Basic Science in Orthopaedics

On this page
🏗️ Bone Composition & Cells - Microscopic Marvels
Bone isn't static scaffolding-it's a living organ that constantly remodels itself in response to mechanical forces, hormonal signals, and metabolic demands. You'll explore how specialized cells orchestrate mineralization and resorption, how trabecular and cortical architecture distribute stress, and why blood supply dictates healing potential. Understanding these foundational principles transforms how you diagnose fractures, plan surgical approaches, and predict complications. Master the basic science, and orthopaedic pathology becomes intuitive rather than memorized.
Bone Matrix Architecture: The Foundation Framework
Bone matrix consists of 65% inorganic mineral content and 35% organic components, creating a composite material with unique mechanical properties. The inorganic phase primarily contains hydroxyapatite crystals [Ca₁₀(PO₄)₆(OH)₂], providing compressive strength of 170 MPa-comparable to concrete. The organic matrix, predominantly Type I collagen (90% of organic content), contributes tensile strength and flexibility.
-
Inorganic Components
- Hydroxyapatite crystals: 65% of bone weight
- Calcium phosphate: 85% of mineral content
- Carbonate substitutions: 4-8% of crystal structure
- Magnesium incorporation: 0.5-1.0%
- Fluoride content: 0.01-0.05%
- Trace elements: Strontium, zinc, copper
-
Organic Matrix Elements
- Type I collagen: 90% of organic matrix
- Non-collagenous proteins: 10% of organic content
- Osteocalcin: 15-20% of non-collagenous proteins
- Osteopontin: Mineralization regulation
- Bone sialoprotein: Cell attachment sites
📌 Remember: CHAMP for bone matrix components - Calcium phosphate, Hydroxyapatite, Alkaline phosphatase, Magnesium, Proteoglycans. Hydroxyapatite provides 65% of bone's compressive strength while collagen contributes 35% of tensile properties.
| Component | Percentage | Function | Mechanical Property | Clinical Significance |
|---|---|---|---|---|
| Hydroxyapatite | 65% | Compressive strength | 170 MPa compression | Osteoporosis target |
| Type I Collagen | 25% | Tensile strength | 150 MPa tension | Osteogenesis imperfecta |
| Water | 5-10% | Viscoelasticity | Shock absorption | Dehydration effects |
| Non-collagenous proteins | 3-5% | Mineralization control | Interface bonding | Biomarker source |
| Cells | <2% | Remodeling regulation | Adaptive response | Therapeutic targets |
Cellular Orchestration: The Bone Remodeling Team
Bone cells form a sophisticated communication network that maintains skeletal homeostasis through coordinated remodeling cycles lasting 120-200 days. Each cell type performs specialized functions while responding to mechanical, hormonal, and metabolic signals.
Osteoblast Mastery: The Bone Builders
- Origin: Mesenchymal stem cells via Runx2 transcription factor
- Function: Matrix synthesis at 1-2 μm/day rate
- Lifespan: 10-20 days active phase
- Key Products: Type I collagen, osteocalcin, alkaline phosphatase
- Alkaline phosphatase levels: 50-120 U/L (formation marker)
- Osteocalcin production: 15-50 ng/mL (bone-specific marker)
Osteoclast Dynamics: The Bone Resorbers
- Origin: Hematopoietic stem cells (monocyte-macrophage lineage)
- Resorption Rate: 25-40 μm³/day per cell
- Lifespan: 2-3 weeks active resorption
- Mechanism: Acid secretion (pH 4.5) dissolves mineral
- Cathepsin K enzyme: Collagen degradation
- TRAP activity: 2.5-6.5 U/L (resorption marker)
💡 Master This: The RANK/RANKL/OPG pathway controls osteoclast formation-RANKL (from osteoblasts) activates osteoclasts, while OPG (osteoprotegerin) blocks this activation. RANKL:OPG ratio >1.0 promotes bone resorption, explaining postmenopausal bone loss when estrogen-mediated OPG production decreases by 40-60%.
Osteocyte Network: The Mechanosensors
- Population: 95% of all bone cells
- Density: 20,000-25,000 cells/mm³ in cortical bone
- Dendrite Network: 50-80 processes per cell
- Mechanosensitivity: Responds to <10 microstrain deformation
- Sclerostin regulation: Mechanical loading inhibitor
- Lacunar-canalicular system: 12-15% of bone volume
⭐ Clinical Pearl: Osteocyte apoptosis triggers microdamage repair within 7-14 days. Bisphosphonates can impair this process by reducing osteocyte viability by 30-40%, contributing to atypical fracture risk with prolonged use >5 years.
