has 10-30% false-positive rate in normal individuals, while Trousseau sign (carpopedal spasm with BP cuff inflation >20 mmHg above systolic × 3 minutes) is 94% specific for hypocalcemia-always confirm clinical signs with laboratory calcium <8.5 mg/dL
- Pseudohypoparathyroidism: PTH Resistance
- Genetic defect: GNAS1 mutation → G-protein signaling impairment
- PTH elevated (resistance at kidney/bone), calcium low, PO₄ high
- Albright hereditary osteodystrophy: short stature, round face, short 4th/5th metacarpals, subcutaneous ossifications
- Type 1a: 50% also have TSH, FSH/LH resistance (multi-hormone resistance)
Connect calcium homeostasis through bone metabolism disorders to understand osteoporosis, osteomalacia, and Paget disease pathophysiology.
🦴 Calcium Command: Parathyroid and Bone Metabolism
⚡ Thyroid Turbulence: Hyper and Hypothyroid States
Thyroid hormones regulate basal metabolic rate, thermogenesis, cardiovascular function, and neurodevelopment through T4 (thyroxine) and T3 (triiodothyronine) secretion. Hyperthyroidism accelerates metabolism (↑ 60-100% BMR), while hypothyroidism slows every physiological process. Thyroid disorders affect 12% of the population during their lifetime, with 5:1 female predominance. Master TSH-thyroid hormone relationships to distinguish primary from secondary disorders and guide treatment.

Thyroid Hormone Production and Regulation
-
Thyroid Hormone Synthesis Steps
- Iodide uptake: sodium-iodide symporter (NIS) concentrates iodide 20-40× plasma levels
- Organification: thyroid peroxidase (TPO) oxidizes iodide, couples to thyroglobulin tyrosine residues
- Coupling: MIT + DIT → T3 (20%), DIT + DIT → T4 (80%)
- Storage: thyroglobulin-bound hormones stored in colloid (2-3 month supply)
- T4 secretion: 80-100 mcg/day, converted peripherally to T3 by deiodinases
- T3: 3-4× more potent than T4, 80% from peripheral T4 conversion
- Free hormone: 0.03% T4, 0.3% T3 unbound (biologically active fraction)
-
HPT Axis Feedback Control
- TRH (hypothalamus) → TSH (pituitary) → T4/T3 (thyroid)
- TSH: log-linear relationship with free T4 (10-fold TSH change per 2-fold T4 change)
- Normal TSH: 0.4-4.0 mIU/L (laboratory-specific ranges vary)
- T4 half-life: 7 days (delayed symptom onset/resolution)
- T3 half-life: 1 day (more rapid effects)
- Subclinical disease: abnormal TSH, normal free T4/T3
- Overt disease: abnormal TSH and free T4/T3
📌 Remember: TSH Tells The Story in thyroid diagnosis TSH Tells The Story: Primary hypothyroidism shows high TSH (>10 mIU/L) with low T4; primary hyperthyroidism shows low TSH (<0.1 mIU/L) with high T4/T3-TSH is the most sensitive screening test, changing before free hormone levels in 95% of thyroid disorders
| Test | Normal Range | Primary Hypothyroidism | Primary Hyperthyroidism | Secondary Hypothyroidism | Subclinical Hypothyroidism |
|---|---|---|---|---|---|
| TSH | 0.4-4.0 mIU/L | >10 mIU/L (↑↑) | <0.1 mIU/L (↓↓) | Low-normal or ↓ | 4.5-10 mIU/L (↑) |
| Free T4 | 0.8-1.8 ng/dL | <0.8 ng/dL (↓) | >1.8 ng/dL (↑) | <0.8 ng/dL (↓) | Normal |
| Free T3 | 2.3-4.2 pg/mL | Low-normal to ↓ | >4.2 pg/mL (↑↑) | Low-normal to ↓ | Normal |
| Anti-TPO | <35 IU/mL | Positive (>100) in 90% Hashimoto | Positive in 70% Graves | Negative | Positive if Hashimoto |
| TSI/TRAb | Negative | Negative | Positive (>140% in 95% Graves) | Negative | Negative |
⭐ Clinical Pearl: Free T4 measurement eliminates binding protein artifacts affecting total T4 in pregnancy (TBG doubles), estrogen therapy, nephrotic syndrome, and critical illness-free T4 reflects true thyroid status in >95% of cases, while total T4 misleads in 15-20% of hospitalized patients
Hyperthyroidism: Accelerated Metabolism
-
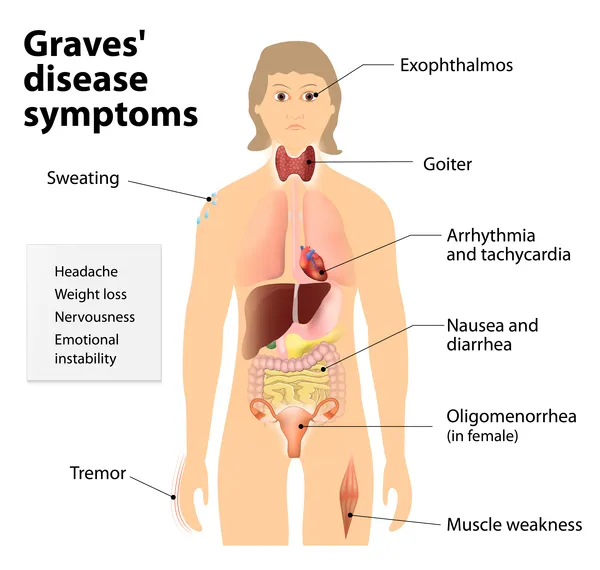
Graves Disease (Most Common, 60-80%)
- Autoimmune: TSH receptor antibodies (TRAb) stimulate thyroid
- TSI (thyroid-stimulating immunoglobulin): positive in 95% of Graves
- TSH: suppressed (<0.1 mIU/L), free T4/T3: elevated
- Unique features: diffuse goiter, ophthalmopathy (30-50%), pretibial myxedema (1-5%)
- Ophthalmopathy: proptosis, diplopia, periorbital edema (glycosaminoglycan deposition)
- Thyroid bruit: audible over gland in 50% (increased vascularity)
- Treatment: antithyroid drugs (methimazole 10-40 mg/day), radioactive iodine, thyroidectomy
-
Toxic Adenoma and Toxic Multinodular Goiter
- Autonomous thyroid nodule(s): TSH-independent hormone production
