Menopause

On this page
🌸 The Menopausal Metamorphosis: Decoding Life's Hormonal Revolution
Menopause marks one of medicine's most universal yet individually variable transitions, where declining ovarian function triggers a cascade affecting nearly every organ system. You'll master the hormonal shifts driving vasomotor symptoms, bone loss, and cardiovascular changes, then build a systematic approach to diagnosis and evidence-based management including hormone therapy risks and benefits. By integrating endocrine, skeletal, cardiovascular, and genitourinary perspectives, you'll confidently guide patients through this decades-long transition with personalized, scientifically grounded care.
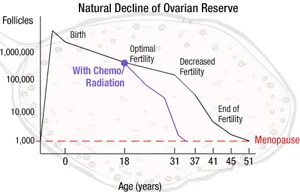
The World Health Organization defines menopause as the permanent cessation of menstruation resulting from loss of ovarian follicular activity, confirmed after 12 consecutive months of amenorrhea without other pathological causes. This definition distinguishes natural menopause from surgical or medical menopause, which can occur at any age.
📌 Remember: MENOPAUSE - Menstruation Ends Naturally Ovarian Production Absent Uterine Silence Estrogen - Occurs after 12 months of amenorrhea with FSH >30 IU/L and estradiol <20 pg/mL
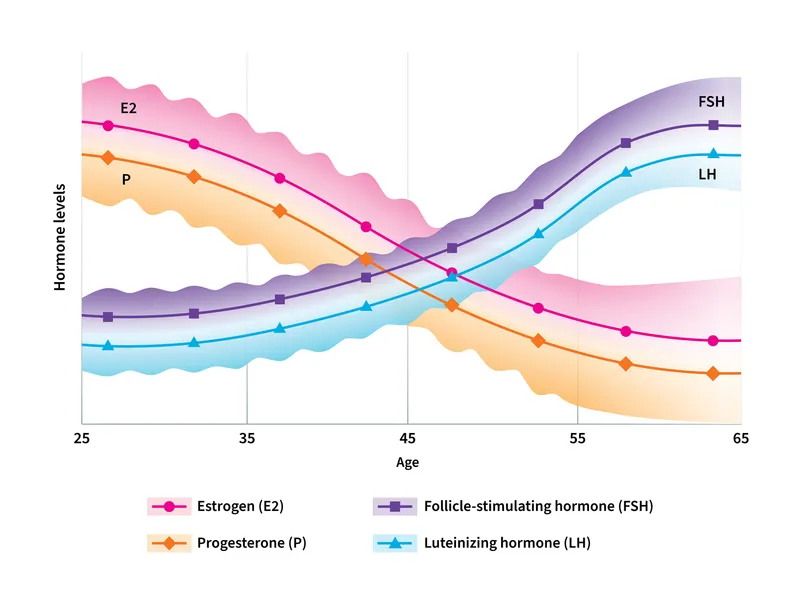
The menopausal transition involves three distinct phases with specific hormonal characteristics:
-
Perimenopause (Menopausal Transition)
- Duration: 2-8 years before final menstrual period
- FSH levels: Begin rising above 10 IU/L in early follicular phase
- Cycle irregularity: Cycles vary by ≥7 days from normal
- Early perimenopause: Subtle cycle changes, occasional anovulation
- Late perimenopause: ≥2 skipped cycles with ≥60-day amenorrheic intervals
-
Menopause (Final Menstrual Period)
- Retrospective diagnosis: Confirmed after 12 months amenorrhea
- Average age: 51.4 years in North American women
- Hormonal markers: FSH >40 IU/L, estradiol <30 pg/mL
-
Postmenopause
- Early postmenopause: First 5 years after final menstrual period
- Late postmenopause: Beyond 5 years postmenopause
- Estradiol levels: Remain consistently <20 pg/mL
| Parameter | Premenopause | Perimenopause | Postmenopause | Clinical Significance | Reference Range |
|---|---|---|---|---|---|
| FSH (IU/L) | 3-10 | 10-40 | >40 | Primary diagnostic marker | Varies by cycle phase |
| Estradiol (pg/mL) | 30-400 | Variable | <30 | Reflects ovarian function | Fluctuates significantly |
| Inhibin B (pg/mL) | 20-150 | <20 | <10 | Early marker of decline | Most sensitive early indicator |
| AMH (ng/mL) | 1.0-4.0 | <1.0 | <0.1 | Ovarian reserve assessment | Age-dependent decline |
| Cycle Length (days) | 21-35 | Variable | Absent | Clinical assessment tool | >7-day variation significant |
The ovarian aging process begins in utero, with peak follicle numbers of 6-7 million at 20 weeks gestation, declining to 1-2 million at birth and 300,000-400,000 at menarche. The rate of follicular atresia accelerates dramatically after age 37-38 years, when approximately 25,000 follicles remain.