Understanding bone's cellular architecture connects directly to biomechanical principles, where these microscopic interactions determine macroscopic skeletal strength and adaptive capacity.
🏗️ Bone Composition & Cells — Microscopic Marvels
⚙️ Bone Formation & Remodeling - Dynamic Development

Intramembranous Ossification: Direct Bone Formation
Intramembranous ossification creates flat bones through direct mesenchymal cell differentiation, bypassing the cartilage template phase. This process occurs in cranial bones, clavicle, and mandible, completing primary ossification by gestational week 12-16.
-
Ossification Centers Formation
- Mesenchymal condensation: Week 6-8 gestation
- Runx2 expression: Master transcription factor activation
- Osterix upregulation: 48-72 hours post-Runx2
- BMP-2/4 signaling: 2-4 fold increase
- Wnt pathway activation: β-catenin stabilization
- FGF signaling: Proliferation control
-
Osteoblast Differentiation Cascade
- Pre-osteoblast stage: 3-5 days duration
- Matrix synthesis phase: 10-15 days active secretion
- Mineralization initiation: 7-10 days post-matrix
- Alkaline phosphatase peak: 5-10 fold increase
- Osteocalcin expression: Day 14-21 marker
- Type I collagen: 90% of secreted proteins
📌 Remember: ROBO pathway for intramembranous ossification - Runx2 activation, Osterix expression, BMP signaling, Osteoblast differentiation. Runx2 knockout results in complete absence of bone and cartilage formation, demonstrating its essential role as the master bone transcription factor.

| Ossification Phase | Timeline | Key Markers | Cellular Events | Clinical Relevance |
|---|---|---|---|---|
| Condensation | Week 6-8 | N-cadherin, N-CAM | Mesenchymal aggregation | Craniosynostosis risk |
| Commitment | Week 8-10 | Runx2, Osterix | Osteoprogenitor specification | Cleidocranial dysplasia |
| Differentiation | Week 10-12 | ALP, Col1a1 | Osteoblast maturation | Osteogenesis imperfecta |
| Mineralization | Week 12-16 | Osteocalcin, BSP | Matrix calcification | Hypophosphatasia |
| Maturation | Week 16+ | Sclerostin, DMP1 | Osteocyte formation | Sclerosteosis |
Endochondral ossification creates long bones through a sophisticated cartilage template that undergoes systematic replacement by bone tissue. This process begins at gestational week 8 and continues through growth plate closure in the second decade of life.
Primary Ossification Center Development
-
Chondrocyte Hypertrophy: Week 8-10 gestation
- Cell volume increase: 5-10 fold expansion
- Type X collagen expression: Hypertrophic marker
- Alkaline phosphatase upregulation: 3-5 fold
- VEGF secretion: Vascular invasion signal
-
Vascular Invasion Process: Week 10-12 gestation
- Periosteal bud formation: Blood vessel penetration
- Osteoprogenitor cell delivery: 10³-10⁴ cells/mm³
- Chondrocyte apoptosis: 70-80% cell death
- Matrix calcification: Hydroxyapatite deposition
💡 Master This: Indian Hedgehog (Ihh) and PTHrP create a negative feedback loop controlling chondrocyte proliferation and hypertrophy. Ihh from pre-hypertrophic chondrocytes stimulates PTHrP production, which inhibits further Ihh expression, maintaining the proliferative zone. Disruption causes premature growth plate closure or achondroplasia-like phenotypes.
Secondary Ossification Centers: Epiphyseal Development
- Timing: Birth to 2 years for major epiphyses
- Process: Similar to primary but smaller scale
- Growth Plate Formation: Bidirectional ossification creates physis
- Epiphyseal side: Slower ossification rate
- Metaphyseal side: Faster replacement
- Zone maintenance: Ihh/PTHrP gradient control
⭐ Clinical Pearl: Growth plate injuries account for 15-30% of pediatric fractures. Salter-Harris Type II fractures (through metaphysis and physis) have >95% good outcomes, while Type IV fractures (crossing epiphysis, physis, and metaphysis) have 30-50% growth disturbance rates due to physeal bar formation.
