Metabolic Bone Diseases

On this page
🦴 The Metabolic Bone Disease Spectrum: Decoding Skeletal Pathology
Your skeleton is not static-it's a dynamic organ constantly rebuilding itself through precise metabolic choreography, and when that balance fails, the consequences range from silent fractures to dramatic deformities. You'll master the pathophysiology behind osteoporosis, osteomalacia, Paget's disease, and the complex bone disruptions triggered by parathyroid and renal dysfunction. By connecting calcium regulation, vitamin D metabolism, and hormonal control to their skeletal manifestations, you'll develop the clinical reasoning to recognize, investigate, and manage these diseases before they break your patients. This is your blueprint for protecting the framework that protects us all.
📌 Remember: VITAMIN - Vitamin D deficiency, Increased PTH, Tumor (malignancy), Aging, Malabsorption, Immobilization, Nutritional deficiency. These seven factors account for 85% of metabolic bone disease presentations in clinical practice.
The pathophysiology centers on three critical systems: the parathyroid-vitamin D axis, bone remodeling cycle, and mineral homeostasis. Normal bone turnover requires 120 days for complete osteoclast-osteoblast cycling, with 10% of the skeleton replaced annually. Disruption at any level cascades into measurable bone loss, with T-scores below -2.5 defining osteoporosis and fracture risk increasing 2-fold per standard deviation decrease.
| Disease Category | Primary Defect | Bone Turnover | Mineralization | Fracture Risk | Treatment Response |
|---|---|---|---|---|---|
| Osteoporosis | Bone Mass Loss | High/Normal | Normal | 2-4x increased | 60-80% reduction |
| Osteomalacia | Mineralization | Low/Normal | Impaired | 3-5x increased | 90% with vitamin D |
| Paget's Disease | Remodeling | Very High | Disorganized | 2-3x increased | 85% with bisphosphonates |
| Hyperparathyroidism | PTH Excess | High | Variable | 2-4x increased | 95% surgical cure |
| Renal Osteodystrophy | CKD-MBD | Variable | Impaired | 4-6x increased | 70% with management |
- Primary Prevention Targets
- Vitamin D sufficiency: ≥30 ng/mL (75 nmol/L)
- Calcium intake: 1200 mg daily for adults >50 years
- Weight-bearing exercise: 150 minutes weekly minimum
- Resistance training: 2-3 sessions weekly
- Impact activities: 30 minutes daily for bone stimulation
- Balance training: 15 minutes daily for fall prevention
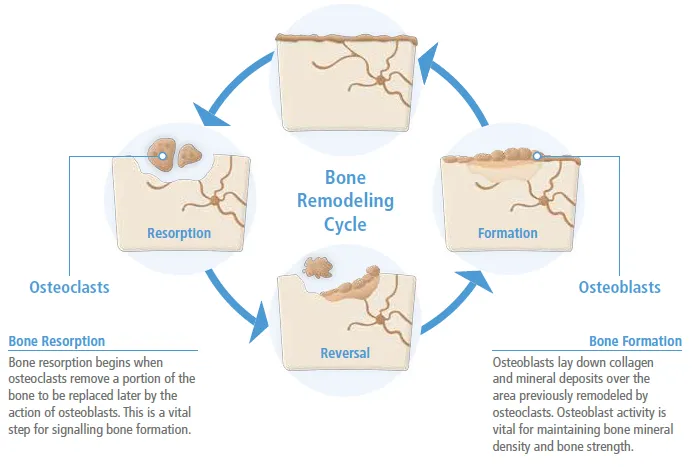
💡 Master This: The RANK-RANKL-OPG pathway controls bone remodeling, with RANKL promoting osteoclast formation and OPG providing inhibition. This 3:1 ratio determines net bone formation versus resorption, making it the primary therapeutic target for 75% of bone-active medications.
Laboratory assessment requires systematic evaluation of bone turnover markers, mineral metabolism, and hormonal status. Serum CTX (C-terminal telopeptide) reflects bone resorption with normal values <400 pg/mL in premenopausal women, while P1NP (procollagen type 1 N-propeptide) indicates formation with reference ranges 15-75 ng/mL. These markers change 30-50% within 3-6 months of effective treatment, providing early therapeutic monitoring.