💡 Master This: The "two-cell, two-gonadotropin" theory explains ovarian hormone production - theca cells produce androgens under LH stimulation, while granulosa cells convert androgens to estrogens via aromatase under FSH influence. Menopause occurs when <1,000 follicles remain, insufficient to maintain this coordinated process.
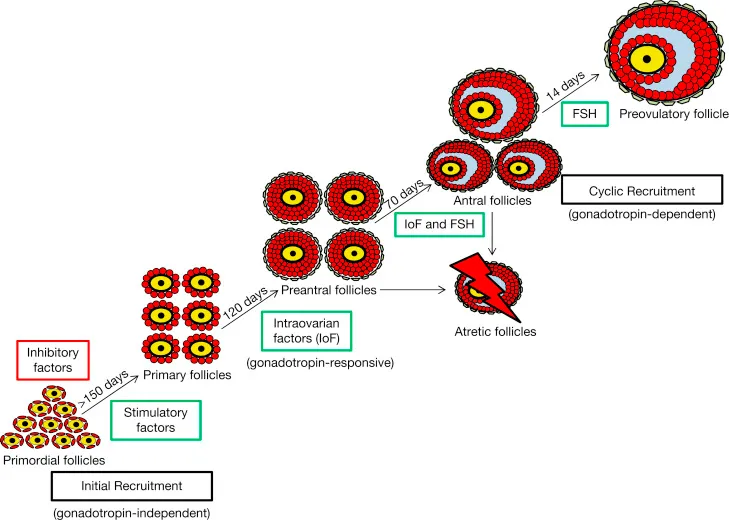
Understanding this hormonal blueprint establishes the foundation for recognizing how estrogen deficiency triggers the cascade of menopausal symptoms and long-term health consequences that define the next phase of this endocrine journey.
🌸 The Menopausal Metamorphosis: Decoding Life's Hormonal Revolution
⚡ The Estrogen Exodus: Unraveling Hormonal Havoc
📌 Remember: FOLLICLE - FSH Overstimulation Loss Luteal Inadequacy Cycles Lengthening Estrogen - The 25,000 follicle threshold triggers FSH elevation and inhibin B decline 2-5 years before menopause
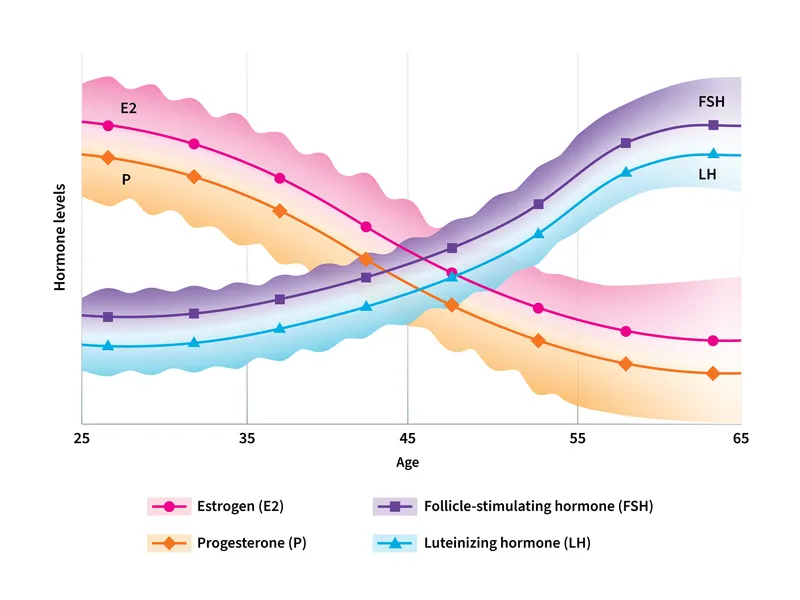
The primary hormonal changes during menopausal transition follow a predictable sequence:
-
Early Perimenopause (2-8 years before menopause)
- Inhibin B decline: Falls below 45 pg/mL (normal: 20-150 pg/mL)
- FSH elevation: Early follicular phase levels >10 IU/L
- Estradiol fluctuation: Wide variations from <20 to >200 pg/mL
- Compensatory hyperstimulation: Remaining follicles produce supraphysiologic estrogen
- Cycle irregularity: ±7 days variation from baseline cycle length
- Anovulatory cycles: Increase from 5% to 15-20%
-
Late Perimenopause (1-3 years before menopause)
- Progressive FSH rise: Levels 20-40 IU/L
- Estradiol instability: Dramatic fluctuations with overall declining trend
- Amenorrheic intervals: ≥60 days between cycles
- Luteal phase defects: Progesterone <3 ng/mL in >50% of cycles
- Cycle length variation: >14 days from baseline
| Hormone | Premenopause | Early Perimenopause | Late Perimenopause | Postmenopause | Clinical Impact |
|---|---|---|---|---|---|
| Estradiol (pg/mL) | 30-400 | 20-300 (erratic) | 10-100 (declining) | <20 | Vasomotor symptoms |
| FSH (IU/L) | 3-10 | 10-25 | 25-40 | >40 | Diagnostic marker |
| LH (IU/L) | 2-15 | 5-20 | 15-30 | >30 | Secondary elevation |
| Progesterone (ng/mL) | 5-20 (luteal) | 1-10 (irregular) | <3 (most cycles) | <1 | Cycle irregularity |
| Testosterone (ng/dL) | 20-70 | 15-50 | 10-40 | 5-25 | Libido changes |
- Quantitative follicular depletion: <1,000 remaining follicles cannot maintain adequate estradiol production
- Qualitative follicular dysfunction: Remaining follicles show reduced aromatase activity and impaired steroidogenesis
- Stromal cell changes: Ovarian stroma increases androgen production while estrogen synthesis declines
- Peripheral conversion alterations: Adipose tissue aromatization becomes the primary estrogen source (estrone >estradiol)
⭐ Clinical Pearl: Estrone becomes the predominant estrogen in postmenopause, produced via peripheral aromatization of androstenedione in adipose tissue. This explains why obese women experience fewer severe vasomotor symptoms but higher endometrial cancer risk.

The failure cascade manifests through specific clinical patterns:
- Vasomotor instability: 75-85% of women experience hot flashes due to hypothalamic thermoregulatory dysfunction
- Urogenital atrophy: Estrogen receptor downregulation causes tissue thinning and pH elevation from 4.5 to 6.5-7.0
