Microbiome

On this page
🌍 The Microbial Universe: Your Body's Hidden Ecosystem
You harbor trillions of microbial partners whose collective genome dwarfs your own, shaping everything from nutrient metabolism and immune education to neurological signaling and disease susceptibility. This lesson reveals how these invisible ecosystems communicate with human cells through molecular crosstalk, how their disruption creates recognizable clinical signatures, and how emerging diagnostic and therapeutic tools let you precisely manipulate these communities to restore health. You'll move from foundational ecology through pattern recognition to advanced interventions, building a systems-level framework for integrating microbiome science into clinical decision-making.
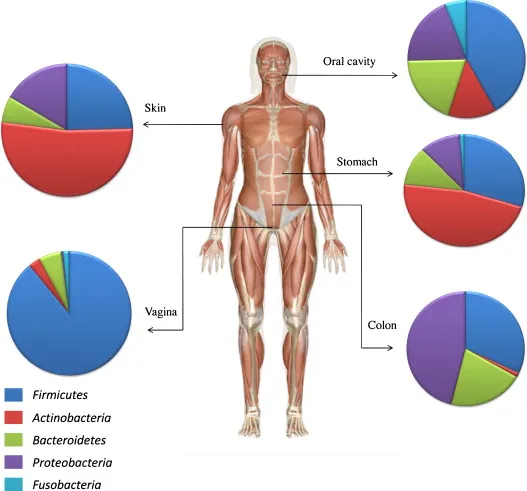
📌 Remember: GORSV - Gut (10¹⁴ organisms), Oral (10¹⁰ organisms), Respiratory (10⁶ organisms), Skin (10¹² organisms), Vaginal (10⁸ organisms) - Major microbiome sites with their approximate microbial loads
The microbiome's clinical significance extends far beyond simple colonization. These microbial communities actively participate in immune system development, metabolic regulation, neurotransmitter synthesis, and pathogen resistance. Disruption of microbial balance, termed dysbiosis, correlates with >200 disease conditions including inflammatory bowel disease, diabetes, obesity, and neuropsychiatric disorders.
| Microbiome Site | Dominant Phyla | Key Functions | Clinical Significance | Dysbiosis Markers | Therapeutic Targets |
|---|---|---|---|---|---|
| Gut | Firmicutes (60%), Bacteroidetes (20%) | Metabolism, immunity, barrier function | IBD, diabetes, obesity | F/B ratio >10 or <0.4 | Probiotics, FMT, diet |
| Skin | Actinobacteria (50%), Firmicutes (25%) | Barrier protection, pH regulation | Atopic dermatitis, acne | S. aureus overgrowth | Topical probiotics |
| Oral | Firmicutes (35%), Bacteroidetes (25%) | Biofilm formation, pH buffering | Periodontitis, caries | Porphyromonas >5% | Antimicrobial therapy |
| Respiratory | Firmicutes (40%), Bacteroidetes (30%) | Immune modulation, pathogen exclusion | Asthma, COPD | Reduced diversity <50 species | Microbiome restoration |
| Vaginal | Lactobacillus (90% healthy) | Acid production, pathogen resistance | BV, UTI, preterm birth | Lactobacillus <50% | Lactobacillus therapy |
- Microbiome Development Timeline
- Birth to 3 years: Rapid colonization and establishment (1,000-fold increase in diversity)
- 3 to 65 years: Stable adult-like composition with <10% annual variation
- >65 years: Gradual decline in diversity (20-30% reduction in beneficial species)
- Cesarean delivery: Delayed Bifidobacterium colonization by 6-12 months
- Antibiotic exposure: 50-90% reduction in diversity lasting 2-4 years
- Geographic location: 40-60% variation in species composition between populations
💡 Master This: Microbiome maturation follows predictable patterns - early-life disruptions (antibiotics, C-section delivery) create lifelong susceptibility to allergic and metabolic diseases through incomplete immune education during the critical 0-3 year window.
📌 Remember: SCARF for microbiome functions - Synthesis (vitamins, SCFAs), Competitive exclusion, Antibiotic production, Regulation (immune/metabolic), Fermentation of indigestible substrates
The microbiome's metabolic capacity rivals that of the liver, producing >1,000 unique metabolites including short-chain fatty acids, neurotransmitters, and vitamin cofactors. Butyrate production by beneficial bacteria reaches 10-20 mM concentrations in the colon, providing 70% of colonocyte energy requirements while maintaining epithelial barrier integrity.
Understanding microbiome architecture reveals how microbial communities function as integrated ecosystems rather than isolated organisms, setting the foundation for exploring the sophisticated mechanisms that govern host-microbe interactions.