Bone Remodeling Cycle: Continuous Renewal
Adult bone remodeling maintains skeletal integrity through Basic Multicellular Units (BMUs) that coordinate resorption and formation. Each BMU processes 0.1-0.2 mm³ of bone over 4-6 months, with 1-2 million active BMUs in the adult skeleton.
Remodeling Sequence Phases
-
Activation Phase: 1-3 days
- Osteocyte apoptosis signals: Microdamage detection
- Lining cell retraction: 80-90% surface exposure
- Pre-osteoclast recruitment: RANKL/M-CSF signaling
- Multinucleation: 2-20 nuclei per osteoclast
-
Resorption Phase: 2-4 weeks
- Howship's lacuna formation: 40-60 μm depth
- Acid secretion: pH 4.0-4.5 microenvironment
- Enzyme release: Cathepsin K, TRAP, MMP-9
- Resorption rate: 25-40 μm³/day per osteoclast
-
Reversal Phase: 1-2 weeks
- Osteoclast apoptosis: 95% cell death
- Coupling factor release: IGF-1, TGF-β, BMPs
- Preosteoblast recruitment: 100-200 cells per BMU
- Cement line formation: 2-5 μm thickness
-
Formation Phase: 3-4 months
- Osteoid synthesis: 1-2 μm/day deposition
- Mineralization lag: 10-15 days delay
- Osteoblast fate: 65% apoptosis, 15% lining cells, 20% osteocytes
- Matrix maturation: 6-12 months complete mineralization
⭐ Clinical Pearl: Coupling efficiency decreases with age-young adults achieve 100% replacement of resorbed bone, while individuals >65 years show 85-90% replacement efficiency, leading to 0.5-1.0% annual bone loss and eventual osteoporosis development.
This dynamic remodeling system connects directly to bone's mechanical properties, where cellular-level processes determine tissue-level strength and adaptation capacity.
⚙️ Bone Formation & Remodeling — Dynamic Development
🏛️ Bone Architecture - Skeleton's Scaffolding

Hierarchical Organization: Seven Levels of Structure
Bone architecture spans multiple organizational levels, each contributing specific mechanical and biological properties. This hierarchical design enables emergent properties-characteristics that arise from structural organization rather than individual components.
Level 1: Molecular Components (1-10 nm)
- Collagen Triple Helix: 300 nm length, 1.5 nm diameter
- Glycine-X-Y repeat: 1014 amino acids per chain
- Hydroxyproline content: 10% of residues
- Cross-linking: Pyridinoline, pyrrole, aldol condensation
- Hydroxyapatite Crystals: 50 nm length, 25 nm width, 2-3 nm thickness
- Crystal orientation: c-axis parallel to collagen
- Carbonate substitution: 4-8% of phosphate sites
Level 2: Fibrillar Structure (50-500 nm)
- Collagen Fibrils: 100-200 nm diameter
- 67 nm periodicity: Gap and overlap zones
- Mineral deposition: 40% in gap zones
- Cross-link density: 1-2 per tropocollagen
- Interfibrillar Matrix: Non-collagenous proteins
- Osteopontin: Fibril-mineral interface
- Osteocalcin: 15-20% of non-collagenous content
📌 Remember: CHAMP for collagen organization - Cross-links (pyridinoline), Hydroxyproline (10%), Amino acid repeat (Gly-X-Y), Mineral deposition (gap zones), Periodicity (67 nm). This organization creates tensile strength of 150 MPa-comparable to cast iron.