- Bone metabolism disruption: Estrogen deficiency increases osteoclast activity by 40-50%, leading to 2-3% annual bone loss
- Cardiovascular risk elevation: Loss of estrogen's cardioprotective effects increases CHD risk by 2-3 fold within 10 years
💡 Master This: The "estrogen threshold hypothesis" explains symptom variability - vasomotor symptoms require >50% estradiol decline, while urogenital changes occur with >80% reduction. Individual sensitivity to estrogen withdrawal varies 10-fold between women.
Understanding these hormonal mechanisms reveals how estrogen deficiency creates the symptom constellation that characterizes the clinical presentation of menopause, setting the stage for targeted therapeutic interventions.
⚡ The Estrogen Exodus: Unraveling Hormonal Havoc
🎯 The Symptom Constellation: Mastering Clinical Recognition
The "see this, think menopause" framework organizes symptoms by system involvement and diagnostic probability:
-
Pathognomonic Symptoms (>90% specificity)
- Hot flashes: Sudden onset warmth lasting 1-5 minutes with diaphoresis
- Night sweats: Sleep-disrupting episodes requiring clothing/bedding changes
- Irregular cycles: >7-day variation from baseline in women >40 years
- Pattern recognition: Lengthening cycles → skipped periods → amenorrhea
- Bleeding changes: Heavier or lighter flow with unpredictable timing
-
Highly Suggestive Symptoms (70-85% association)
- Sleep disturbances: Difficulty maintaining sleep in 60-80% of perimenopausal women
- Mood changes: Irritability, anxiety, or depression in previously stable women
- Cognitive symptoms: "Brain fog", memory lapses, concentration difficulties
- Executive function: Working memory deficits during hormonal fluctuations
- Verbal memory: Word-finding difficulties in 40-60% of women
📌 Remember: FLASHES - Frequency Lasting Associated Sleep Hormonal Estrogen Severity - Hot flashes occur in 75-85% of women, lasting 1-5 minutes, 7-10 episodes daily at peak, triggered by estradiol fluctuations >20 pg/mL
| Symptom Category | Prevalence (%) | Onset Timing | Duration | Severity Markers | Clinical Assessment |
|---|---|---|---|---|---|
| Vasomotor | 75-85 | Early perimenopause | 2-7 years | >7 episodes/day | Hot flash frequency/severity scale |
| Urogenital | 50-70 | Late perimenopause | Progressive | pH >6.0, VMI <40 | Vaginal maturation index |
| Psychological | 40-60 | Variable | 2-5 years | PHQ-9 >10 | Standardized mood scales |
| Sleep | 60-80 | Early perimenopause | 3-8 years | >3 awakenings/night | Pittsburgh Sleep Quality Index |
| Cognitive | 40-65 | Perimenopause peak | 1-3 years | Subjective complaints | Cognitive assessment tools |
-
Hot Flash Characteristics
- Frequency: 1-20 episodes daily (average 7-10)
- Duration: 30 seconds to 10 minutes (typical 2-4 minutes)
- Intensity: Mild (awareness) to severe (incapacitating)
- Physiologic changes: Core temperature rise 0.5-1.0°C
- Cardiovascular response: Heart rate increase 10-15 bpm
- Skin conductance: 3-5 fold increase during episodes
-
Trigger Identification
- Environmental: Warm temperatures, crowded spaces, spicy foods
- Behavioral: Alcohol consumption, caffeine intake, stress
- Hormonal: Estradiol fluctuations >20 pg/mL within 24-48 hours
⭐ Clinical Pearl: The "thermoregulatory zone" narrows from normal 0.4°C range to <0.1°C during menopause. Any trigger causing minimal core temperature elevation precipitates vasomotor symptoms due to hypothalamic hypersensitivity.