⭐ Clinical Pearl: Secondary osteoporosis accounts for 30% of postmenopausal and 50% of premenopausal cases. The most common causes include glucocorticoid use (>7.5 mg prednisone daily for >3 months), hyperparathyroidism, and malabsorption syndromes.
Understanding metabolic bone diseases transforms clinical practice by revealing the interconnected nature of skeletal pathology, setting the foundation for exploring specific disease mechanisms and their therapeutic implications.
🦴 The Metabolic Bone Disease Spectrum: Decoding Skeletal Pathology
⚡ Osteoporosis Command Center: The Silent Epidemic Architecture
📌 Remember: SHATTERED - Steroids, Hyperthyroidism, Alcohol, Thin build, Testosterone low, Early menopause, Renal disease, Erosive arthritis, Dietary calcium deficiency. These risk factors account for 80% of osteoporotic fractures in clinical practice.
The pathophysiology involves estrogen deficiency, aging, and secondary factors disrupting the RANK-RANKL-OPG signaling pathway. Postmenopausal estrogen loss increases RANKL expression by 300%, promoting osteoclast formation and bone resorption. Simultaneously, osteoblast function declines 2-3% annually, creating negative bone balance of -1 to -3% yearly during the first 5-10 years post-menopause.
| Fracture Site | Lifetime Risk (Women) | Lifetime Risk (Men) | Mortality Impact | Healthcare Cost | Recovery Time |
|---|---|---|---|---|---|
| Hip | 17.5% | 6.0% | 20% at 1 year | $40,000 per fracture | 6-12 months |
| Vertebral | 15.6% | 5.0% | 23% increased mortality | $9,000 per fracture | 4-6 months |
| Wrist | 16.0% | 2.5% | Minimal | $5,000 per fracture | 6-8 weeks |
| Any Major | 39.7% | 13.1% | Variable | $19 billion annually | Variable |
- FRAX Risk Assessment Framework
- 10-year major fracture probability ≥20% indicates treatment
- 10-year hip fracture probability ≥3% warrants intervention
- Risk factors amplifying FRAX scores:
- Previous fracture: Increases risk 1.5-2.0 fold
- Parental hip fracture: Adds 1.4-fold risk
- Current smoking: Increases risk 1.3-fold
- Alcohol ≥3 units daily: Adds 1.2-fold risk
- Rheumatoid arthritis: Increases risk 1.4-fold

💡 Master This: Trabecular bone (spine, hip) turns over 8 times faster than cortical bone, explaining why vertebral fractures occur 5-10 years earlier than hip fractures. This differential turnover rate determines both fracture timing and treatment response patterns.
Secondary osteoporosis evaluation requires comprehensive screening when T-scores are unexpectedly low or fractures occur at young age. Essential laboratory assessment includes 25(OH)D, PTH, TSH, serum protein electrophoresis, 24-hour urine calcium, and testosterone (men). Glucocorticoid-induced osteoporosis develops within 3-6 months of therapy, with fracture risk increasing 75% at the spine and 18% at the hip.
⭐ Clinical Pearl: Bone turnover markers predict treatment response within 3-6 months. CTX reduction ≥60% with antiresorptive therapy and P1NP increase ≥20% with anabolic agents indicate therapeutic efficacy before BMD changes become apparent.
Treatment selection depends on fracture risk, bone turnover status, and patient factors. Bisphosphonates remain first-line therapy, reducing vertebral fractures by 40-70% and hip fractures by 40-50%. Denosumab provides similar efficacy with 6-month dosing, while teriparatide offers anabolic effects for severe osteoporosis, increasing BMD by 9-13% at the spine over 18 months.
This comprehensive understanding of osteoporosis mechanisms enables precise risk stratification and targeted therapeutic interventions, connecting to the broader spectrum of metabolic bone diseases through shared pathophysiological pathways.