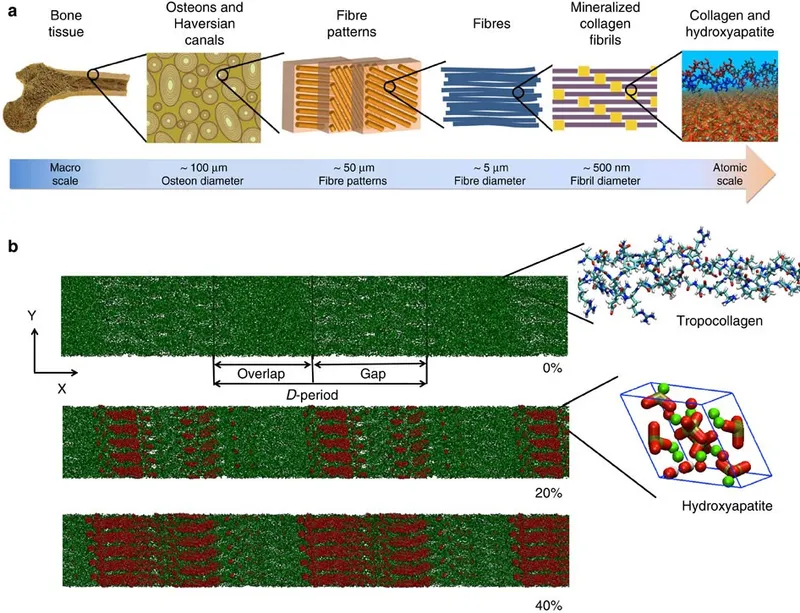
| Structural Level | Size Range | Key Components | Mechanical Contribution | Clinical Relevance |
|---|---|---|---|---|
| Molecular | 1-10 nm | Collagen, hydroxyapatite | Tensile/compressive strength | Osteogenesis imperfecta |
| Fibrillar | 50-500 nm | Mineralized fibrils | Crack deflection | Osteomalacia |
| Lamellar | 3-7 μm | Organized lamellae | Anisotropic properties | Paget's disease |
| Osteonal | 100-300 μm | Haversian systems | Fracture toughness | Stress fractures |
| Trabecular | 100-500 μm | Trabecular network | Energy absorption | Osteoporosis |
| Cortical | 1-10 mm | Compact bone | Load bearing | Cortical porosity |
| Whole bone | 10-500 mm | Complete structure | Organ-level function | Fracture patterns |
Cortical bone forms the outer shell of long bones and comprises 80% of skeletal mass while providing 90% of mechanical strength. Its dense, organized structure achieves porosity <10% compared to trabecular bone's 50-90% porosity.
Haversian System Organization
- Osteon Dimensions: 100-300 μm diameter, 5-10 mm length
- Haversian Canal: 50-100 μm diameter
- Blood vessel accommodation: 1-2 capillaries
- Nerve fiber innervation: Sympathetic terminals
- Interstitial fluid flow: 10⁻⁹ m/s velocity
- Concentric Lamellae: 3-20 layers per osteon
- Lamellar thickness: 3-7 μm
- Collagen orientation: ±30° alternating angles
- Mineral density gradient: 5-10% variation
Volkmann's Canal Network
- Orientation: Perpendicular to Haversian canals
- Function: Interconnect vascular systems
- Density: 50-100 canals/cm² cortical surface
- Clinical Significance: Osteomyelitis spread pathway
💡 Master This: Cement lines represent mineralization boundaries between old and new bone, creating weak interfaces where microcracks preferentially propagate. These lines have 30-50% lower mineral density and reduced collagen cross-linking, explaining why fatigue fractures often follow cement line patterns in cortical bone.
Cortical Porosity and Aging
- Young Adult: 5-10% porosity
- Elderly: 15-25% porosity increase
- Pore Size Distribution: 10-100 μm diameter
- Mechanical Impact: 10% porosity increase = 30% strength reduction
⭐ Clinical Pearl: Cortical porosity increases exponentially after age 50, particularly in women post-menopause. Each 1% increase in cortical porosity reduces bone strength by 3-5%, explaining why hip fracture risk increases 2-3 fold per decade after age 65 despite modest decreases in bone mineral density.

Trabecular Architecture: The Internal Framework
Trabecular bone creates an internal scaffolding that optimizes strength-to-weight ratio while providing metabolic responsiveness. Its high surface area (5-10 times greater than cortical bone) enables rapid mineral exchange and remodeling.
Trabecular Network Properties
- Trabecular Thickness: 100-200 μm average
- Trabecular Separation: 300-1500 μm spacing
- Connectivity Density: 3-15 connections/mm³
- Bone Volume Fraction: 15-35% in healthy adults
- Vertebral body: 25-30% BV/TV
- Femoral neck: 15-25% BV/TV
- Distal radius: 10-20% BV/TV
Architectural Anisotropy
- Primary Orientation: Aligned with principal stress directions
- Degree of Anisotropy: 1.5-3.0 (ratio of properties)
- Fabric Tensor: Mathematical description of orientation
- Mechanical Advantage: 2-3 fold strength increase along preferred orientation
📌 Remember: STAR for trabecular properties - Surface area (high), Thickness (100-200 μm), Anisotropy (stress-aligned), Remodeling (rapid turnover). Trabecular bone remodels 8-10 times faster than cortical bone, making it the primary target for osteoporosis and metabolic bone diseases.