Urogenital symptoms develop progressively with cumulative estrogen deficiency:
-
Vulvovaginal Atrophy (Genitourinary Syndrome of Menopause)
- Vaginal dryness: 50-60% of postmenopausal women
- Dyspareunia: Pain during intercourse in 30-45%
- Vaginal pH elevation: From 3.8-4.5 to 6.0-7.5
- Epithelial thinning: Maturation index <40% superficial cells
- Elasticity loss: Vaginal rugae flattening and tissue friability
- Lubrication decrease: >50% reduction in arousal response
-
Urinary Symptoms
- Urgency/frequency: Detrusor instability in 30-40%
- Recurrent UTIs: pH elevation promotes pathogenic bacteria
- Stress incontinence: Urethral tissue atrophy and pelvic floor changes
💡 Master This: Estrogen receptors are abundant in vaginal epithelium, urethral tissue, and bladder trigone. Estrogen deficiency causes collagen degradation, decreased blood flow, and tissue atrophy within 6-12 months of menopause onset.
Psychological symptom patterns require careful differentiation from primary mood disorders:
- Perimenopausal Depression
- Risk increase: 2-4 fold higher than premenopausal women
- Hormonal correlation: Estradiol variability >stable low levels
- Symptom profile: Irritability, anxiety, mood lability predominate
- Sleep disruption: Secondary to vasomotor symptoms in 60-70%
- Cognitive complaints: Subjective >objective deficits on testing

Understanding these symptom patterns enables systematic assessment and targeted interventions for the complex clinical presentations that characterize menopausal transition.
🎯 The Symptom Constellation: Mastering Clinical Recognition
🔬 The Diagnostic Detective: Systematic Assessment Mastery
Diagnostic certainty in menopause relies on pattern recognition rather than single laboratory values, as hormonal fluctuations during perimenopause create wide variability in biochemical markers. The clinical context combined with age-appropriate symptoms provides greater diagnostic accuracy than isolated hormone levels.
📌 Remember: DIAGNOSIS - Duration Irregular Age Gonadotropins No Other Symptoms Investigate Stages - 12 months amenorrhea in women >45 years with typical symptoms requires no laboratory confirmation
The diagnostic framework follows evidence-based guidelines with specific testing indications:
-
Clinical Diagnosis (No laboratory testing required)
- Age >45 years with typical symptoms
- Progressive cycle irregularity over 6-12 months
- Vasomotor symptoms in appropriate clinical context
- Bilateral oophorectomy: Immediate surgical menopause
- Chemotherapy/radiation: Iatrogenic ovarian failure
-
Laboratory Testing Indications
- Age <45 years with menopausal symptoms
- Unclear clinical picture or atypical presentation
- Differential diagnosis considerations
- Premature ovarian insufficiency: Age <40 years
- Secondary amenorrhea: Rule out other causes
- Hormone therapy decisions requiring baseline values
| Test | Timing | Menopausal Range | Clinical Utility | Limitations | Cost-Effectiveness |
|---|---|---|---|---|---|
| FSH | Early follicular | >25-30 IU/L | High specificity | Fluctuates in perimenopause | First-line when indicated |
| Estradiol | Any time | <30 pg/mL | Confirms deficiency | Wide daily variation | Adjunctive to FSH |
| LH | Any time | >30 IU/L | Secondary marker | Less specific than FSH | Limited clinical value |
| Inhibin B | Early follicular | <20 pg/mL | Earliest marker | Expensive, limited availability | Research primarily |
| AMH | Any time | <0.1 ng/mL | Cycle-independent | Age-dependent decline | Emerging clinical use |
-
Primary Amenorrhea (if age <16 years)
- Genetic disorders: Turner syndrome, Swyer syndrome
- Anatomical abnormalities: Müllerian agenesis, imperforate hymen
- Endocrine disorders: Constitutional delay, hypothalamic dysfunction
-
Secondary Amenorrhea (in reproductive-age women)
- Pregnancy: Always exclude with β-hCG
- Hypothalamic causes: Stress, weight loss, exercise, medications
- Pituitary disorders: Prolactinoma, Sheehan syndrome, empty sella
- Thyroid dysfunction: Hypo/hyperthyroidism affects 15-20% of women
- PCOS: Oligomenorrhea with hyperandrogenism
- Premature ovarian insufficiency: Age <40 years with FSH >40 IU/L
⭐ Clinical Pearl: Single FSH measurement during perimenopause has limited diagnostic value due to monthly fluctuations. Two elevated values (>25 IU/L) 6 weeks apart provide 85% diagnostic accuracy for late perimenopause.