⚡ Osteoporosis Command Center: The Silent Epidemic Architecture
🔬 Osteomalacia & Rickets: The Mineralization Malfunction Matrix
Osteomalacia and rickets represent the same pathophysiological process affecting closed versus open growth plates, respectively. Both conditions result from inadequate mineralization of bone matrix, creating excess unmineralized osteoid that comprises >12% of bone volume (normal <5%). The global prevalence reaches 40% in some populations, with vitamin D deficiency accounting for 85% of cases worldwide.
📌 Remember: VITAMIN D - Vascular disease, Inadequate sunlight, Tumor (malignancy), Aging, Malabsorption, Immobilization, Nutritional deficiency, Drugs (anticonvulsants). These factors create the clinical spectrum of vitamin D deficiency affecting 1 billion people globally.
The pathophysiology centers on disrupted calcium homeostasis through three primary mechanisms: vitamin D deficiency, phosphate depletion, and mineralization inhibitors. Normal mineralization requires adequate calcium (>10 mg/dL), phosphate (>3.0 mg/dL), and alkaline phosphatase activity. When 25(OH)D falls below 20 ng/mL, intestinal calcium absorption decreases from 30-40% to 10-15%, triggering compensatory PTH elevation and phosphate wasting.
| Parameter | Normal Values | Osteomalacia | Rickets | Severe Deficiency |
|---|---|---|---|---|
| 25(OH)D | 30-100 ng/mL | <20 ng/mL | <10 ng/mL | <5 ng/mL |
| PTH | 15-65 pg/mL | 65-150 pg/mL | 100-300 pg/mL | >300 pg/mL |
| Alkaline Phosphatase | 44-147 IU/L | 150-400 IU/L | 200-800 IU/L | >800 IU/L |
| 24h Urine Calcium | 100-300 mg | <50 mg | <25 mg | <10 mg |
| Osteoid Volume | <5% | 12-25% | 15-35% | >35% |
- Clinical Manifestations by System
- Skeletal symptoms (present in 90% of cases):
- Bone pain: Diffuse, aching, worse with movement
- Muscle weakness: Proximal myopathy in 70%
- Fractures: Low-trauma, delayed healing
- Rickets-specific deformities:
- Craniotabes: Soft skull bones in infants
- Delayed fontanelle closure: Beyond 18 months
- Dental problems: Delayed eruption, increased caries
- Growth retardation: Height <3rd percentile
- Biochemical abnormalities:
- Hypocalcemia: Serum calcium <8.5 mg/dL
- Hypophosphatemia: Phosphate <2.5 mg/dL
- Elevated PTH: >65 pg/mL in 95% of cases
- Skeletal symptoms (present in 90% of cases):

💡 Master This: The calcium-phosphate product must exceed 30 mg²/dL² for normal mineralization. When this product falls below 25, mineralization defects occur, explaining why both hypocalcemia and hypophosphatemia can cause osteomalacia through different mechanisms.
Differential diagnosis requires distinguishing osteomalacia from osteoporosis, hyperparathyroidism, and malignancy. Key discriminators include elevated alkaline phosphatase (present in 85% of osteomalacia), low 25(OH)D, and characteristic radiographic changes. Bone biopsy remains the gold standard, showing osteoid seam width >12 μm and mineralization lag time >100 days (normal <20 days).
| Etiology Category | Specific Causes | Prevalence | Key Laboratory Findings | Treatment Response |
|---|---|---|---|---|
| Vitamin D Deficiency | Dietary, sunlight, malabsorption | 85% | Low 25(OH)D, high PTH | 90% cure rate |
| Phosphate Depletion | Renal wasting, antacids | 10% | Low phosphate, normal 25(OH)D | 80% improvement |
| Mineralization Defects | Bisphosphonates, fluoride | 3% | Normal minerals, high ALP | 60% improvement |
| Genetic Disorders | Hypophosphatasia, vitamin D resistance | 2% | Variable patterns | Variable response |
Treatment protocols depend on severity and underlying etiology. Standard vitamin D deficiency responds to 50,000 IU weekly for 8 weeks, followed by maintenance dosing of 1,000-2,000 IU daily. Severe deficiency may require loading doses of 300,000-600,000 IU over 1-2 weeks. Phosphate replacement uses 1-3 grams daily in divided doses, while calcium supplementation provides 1,200-1,500 mg daily.