Trabecular Bone Loss Patterns
- Thinning: 2-5 μm/year after age 30
- Perforation: Critical thickness <100 μm
- Connectivity Loss: Irreversible structural damage
- Mechanical Consequences: Exponential strength reduction
⭐ Clinical Pearl: Trabecular connectivity loss is irreversible-once trabeculae perforate and disconnect, anabolic therapy cannot restore the original architecture. This explains why prevention is more effective than treatment for osteoporosis, and why early intervention before connectivity loss is crucial for maintaining bone strength.
Understanding bone's architectural hierarchy connects directly to its mechanical behavior, where structural organization at multiple scales determines the tissue's remarkable ability to resist fracture while maintaining biological function.
🏛️ Bone Architecture — Skeleton's Scaffolding
⚡ Bone Mechanics & Blood Supply - Force & Flow

Mechanical Properties: Engineering Excellence
Bone exhibits anisotropic mechanical properties, meaning strength varies with loading direction. This directional dependence reflects the tissue's adaptive response to habitual loading patterns, optimizing material distribution for functional demands.
Cortical Bone Mechanical Properties
- Compressive Strength: 130-180 MPa (longitudinal)
- Transverse direction: 50-70 MPa (60% reduction)
- Age-related decline: 2-5% per decade after age 40
- Gender differences: 10-15% higher in males
- Tensile Strength: 120-150 MPa (longitudinal)
- Transverse direction: 30-50 MPa (70% reduction)
- Strain rate sensitivity: 20-30% increase at high rates
- Elastic Modulus: 15-20 GPa (longitudinal)
- Transverse direction: 8-12 GPa
- Wet vs. dry: 25% reduction when hydrated
Trabecular Bone Mechanical Characteristics
- Compressive Strength: 2-15 MPa (density-dependent)
- Power law relationship: Strength ∝ (density)^1.8-2.0
- Architectural dependence: 50% from density, 50% from structure
- Elastic Modulus: 50-500 MPa
- Fabric tensor influence: 2-3 fold directional variation
- Connectivity impact: Exponential relationship
📌 Remember: FAST for bone mechanical properties - Fatigue resistance (10⁶ cycles), Anisotropy (directional strength), Strain rate effects (20-30% increase), Toughness (crack resistance). Bone's toughness (energy to fracture) is 3-5 times higher than its constituent materials due to hierarchical structure and crack deflection mechanisms.
| Property | Cortical (Longitudinal) | Cortical (Transverse) | Trabecular | Clinical Significance |
|---|---|---|---|---|
| Compressive Strength | 130-180 MPa | 50-70 MPa | 2-15 MPa | Vertebral fractures |
| Tensile Strength | 120-150 MPa | 30-50 MPa | 1-5 MPa | Long bone fractures |
| Elastic Modulus | 15-20 GPa | 8-12 GPa | 0.05-0.5 GPa | Implant matching |
| Ultimate Strain | 1.5-3.0% | 0.5-1.0% | 5-10% | Energy absorption |
| Toughness | 2-6 kJ/m² | 1-3 kJ/m² | 0.1-1 kJ/m² | Fracture resistance |
Bone experiences cyclic loading throughout life, accumulating millions of loading cycles that can lead to fatigue damage. Understanding fatigue mechanisms explains stress fractures and age-related bone fragility.
Microdamage Formation and Repair
- Microcrack Density: 0.5-2.0 cracks/mm² in healthy bone
- Critical Crack Length: 100-200 μm for propagation
- Crack Growth Rate: 10⁻⁸-10⁻⁶ m/cycle
- Repair Timeline: 7-14 days for targeted remodeling activation
Fatigue Life Relationships
- High-Cycle Fatigue: >10⁶ cycles at <50% ultimate strength
- Low-Cycle Fatigue: <10⁴ cycles at >80% ultimate strength
- Endurance Limit: 60-70% of ultimate strength
- Age Effects: 50% reduction in fatigue life by age 80
💡 Master This: Wolff's Law states that bone adapts to mechanical demands, but this adaptation has limits. When loading rate exceeds remodeling capacity, microdamage accumulates faster than repair, leading to stress fractures. Athletes training >20 hours/week show 3-5 fold increased stress fracture risk due to this imbalance.