Specialized testing for complex presentations:
-
Premature Ovarian Insufficiency Workup
- Karyotype analysis: Rule out Turner mosaicism
- FMR1 premutation: Fragile X carrier status
- Autoimmune markers: 21-hydroxylase antibodies, thyroid antibodies
- Adrenal function: Morning cortisol, ACTH stimulation test
- Bone density: DEXA scan for osteoporosis risk
-
Atypical Presentations
- Testosterone levels: Rule out androgen-secreting tumors
- Prolactin: Exclude prolactinoma if galactorrhea present
- TSH: Thyroid dysfunction mimics menopausal symptoms
💡 Master This: The "clinical gestalt" approach combines age, symptom pattern, menstrual history, and family history to achieve >95% diagnostic accuracy without laboratory testing in typical presentations. Biochemical confirmation is reserved for atypical cases or therapeutic decisions.
Documentation standards ensure comprehensive assessment:
- Menstrual history: Last menstrual period, cycle patterns, bleeding changes
- Symptom inventory: Vasomotor, urogenital, psychological, sleep disturbances
- Risk factor assessment: Family history, surgical history, medications
- Quality of life impact: Functional impairment, relationship effects, work productivity
This systematic diagnostic approach establishes the foundation for evidence-based treatment decisions and personalized management strategies that address individual patient needs and risk profiles.
🔬 The Diagnostic Detective: Systematic Assessment Mastery
⚖️ The Treatment Algorithm: Evidence-Based Management Mastery
Treatment algorithms follow international guidelines with evidence-based decision trees that prioritize symptom relief, quality of life improvement, and long-term health optimization. The individualized approach considers contraindications, patient preferences, and risk stratification to achieve optimal therapeutic outcomes.
📌 Remember: TREATMENT - Timing Risks Efficacy Alternatives Titration Monitoring Evaluation Non-hormonal Tailored - Hormone therapy within 10 years of menopause or age <60 provides optimal benefit-risk ratio
First-line management follows stepped-care approach:
-
Lifestyle Interventions (All patients)
- Dietary modifications: Phytoestrogen-rich foods (40-80 mg/day isoflavones)
- Exercise prescription: 150 minutes/week moderate activity + resistance training
- Weight management: BMI 18.5-24.9 reduces vasomotor symptoms by 30-40%
- Smoking cessation: Reduces hot flash frequency by 25-30%
- Alcohol limitation: <7 drinks/week improves sleep quality
- Stress reduction: Mindfulness, yoga, cognitive behavioral therapy
-
Symptom-Specific Interventions
- Vasomotor symptoms: Layered clothing, cooling techniques, trigger avoidance
- Sleep hygiene: Cool bedroom (65-68°F), consistent schedule, screen limitation
- Urogenital health: Regular sexual activity, vaginal moisturizers, pelvic floor exercises
| Treatment Category | Efficacy (% Reduction) | Time to Effect | Duration of Use | Monitoring Requirements | Cost Range |
|---|---|---|---|---|---|
| Lifestyle Only | 20-40% | 4-8 weeks | Indefinite | Clinical assessment | Low |
| Hormone Therapy | 75-90% | 2-4 weeks | 3-5 years typical | Annual comprehensive | Moderate |
| Non-hormonal Rx | 40-60% | 4-8 weeks | Variable | Drug-specific | Moderate-High |
| Complementary | 15-30% | 6-12 weeks | Variable | Minimal | Low-Moderate |
| Combined Approach | 80-95% | 2-6 weeks | Individualized | Comprehensive | Variable |
-
Candidate Selection Criteria
- Age <60 years or within 10 years of menopause
- Moderate-severe vasomotor symptoms affecting quality of life
- No absolute contraindications to estrogen therapy
- Cardiovascular risk assessment: <10% 10-year risk preferred
- Thrombotic risk evaluation: Personal/family history consideration
- Breast cancer screening: Up-to-date mammography
-
HRT Regimen Selection
- Estrogen type: 17β-estradiol preferred over conjugated equine estrogens
- Delivery route: Transdermal for VTE risk reduction vs oral for convenience
- Progestogen addition: Required for intact uterus to prevent endometrial hyperplasia
- Continuous combined: Daily estrogen + progestogen for postmenopausal women
- Sequential regimen: Cyclic progestogen for perimenopausal women
⭐ Clinical Pearl: Transdermal estradiol (0.05-0.1 mg/day) with micronized progesterone (100-200 mg/day) provides optimal safety profile with 90% symptom reduction and minimal VTE risk increase (RR 1.2 vs oral RR 2.1).