This mechanistic understanding of mineralization defects provides the foundation for exploring more complex metabolic bone diseases where similar pathways become disrupted through different mechanisms.
🔬 Osteomalacia & Rickets: The Mineralization Malfunction Matrix
🎯 Paget's Disease: The Bone Remodeling Rebellion
📌 Remember: PAGET'S - Pain (bone), Alkaline phosphatase elevated, Giant osteoclasts, Enlarged bones, Thick cortices, Sclerotic appearance. These features define the classic presentation affecting 3-4% of the population over age 40 in endemic areas.
The pathophysiology begins with abnormal osteoclasts containing viral-like inclusions and increased nuclei (up to 100 versus normal 3-5). These giant osteoclasts create excessive bone resorption, followed by compensatory osteoblast activation producing disorganized collagen with woven bone pattern. The result is mechanically inferior bone with increased fracture risk despite normal or increased bone density.
| Disease Phase | Osteoclast Activity | Osteoblast Activity | Radiographic Appearance | Alkaline Phosphatase | Symptoms |
|---|---|---|---|---|---|
| Lytic | Very High | Low | Osteoporosis circumscripta | 5-10x elevated | Severe pain |
| Mixed | High | High | Mixed lytic-sclerotic | 10-25x elevated | Moderate pain |
| Sclerotic | Low | Moderate | Dense, enlarged bone | 2-5x elevated | Mild pain |
| Inactive | Normal | Normal | Residual deformity | Normal | Minimal pain |
- Clinical Manifestations by System
- Skeletal complications (present in 70% of symptomatic cases):
- Bone pain: Deep, aching, worse at night
- Deformity: Bowing of long bones, skull enlargement
- Fractures: Chalk-stick fractures, delayed healing
- Arthritis: Secondary to joint deformity
- Neurological complications (15-20% of cases):
- Hearing loss: Conductive or sensorineural
- Spinal stenosis: From vertebral enlargement
- Cranial nerve compression: Multiple nerves affected
- Cardiovascular complications (5-10% of extensive disease):
- High-output heart failure: From increased vascularity
- Calcific aortic stenosis: Accelerated calcification
- Skeletal complications (present in 70% of symptomatic cases):

💡 Master This: Sarcomatous transformation occurs in <1% of patients but represents the most feared complication. Risk factors include polyostotic disease, long duration, and previous radiation. Sudden pain increase or soft tissue mass warrants immediate MRI and biopsy.
Diagnostic criteria require characteristic radiographic changes plus elevated alkaline phosphatase or positive bone scan. Bone-specific alkaline phosphatase provides greater specificity than total alkaline phosphatase, while urinary NTX or CTX reflects resorption activity. Bone biopsy shows pathognomonic mosaic pattern with irregular cement lines but is rarely necessary.
| Complication | Frequency | Risk Factors | Clinical Presentation | Management Approach |
|---|---|---|---|---|
| Fractures | 10-15% | Long bone involvement | Chalk-stick pattern | Prophylactic fixation |
| Hearing Loss | 30-50% | Skull involvement | Progressive deafness | Hearing aids, surgery |
| Heart Failure | 5-10% | Extensive disease | High-output failure | Bisphosphonates, diuretics |
| Sarcoma | <1% | Polyostotic, duration | Pain increase, mass | Amputation, chemotherapy |
| Spinal Stenosis | 10-20% | Vertebral involvement | Neurogenic claudication | Decompression surgery |
Bisphosphonate therapy remains the gold standard, with zoledronic acid 5 mg IV providing sustained remission for 2-5 years in 90% of patients. Oral bisphosphonates require higher doses than osteoporosis treatment: risedronate 30 mg daily for 2 months or alendronate 40 mg daily for 6 months. Treatment goals include symptom relief, alkaline phosphatase normalization, and complication prevention.

This understanding of Paget's disease pathophysiology illuminates how localized bone remodeling disorders can create systemic complications, connecting to broader concepts of bone turnover regulation and therapeutic intervention strategies.