Vascular Architecture: The Perfusion Network
Bone's blood supply represents 5-10% of cardiac output despite comprising 15% of body weight, reflecting its high metabolic activity. The vascular network must navigate the tissue's mineralized matrix while providing nutrients to embedded osteocytes.
Cortical Bone Vascularization
- Periosteal Vessels: 70-80% of cortical blood supply
- Penetrating arteries: 100-200 μm diameter
- Branching pattern: Dichotomous division
- Penetration depth: 1-2 mm into cortex
- Endosteal Circulation: 20-30% of supply
- Medullary artery branches: 50-100 μm diameter
- Centrifugal flow pattern: Inside-out perfusion
- Haversian Circulation: Capillary networks in osteons
- Capillary diameter: 5-10 μm
- Flow velocity: 0.5-1.0 mm/s
- Oxygen tension: 30-50 mmHg
Trabecular Bone Blood Flow
- Sinusoidal Circulation: Low-pressure system
- Flow Rate: 2-3 times higher than cortical bone
- Oxygen Extraction: 20-30% (vs. 40-50% in cortical)
- Metabolic Exchange: Rapid due to high surface area
⭐ Clinical Pearl: Cortical bone blood flow decreases 30-50% with aging, particularly affecting the outer cortical regions supplied by periosteal vessels. This explains why cortical bone healing is slower in elderly patients and why nonunions are more common in cortical bone fractures compared to cancellous bone injuries.
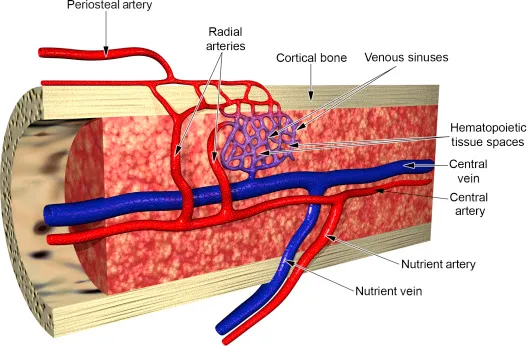
Vascular Response to Mechanical Loading
- Flow-Mediated Adaptation: 20-40% increase with exercise
- Mechanotransduction: Osteocyte-mediated vessel regulation
- VEGF Expression: 2-5 fold increase with loading
- Angiogenesis: New vessel formation in 7-14 days
Clinical Vascular Considerations
- Avascular Necrosis Risk Sites:
- Femoral head: Retrograde blood supply
- Scaphoid waist: Watershed region
- Fifth metatarsal base: Poor collateral circulation
- Healing Implications: Vascular density correlates with healing rate
- Well-vascularized: 6-8 weeks (cancellous bone)
- Poorly-vascularized: 12-16 weeks (cortical bone)
⭐ Clinical Pearl: Smoking reduces bone blood flow by 25-40% through nicotine-induced vasoconstriction and carbon monoxide reducing oxygen delivery. This explains why smokers have 2-3 times higher nonunion rates and delayed healing times 50-100% longer than non-smokers.
Understanding the intricate relationship between bone mechanics and vascular supply reveals how loading patterns influence both structural adaptation and healing capacity, connecting directly to clinical applications in fracture management and bone health optimization.
⚡ Bone Mechanics & Blood Supply — Force & Flow
🎯 High‑Yield Points - ⚡ Biggest Takeaways

Essential Clinical Correlations
Bone Composition Clinical Impact
- Hydroxyapatite:Collagen Ratio determines fracture pattern
- High mineral content: Brittle fractures (osteoporosis)
- Low mineral content: Soft bone deformity (osteomalacia)
- Abnormal collagen: Multiple fractures (osteogenesis imperfecta)
- Age-Related Changes: Progressive deterioration after age 30
- Cortical porosity: Doubles every 20 years after age 50
- Trabecular connectivity: Irreversible loss after perforation
- Collagen cross-linking: Advanced glycation reduces toughness
⭐ Clinical Pearl: T-score interpretation requires understanding bone composition-a T-score of -2.5 represents 30% bone loss, but fracture risk depends on bone quality (collagen integrity, microdamage) not just quantity (mineral density). Patients with diabetes show normal BMD but increased fracture risk due to advanced glycation end-products affecting collagen.