Non-hormonal pharmacotherapy for HRT-ineligible patients:
-
Selective Serotonin Reuptake Inhibitors (SSRIs)
- Paroxetine (7.5 mg/day): FDA-approved for vasomotor symptoms
- Efficacy: 50-65% reduction in hot flash frequency
- Onset: 2-4 weeks for maximum benefit
- Venlafaxine (37.5-75 mg/day): 60-70% reduction in moderate-severe symptoms
- Citalopram (10-20 mg/day): 50% reduction with good tolerability
-
Other Pharmacological Options
- Gabapentin (300-900 mg/day): 45-60% reduction, especially night sweats
- Clonidine (0.1-0.2 mg/day): 30-40% reduction, limited by side effects
- Fezolinetant (45 mg/day): New NK3 receptor antagonist, 65% reduction
💡 Master This: Shared decision-making integrates patient values, symptom severity, risk tolerance, and treatment preferences to achieve personalized management plans. Regular reassessment every 6-12 months ensures optimal outcomes and appropriate therapy duration.
Monitoring protocols ensure treatment safety and efficacy:
-
Initial Assessment (Baseline)
- Comprehensive history: Symptoms, risk factors, contraindications
- Physical examination: Blood pressure, BMI, breast/pelvic exam
- Laboratory studies: Lipid profile, glucose, liver function if indicated
-
Follow-up Schedule
- 3 months: Symptom response, side effects, adherence assessment
- 6-12 months: Comprehensive evaluation, risk reassessment
- Annual: Mammography, cervical cytology, bone density if indicated
This evidence-based treatment framework provides the foundation for individualized care plans that optimize symptom relief while minimizing long-term health risks through systematic monitoring and therapeutic adjustments.
⚖️ The Treatment Algorithm: Evidence-Based Management Mastery
🔗 The Systemic Symphony: Multi-System Integration Mastery
Systems integration during menopause reveals how estrogen receptors distributed throughout the body create far-reaching consequences beyond reproductive function. Understanding these multi-system interactions enables comprehensive management that addresses immediate symptoms while preventing long-term complications affecting >50 million postmenopausal women in the United States.