🎯 Paget's Disease: The Bone Remodeling Rebellion
⚖️ Hyperparathyroidism: The Calcium Command Crisis
📌 Remember: STONES - Skeletal disease, Thirty (psychiatric symptoms), Osteitis fibrosa, Nephrolithiasis, Elevated calcium, Serum PTH high. This classic mnemonic captures the multisystem effects of chronic hypercalcemia affecting cardiovascular, renal, skeletal, and neurological systems.
The pathophysiology involves autonomous PTH secretion from parathyroid adenomas (80%), hyperplasia (15%), or carcinoma (<5%). PTH excess increases renal calcium reabsorption, intestinal calcium absorption via 1,25(OH)₂D, and bone resorption through RANKL upregulation. The result is hypercalcemia (>10.5 mg/dL), hypercalciuria (>400 mg/24h), and accelerated bone turnover.
| Parameter | Normal Range | Mild PHPT | Moderate PHPT | Severe PHPT | Crisis Level |
|---|---|---|---|---|---|
| Serum Calcium | 8.5-10.5 mg/dL | 10.6-11.0 | 11.1-12.0 | 12.1-14.0 | >14.0 |
| PTH | 15-65 pg/mL | 65-150 | 150-300 | 300-800 | >800 |
| 24h Urine Calcium | 100-300 mg | 300-400 | 400-600 | >600 | >800 |
| Bone Turnover | Normal | 1.5x elevated | 2-3x elevated | >3x elevated | >5x elevated |
| Fracture Risk | Baseline | 1.5x increased | 2-3x increased | >3x increased | Acute risk |
- Clinical Manifestations by System
- Skeletal effects (present in 80% of cases):
- Osteoporosis: Preferentially affects cortical bone
- Fractures: 2-3 fold increased risk, especially wrist
- Bone pain: Diffuse, aching, worse with activity
- Arthritis: Secondary to chondrocalcinosis
- Renal complications (15-20% of patients):
- Nephrolithiasis: Calcium oxalate or calcium phosphate
- Nephrocalcinosis: Medullary calcification
- Chronic kidney disease: From hypercalciuria
- Neuropsychiatric symptoms (50-70% of cases):
- Depression: Present in 50% of patients
- Cognitive impairment: Memory and concentration
- Anxiety: Panic attacks in severe cases
- Fatigue: Weakness and lethargy
- Skeletal effects (present in 80% of cases):
💡 Master This: PTH acts differently on cortical versus trabecular bone. Cortical bone (radius, hip) shows preferential loss with 2-3% annual decline, while trabecular bone (spine) may be preserved or even increased due to anabolic PTH effects at intermittent exposure.
Diagnostic approach requires confirmed hypercalcemia on multiple measurements plus elevated or inappropriately normal PTH. Ionized calcium provides greater accuracy than albumin-corrected total calcium. 25(OH)D deficiency must be corrected before PTH interpretation, as vitamin D levels <20 ng/mL can mask or exaggerate hyperparathyroidism.
| Surgical Indication | Criteria | Evidence Level | Cure Rate | Complication Risk |
|---|---|---|---|---|
| Symptomatic Disease | Stones, bones, psychiatric | Class I | 95-98% | <2% |
| Age <50 years | Asymptomatic patients | Class IIa | 95-98% | <2% |
| Severe Hypercalcemia | >11.5 mg/dL | Class I | 95-98% | <2% |
| Osteoporosis | T-score ≤-2.5 any site | Class IIa | 95-98% | <2% |
| Renal Dysfunction | eGFR <60 mL/min | Class IIa | 95-98% | <2% |
Medical management is reserved for poor surgical candidates or patient preference. Cinacalcet reduces serum calcium by 0.5-1.0 mg/dL through calcium-sensing receptor activation. Bisphosphonates improve bone density by 3-5% but don't address hypercalcemia. Adequate hydration and loop diuretics provide acute management for hypercalcemic crisis.

This comprehensive understanding of hyperparathyroidism reveals how hormonal dysregulation creates multisystem disease, connecting calcium homeostasis to the broader spectrum of metabolic bone diseases and their therapeutic management strategies.