Cellular Activity Biomarkers
- Formation Markers: Predict anabolic response
- Osteocalcin: 15-50 ng/mL (bone-specific)
- P1NP: 20-80 ng/mL (collagen synthesis)
- Alkaline phosphatase: 50-120 U/L (mineralization)
- Resorption Markers: Monitor catabolic activity
- CTX: 100-600 pg/mL (collagen breakdown)
- NTX: 5-25 nM BCE/mM creatinine
- TRAP5b: 2.5-6.5 U/L (osteoclast activity)
📌 Remember: FORM for formation markers - Formation (osteocalcin), Osteoblast activity (ALP), Remodeling (P1NP), Mineralization (bone ALP). BREAK for resorption markers - Bone breakdown (CTX), Resorption (NTX), Enzyme activity (TRAP), Acid production (DPD), Key monitoring (CTX most sensitive).

| Clinical Scenario | Key Markers | Expected Values | Therapeutic Implications |
|---|---|---|---|
| Osteoporosis | CTX ↑, Osteocalcin ↑ | CTX >600 pg/mL | Antiresorptive therapy |
| Paget's Disease | ALP ↑↑, CTX ↑↑ | ALP >300 U/L | Bisphosphonate treatment |
| Hyperparathyroidism | CTX ↑, P1NP ↑ | PTH >65 pg/mL | Surgical consideration |
| Anabolic Response | P1NP ↑, CTX ↓ | P1NP >100 ng/mL | Teriparatide efficacy |
| Bisphosphonate Effect | CTX ↓↓, Osteocalcin ↓ | CTX <200 pg/mL | Treatment monitoring |
Mechanical Property Clinical Translation
- Cortical Bone Assessment: Hip fracture prediction
- Femoral neck BMD: Primary predictor
- Cortical thickness: <3 mm = high risk
- Buckling ratio: >10 indicates instability
- Trabecular Bone Evaluation: Vertebral fracture risk
- Spine BMD: T-score correlation
- Trabecular bone score: Microarchitecture assessment
- Connectivity index: Structural integrity
FRAX Integration with Bone Biology
- 10-Year Fracture Probability: Combines clinical factors with BMD
- Major Osteoporotic Fracture: >20% = treatment threshold
- Hip Fracture Risk: >3% = intervention indicated
- Adjustment Factors: TBS, falls risk, diabetes
💡 Master This: Bone strength = Bone density × Bone quality. DEXA measures density but misses quality factors (microdamage, collagen cross-linking, microarchitecture). This explains why 50% of fragility fractures occur in patients with T-scores >-2.5, emphasizing the importance of clinical risk factors beyond BMD.
Therapeutic Target Mastery
Anabolic vs. Antiresorptive Strategy
-
Anabolic Therapy (Teriparatide, Abaloparatide):
- Mechanism: PTH receptor activation → cAMP → osteoblast proliferation
- Optimal Candidates: Severe osteoporosis (T-score <-3.0)
- Response Monitoring: P1NP increase >50% within 3 months
- Duration Limit: 24 months maximum (osteosarcoma risk)
-
Antiresorptive Therapy (Bisphosphonates, Denosumab):
- Mechanism: Osteoclast inhibition → reduced bone turnover
- Optimal Candidates: High turnover osteoporosis
- Response Monitoring: CTX decrease >70% within 3 months
- Duration Considerations: Drug holidays after 5-10 years
⭐ Clinical Pearl: Sequential therapy maximizes bone gain-anabolic first (18-24 months) followed by antiresorptive provides superior outcomes compared to antiresorptive-first approaches. Anabolic therapy after bisphosphonates shows blunted response due to suppressed bone turnover, requiring 6-12 month washout for optimal effect.
Monitoring Treatment Efficacy
- BMD Response: 3-5% increase expected annually
- Biomarker Changes: Significant within 3-6 months
- Fracture Reduction: Vertebral 40-70%, Hip 20-40%
- Treatment Failure: <3% BMD increase or continued fractures
This comprehensive understanding of bone structure and function provides the foundation for evidence-based clinical decision-making in orthopedic practice, connecting cellular mechanisms to patient outcomes through quantitative assessment and targeted therapeutic interventions.
🎯 High‑Yield Points — ⚡ Biggest Takeaways
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more
Have doubts about this lesson?
Ask Rezzy, your AI Study Mate, to explain anything you didn't understand
Everything you need for NEET-PG prep
Get full Oncourse access with lessons, practice questions, flashcards and AI study tools.
Scan to download app