The estrogen receptor network spans multiple organ systems with tissue-specific responses:
-
Cardiovascular System (ERα and ERβ distribution)
- Vascular endothelium: Nitric oxide production, vasodilation, anti-inflammatory effects
- Myocardium: Calcium handling, contractility, arrhythmia protection
- Lipid metabolism: HDL elevation (10-15%), LDL reduction (10-20%)
- Coronary risk: 2-3 fold increase within 10 years postmenopause
- Stroke risk: 1.5-2 fold elevation in early postmenopause
- Hypertension: Prevalence increases from 30% to 60% postmenopause
-
Skeletal System (Bone remodeling integration)
- Osteoblast activity: Estrogen deficiency reduces bone formation by 30-40%
- Osteoclast regulation: RANKL/OPG pathway disruption increases resorption by 50-60%
- Calcium homeostasis: Intestinal absorption decreases 10-15%
- Bone loss rate: 2-3% annually for 5-7 years postmenopause
- Fracture risk: Hip fractures increase 2-3 fold by age 70
- Vertebral compression: 25% of women by age 65
📌 Remember: SYSTEMS - Skeletal Years Stroke Thrombosis Endothelial Metabolic Sleep - Estrogen deficiency affects >8 organ systems with peak bone loss in first 5 years, cardiovascular risk doubling by 10 years, and metabolic changes within 2-3 years
| System | Primary Effects | Timeline | Risk Magnitude | Prevention Strategy | Monitoring Frequency |
|---|---|---|---|---|---|
| Cardiovascular | Endothelial dysfunction | 2-5 years | 2-3x CHD risk | Lifestyle + HRT if appropriate | Annual |
| Skeletal | Accelerated bone loss | 1-2 years | 2-3x fracture risk | Calcium/Vitamin D + exercise | 1-2 years |
| Metabolic | Insulin resistance | 1-3 years | 1.5-2x diabetes risk | Weight management + activity | Annual |
| Neurological | Cognitive changes | Variable | Dementia risk unclear | Mental stimulation + HRT? | As needed |
| Urogenital | Tissue atrophy | 6 months-2 years | Progressive dysfunction | Local estrogen + lubricants | Annual |
-
Metabolic Syndrome Development
- Visceral adiposity: Central fat accumulation increases 30-40% postmenopause
- Insulin resistance: HOMA-IR increases 25-35% within 3-5 years
- Dyslipidemia: Triglycerides increase 20-30%, HDL decreases 10-15%
- Inflammatory markers: CRP elevation 2-3 fold in early postmenopause
- Endothelial function: Flow-mediated dilation decreases 15-25%
- Blood pressure: Systolic increases 5-10 mmHg over 5 years
-
Thrombotic Risk Integration
- Coagulation factors: Factor VII, fibrinogen increase 10-20%
- Platelet aggregation: Enhanced responsiveness to agonists
- Fibrinolytic capacity: PAI-1 elevation impairs clot dissolution
Bone-muscle-metabolic axis reveals interconnected pathways:
- Sarcopenia Development
- Muscle mass loss: 1-2% annually after age 50
- Strength decline: 2-3% annually in postmenopausal women
- Functional capacity: Reduced by 20-30% over 10 years
- Protein synthesis: Estrogen deficiency impairs muscle protein balance
- Mitochondrial function: Decreased efficiency affects energy metabolism
- Fall risk: Increases 2-3 fold with combined bone-muscle loss
⭐ Clinical Pearl: The "bone-muscle-fat triad" demonstrates how estrogen deficiency simultaneously promotes bone loss, muscle wasting, and visceral fat accumulation, creating synergistic effects that amplify individual system risks by 2-4 fold.
Neurological-psychological integration shows complex brain-hormone interactions:
-
Cognitive Function Changes
- Executive function: Working memory deficits during perimenopause
- Verbal memory: Word-finding difficulties in 40-60% of women
- Processing speed: Slowing during hormonal fluctuations
- Brain imaging: Hippocampal and prefrontal changes on MRI
- Neurotransmitters: Serotonin, dopamine system alterations
- Neuroprotection: Estrogen's role in Alzheimer's prevention under investigation
-
Sleep-Mood-Cognition Cycle
- Sleep fragmentation: Vasomotor symptoms disrupt REM sleep
- Mood instability: Sleep deprivation exacerbates hormonal mood effects
- Cognitive performance: Poor sleep amplifies memory complaints
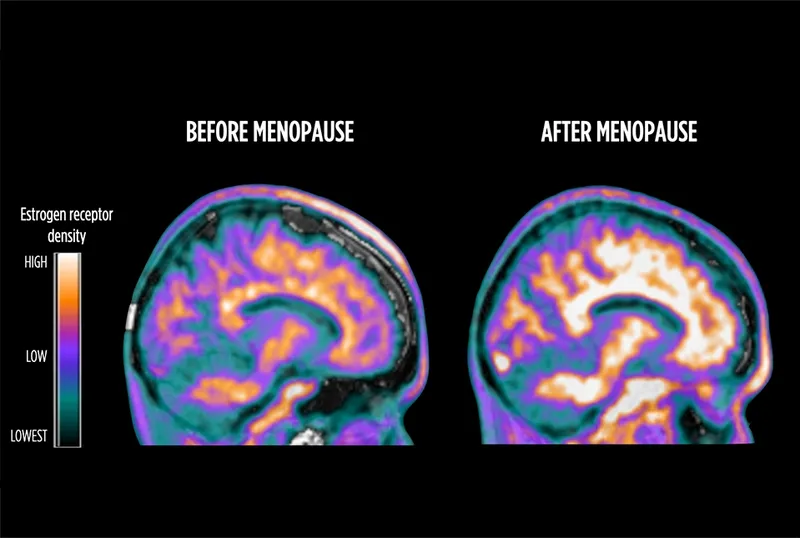
💡 Master This: Systems thinking in menopause management recognizes that treating vasomotor symptoms with hormone therapy simultaneously provides cardiovascular protection, bone preservation, and cognitive benefits during the critical window of early postmenopause.