⚖️ Hyperparathyroidism: The Calcium Command Crisis
🔗 Renal Osteodystrophy: The Kidney-Bone Connection Catastrophe

Renal osteodystrophy encompasses the spectrum of bone diseases associated with chronic kidney disease-mineral bone disorder (CKD-MBD), affecting bone turnover, mineralization, and volume. The condition develops progressively as GFR declines, with biochemical abnormalities appearing at stage 3 CKD (GFR 30-59) and bone disease becoming universal by stage 5 (GFR <15). Fracture risk increases 2-4 fold compared to age-matched controls.
📌 Remember: KIDNEY - Kalium (potassium) retention, Iron deficiency, Decreased 1,25(OH)₂D, No phosphate excretion, Elevated PTH, Yearning for calcium. These interconnected abnormalities create the complex pathophysiology of CKD-MBD affecting cardiovascular and skeletal systems simultaneously.
The pathophysiology involves multiple interconnected mechanisms beginning with impaired phosphate excretion and reduced 1α-hydroxylase activity. Phosphate retention stimulates FGF23 secretion, which inhibits 1,25(OH)₂D production and increases phosphate excretion. Declining 1,25(OH)₂D reduces intestinal calcium absorption, triggering secondary hyperparathyroidism and progressive bone disease.
| CKD Stage | GFR Range | PTH Target | Phosphate Target | Calcium Target | Bone Disease Prevalence |
|---|---|---|---|---|---|
| Stage 3 | 30-59 | 35-70 pg/mL | 2.7-4.6 mg/dL | 8.4-9.5 mg/dL | 25% |
| Stage 4 | 15-29 | 70-110 pg/mL | 2.7-4.6 mg/dL | 8.4-9.5 mg/dL | 50% |
| Stage 5 | <15 | 150-300 pg/mL | 3.5-5.5 mg/dL | 8.4-9.5 mg/dL | 90% |
| Dialysis | <15 | 150-300 pg/mL | 3.5-5.5 mg/dL | 8.4-9.5 mg/dL | 95% |
- Bone Disease Classification (TMV System)
- Turnover assessment (based on bone formation rate):
- High turnover: >95th percentile normal
- Normal turnover: 5th-95th percentile
- Low turnover: <5th percentile normal
- Mineralization evaluation (based on osteoid maturation):
- Normal mineralization: <20 days lag time
- Abnormal mineralization: >20 days lag time
- Volume determination (based on bone volume/tissue volume):
- Normal volume: 15-25% trabecular bone
- High volume: >25% trabecular bone
- Low volume: <15% trabecular bone
- Turnover assessment (based on bone formation rate):
💡 Master This: FGF23 serves as the earliest biomarker of CKD-MBD, rising 100-1000 fold above normal in stage 5 CKD. This phosphaturic hormone creates a vicious cycle by suppressing 1,25(OH)₂D production while promoting phosphate excretion, ultimately contributing to cardiovascular calcification and mortality.
Diagnostic approach requires comprehensive biochemical assessment including PTH, 25(OH)D, 1,25(OH)₂D, alkaline phosphatase, bone-specific alkaline phosphatase, and FGF23. Bone biopsy remains the gold standard for definitive diagnosis but is rarely performed due to invasive nature. Bone turnover markers provide surrogate assessment with PTH levels guiding therapeutic decisions.
| Bone Disease Type | PTH Level | Alkaline Phosphatase | Bone Formation Rate | Fracture Risk | Treatment Approach |
|---|---|---|---|---|---|
| Osteitis Fibrosa | >300 pg/mL | >150 IU/L | High | Moderate | Phosphate binders, calcimimetics |
| Adynamic Bone | <100 pg/mL | <100 IU/L | Low | High | Reduce calcium, vitamin D analogs |
| Mixed Disease | 150-300 pg/mL | 100-150 IU/L | Variable | High | Individualized therapy |
| Osteomalacia | Variable | >150 IU/L | Low-Normal | Very High | Vitamin D, phosphate |
Treatment strategies focus on preventing progression and managing complications through phosphate control, vitamin D therapy, and PTH modulation. Phosphate binders (calcium-based, sevelamer, lanthanum) target phosphate levels 3.5-5.5 mg/dL. Active vitamin D analogs (calcitriol, paricalcitol) suppress PTH while calcimimetics (cinacalcet) provide PTH control without increasing calcium-phosphate product.