- TSH: suppressed, free T4/T3: elevated, radioiodine uptake: focal hot nodule(s)
- Age: typically >40 years (vs. Graves peak 20-40 years)
- TRAb: negative (distinguishes from Graves)
- Toxic adenoma: single autonomous nodule >3 cm
- Toxic MNG: multiple nodules with suppressed surrounding tissue
- Treatment: radioactive iodine or surgery (antithyroid drugs temporize only)
💡 Master This: Thyroid storm represents life-threatening hyperthyroidism with mortality 10-30%-triggered by surgery, infection, or iodine exposure in uncontrolled thyrotoxicosis, presenting with fever >40°C, tachycardia >140 bpm, altered mental status, and multiorgan dysfunction requiring immediate treatment with propylthiouracil 600-1000 mg loading, propranolol 60-80 mg q4h, hydrocortisone 100 mg q8h, and supportive care
- Thyroiditis-Induced Thyrotoxicosis
- Subacute (de Quervain): viral prodrome, painful thyroid, ↑ ESR (>50 mm/hr)
- Painless (lymphocytic): postpartum (5-10% of pregnancies), autoimmune
- Radioiodine uptake: low (<5%) distinguishes from Graves (uptake >25%)
- Natural history: thyrotoxic phase (1-3 months) → hypothyroid phase (4-6 months) → recovery
- Treatment: symptomatic with β-blockers (propranolol 20-40 mg q6h)
- Antithyroid drugs ineffective (no hormone synthesis occurring)
Hypothyroidism: Metabolic Slowdown
-
Hashimoto Thyroiditis (Most Common, 90%)
- Autoimmune: T-cell mediated destruction, anti-TPO antibodies (90%), anti-thyroglobulin (80%)
- TSH: elevated (>10 mIU/L in overt, 4.5-10 in subclinical)
- Free T4: low (<0.8 ng/dL) in overt, normal in subclinical
- Goiter: firm, non-tender, 20-50% of cases (atrophic variant has no goiter)
- Progression: 2-5%/year from subclinical to overt hypothyroidism
- Associated autoimmunity: Type 1 DM, Addison, celiac, vitiligo, pernicious anemia
- Treatment: levothyroxine 1.6 mcg/kg/day, titrate to TSH 0.4-4.0 mIU/L
-
Central (Secondary/Tertiary) Hypothyroidism
- Pituitary failure: TSH deficiency (secondary)
- Hypothalamic failure: TRH deficiency (tertiary)
- TSH: low-normal or inappropriately normal despite low free T4
- Free T4: low (<0.8 ng/dL), but TSH does not elevate appropriately
- Causes: pituitary adenoma, Sheehan syndrome, craniopharyngioma, TBI
- Screen other axes: ACTH, LH/FSH, GH deficiency commonly coexist
- Treatment: levothyroxine replacement (must treat adrenal insufficiency first to avoid crisis)
⭐ Clinical Pearl: Myxedema coma represents severe hypothyroidism with 30-60% mortality-presents with hypothermia <35°C, bradycardia <60 bpm, hyponatremia, hypoglycemia, and altered mental status, requiring IV levothyroxine 200-400 mcg loading then 50-100 mcg daily, hydrocortisone 100 mg q8h, and intensive supportive care
- Subclinical Thyroid Disease
- Subclinical hypothyroidism: TSH 4.5-10 mIU/L, normal free T4
- Treatment threshold: TSH >10 mIU/L or TSH 7-10 with symptoms/antibodies
- Progression risk: 2-5%/year to overt disease
- Subclinical hyperthyroidism: TSH <0.4 mIU/L, normal free T4/T3
- Atrial fibrillation risk: 3-fold increase with TSH <0.1 mIU/L
- Osteoporosis risk: 2-fold increase in postmenopausal women
- Treatment: consider if TSH <0.1 mIU/L, age >65, cardiac disease, osteoporosis
Thyroid dysfunction creates systemic metabolic derangements affecting cardiovascular, neurological, and reproductive systems. Connect these hormone imbalances through specific complications and treatment strategies.
⚡ Thyroid Turbulence: Hyper and Hypothyroid States
🔬 Adrenal Architecture: Cortex and Medulla Disorders
The adrenal glands produce cortisol (stress response, metabolism), aldosterone (sodium/volume homeostasis), androgens (secondary sex characteristics), and catecholamines (fight-or-flight response). Adrenal cortex disorders manifest as hormone excess (Cushing syndrome, primary aldosteronism) or deficiency (Addison disease), while medulla disorders produce catecholamine excess (pheochromocytoma). Understanding the 3-zone cortex and medulla architecture predicts all adrenal pathology patterns.

Adrenal Cortex: Three-
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more
Have doubts about this lesson?
Ask Rezzy, your AI Study Mate, to explain anything you didn't understand
Everything you need for NEET-PG prep
Get full Oncourse access with lessons, practice questions, flashcards and AI study tools.
Scan to download app




