Integrated management strategies address multiple systems simultaneously:
-
Hormone Therapy Benefits (Multi-system approach)
- Cardiovascular: 30-40% CHD reduction when started <10 years postmenopause
- Skeletal: 30-50% fracture reduction with continued use
- Cognitive: Potential protection when initiated early in transition
-
Lifestyle Integration
- Exercise prescription: Resistance training for bone/muscle, aerobic for cardiovascular
- Nutrition optimization: Calcium/Vitamin D for bone, Mediterranean diet for cardiovascular
- Stress management: Benefits across all systems through cortisol modulation
This systems integration approach transforms menopause management from symptom-focused treatment to comprehensive health optimization that addresses interconnected pathways and long-term wellness.
🔗 The Systemic Symphony: Multi-System Integration Mastery
🎯 The Clinical Mastery Arsenal: Rapid Reference Framework
Essential Clinical Arsenal provides immediate reference for high-stakes decisions:
📌 Remember: MASTERY - Menopause Assessment Symptoms Treatment Evidence Risks Years - 12 months amenorrhea >age 45 = menopause, FSH >30 confirms perimenopause, HRT within 10 years = optimal window
Critical Numbers for Immediate Recall:
-
Diagnostic Thresholds
- Menopause confirmation: 12 months amenorrhea
- FSH elevation: >25-30 IU/L (postmenopausal)
- Estradiol deficiency: <30 pg/mL (postmenopausal)
- Average menopause age: 51.4 years (range 45-55)
-
Treatment Efficacy Benchmarks
- HRT symptom reduction: 75-90% for vasomotor symptoms
- Non-hormonal options: 40-60% reduction maximum
- Lifestyle interventions: 20-40% improvement
- Time to HRT effect: 2-4 weeks for vasomotor relief
| Clinical Scenario | Immediate Action | Key Numbers | Red Flags | Next Steps |
|---|---|---|---|---|
| Age 52, hot flashes | Clinical diagnosis | No labs needed | Breast mass, bleeding | Symptom management |
| Age 42, amenorrhea | FSH + pregnancy test | FSH >40 = POI | Galactorrhea, headache | Full POI workup |
| Irregular bleeding | Exclude pregnancy | Age >45 + symptoms | Heavy bleeding >7 days | Endometrial assessment |
| HRT candidate | Risk assessment | <10% CVD risk ideal | VTE history, breast CA | Shared decision making |
| Severe symptoms | Rapid intervention | >7 hot flashes/day | Depression, suicidality | Multidisciplinary care |
Rapid Assessment Framework:
-
60-Second Menopause Assessment
- Age + amenorrhea duration = diagnostic probability
- Vasomotor symptom severity = treatment urgency
- Contraindication screening = therapy options
- Quality of life impact = intervention intensity
-
Treatment Decision Tree (<2 minutes)
- Mild symptoms → Lifestyle first
- Moderate-severe + HRT candidate → Hormone therapy
- HRT contraindicated → Non-hormonal pharmacotherapy
- Urogenital only → Local estrogen therapy
💡 Master This: Pattern recognition beats algorithmic thinking in complex cases. Experienced clinicians integrate multiple data points simultaneously to achieve diagnostic accuracy >95% without extensive testing in typical presentations.
High-Yield Clinical Pearls for immediate application:
- Perimenopausal bleeding: >7 days heavy flow or bleeding >21 days requires endometrial assessment
- Premature menopause: Age <40 mandates karyotype, autoimmune, and genetic testing
- HRT timing: Transdermal preferred for VTE risk, oral acceptable for low-risk patients
- Monitoring frequency: 3 months initial, 6-12 months ongoing, annual comprehensive
Emergency Situations requiring immediate action:
- Severe depression with suicidal ideation during perimenopause
- Acute cardiovascular events in HRT users
- Suspected breast cancer during hormone therapy
- Severe bleeding requiring urgent intervention
This clinical mastery framework transforms complex menopausal medicine into accessible, evidence-based tools that enable confident, efficient, and effective patient care across diverse clinical presentations and practice settings.
🎯 The Clinical Mastery Arsenal: Rapid Reference Framework
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more
Have doubts about this lesson?
Ask Rezzy, your AI Study Mate, to explain anything you didn't understand
Everything you need for NEET-PG prep
Get full Oncourse access with lessons, practice questions, flashcards and AI study tools.
Scan to download app




