This comprehensive understanding of renal osteodystrophy demonstrates how organ system failure creates complex metabolic bone disease, requiring multifaceted therapeutic approaches that address mineral metabolism, bone health, and cardiovascular protection simultaneously.
🔗 Renal Osteodystrophy: The Kidney-Bone Connection Catastrophe
🎯 Metabolic Bone Disease Mastery: The Clinical Command Arsenal
📌 Remember: METABOLIC - Mineralization defects, Endocrine disorders, Turnover abnormalities, Aging effects, Bone formation defects, Osteoclast dysfunction, Lifestyle factors, Iatrogenic causes, Chronic diseases. This framework encompasses 95% of metabolic bone disease presentations in clinical practice.
- Essential Clinical Arsenal
- Rapid Assessment Tools:
- FRAX calculator: 10-year fracture probability
- T-score interpretation: -1.0 to -2.5 (osteopenia), ≤-2.5 (osteoporosis)
- Bone turnover markers: CTX (resorption), P1NP (formation)
- Vitamin D status: ≥30 ng/mL optimal, <20 ng/mL deficient
- Critical Laboratory Thresholds:
- PTH elevation: >65 pg/mL suggests secondary hyperparathyroidism
- Alkaline phosphatase: >150 IU/L indicates increased bone turnover
- 25(OH)D deficiency: <20 ng/mL affects 40% of adults globally
- Calcium-phosphate product: >55 mg²/dL² increases calcification risk
- Rapid Assessment Tools:
| Disease Pattern | Key Discriminator | Diagnostic Threshold | Treatment Response | Monitoring Parameter |
|---|---|---|---|---|
| Osteoporosis | Low BMD | T-score ≤-2.5 | BMD increase 3-8% | Annual DXA |
| Osteomalacia | High ALP + Low 25(OH)D | ALP >150 + 25(OH)D <20 | ALP normalize 3-6 months | Quarterly labs |
| Paget's Disease | Very High ALP | ALP >300 IU/L | ALP decrease >75% | Every 6 months |
| Hyperparathyroidism | High Ca + High PTH | Ca >10.5 + PTH >65 | Ca normalize post-surgery | Post-op monitoring |
| Renal Osteodystrophy | High PTH + Low GFR | PTH >150 + GFR <60 | PTH target by CKD stage | Monthly labs |
💡 Master This: Bone turnover markers change 30-50% within 3-6 months of effective treatment, providing early therapeutic monitoring before BMD changes become apparent. CTX reduction ≥60% with antiresorptive therapy and P1NP increase ≥20% with anabolic agents indicate therapeutic efficacy.
Treatment Selection Matrix requires matching therapeutic mechanism to underlying pathophysiology. Antiresorptive agents (bisphosphonates, denosumab) work best for high-turnover osteoporosis, while anabolic agents (teriparatide, abaloparatide) excel in severe osteoporosis with low formation. Combination therapy may benefit complex cases with multiple pathologies.
⭐ Clinical Pearl: Drug holidays from bisphosphonates should be considered after 3-5 years of therapy in low-risk patients with stable BMD and no recent fractures. High-risk patients (T-score ≤-2.5, previous fractures, age >75) should continue therapy with annual reassessment.
This comprehensive mastery framework transforms complex metabolic bone diseases into manageable clinical entities through systematic assessment, targeted therapy, and evidence-based monitoring, enabling optimal patient outcomes across the entire spectrum of bone metabolic disorders.
🎯 Metabolic Bone Disease Mastery: The Clinical Command Arsenal
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more
Have doubts about this lesson?
Ask Rezzy, your AI Study Mate, to explain anything you didn't understand
Everything you need for NEET-PG prep
Get full Oncourse access with lessons, practice questions, flashcards and AI study tools.
Scan to download app
