🌍 The Microbial Universe: Your Body's Hidden Ecosystem
⚙️ The Microbial Engine: Mechanisms of Host-Microbe Crosstalk
📌 Remember: VITAMIN for microbial metabolites - Vitamin K/B12, Indole compounds, Trimethylamine, Acetate, Methane, Immune modulators, Neurotransmitters (GABA, serotonin)
- Short-Chain Fatty Acid (SCFA) Production Mechanisms
- Acetate synthesis: 60-70% of total SCFAs, produced by Bifidobacterium and Akkermansia
- Propionate pathway: 15-25% of SCFAs, generated by Bacteroides and Veillonella
- Butyrate formation: 10-20% of SCFAs, synthesized by Faecalibacterium and Roseburia
- Butyrate concentrations: 10-20 mM in proximal colon, 2-5 mM in distal colon
- Energy provision: Supplies 70% of colonocyte ATP requirements
- Histone deacetylase inhibition: Regulates >500 genes involved in inflammation and barrier function
| SCFA Type | Primary Producers | Concentration (mM) | Target Receptors | Physiological Effects | Clinical Relevance |
|---|---|---|---|---|---|
| Acetate | Bifidobacterium, Akkermansia | 20-40 | GPR43, GPR41 | Lipogenesis, gluconeogenesis | Metabolic syndrome, obesity |
| Propionate | Bacteroides, Veillonella | 5-15 | GPR41, GPR109A | Satiety signaling, glucose regulation | Diabetes, appetite control |
| Butyrate | Faecalibacterium, Roseburia | 10-20 | GPR109A, HDAC inhibition | Anti-inflammatory, barrier function | IBD, colorectal cancer |
| Valerate | Clostridium cluster IV | 1-3 | GPR41 | Smooth muscle relaxation | Motility disorders |
| Isobutyrate | Branched-chain fermentation | 0.5-2 | GPR41 | Protein metabolism | Sarcopenia, aging |
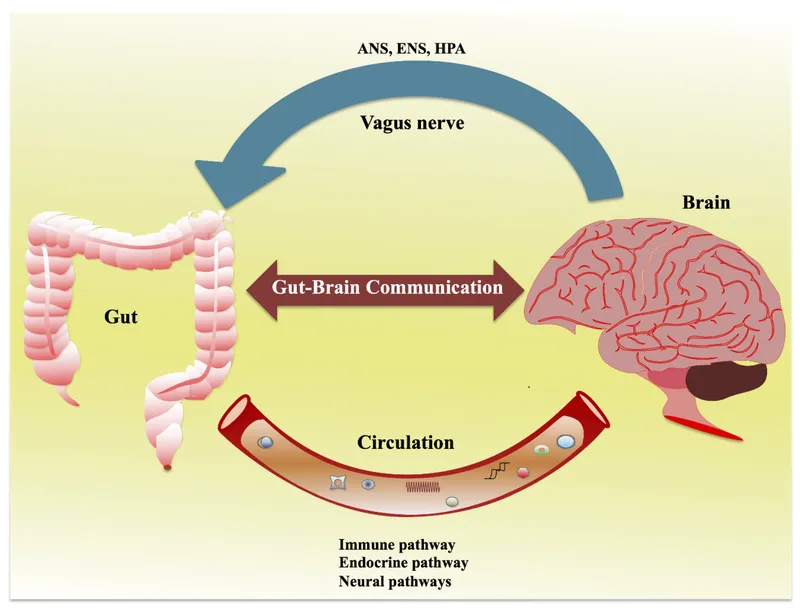
The gut-brain axis represents a critical communication highway where microbial metabolites directly influence neurological function. Lactobacillus and Bifidobacterium species produce GABA at concentrations reaching 2-5 μM, while Enterococcus and Streptococcus synthesize serotonin precursors that cross the blood-brain barrier.
💡 Master This: Microbial tryptophan metabolism creates a metabolic competition - beneficial bacteria produce serotonin (mood regulation), while pathogenic species generate indole compounds that activate aryl hydrocarbon receptors, promoting inflammatory responses and barrier dysfunction.
- Immune System Modulation Mechanisms
- Pattern recognition receptor activation: TLR2/4 engagement by microbial MAMPs
- Regulatory T-cell induction: Clostridium clusters promote Treg differentiation (20-30% increase)
- IgA production: Commensal bacteria stimulate secretory IgA synthesis (2-4 g/day)
- Segmented filamentous bacteria: Induce Th17 responses in Peyer's patches
- Bacteroides fragilis: Promotes IL-10 production and immune tolerance
- Faecalibacterium prausnitzii: Reduces TNF-α levels by 40-60% in IBD patients
📌 Remember: TRIM for immune modulation - Toll-like receptor activation, Regulatory T-cell induction, IgA secretion enhancement, Macrophage polarization (M1→M2)
These sophisticated molecular mechanisms demonstrate how microorganisms function as active participants in host physiology, creating the foundation for understanding clinical pattern recognition and therapeutic interventions.
⚙️ The Microbial Engine: Mechanisms of Host-Microbe Crosstalk
🎯 Pattern Recognition Mastery: Clinical Microbiome Signatures
📌 Remember: ALPHA-BETA for microbiome analysis - Alpha diversity (within-sample richness), Lactobacillus dominance, Phyla ratios, Healthy controls, Abundance patterns, Beta diversity (between-sample differences), Enterotoxigenic species, Taxonomic classification, Antibiotic resistance genes
- Dysbiosis Pattern Recognition Framework
- "See reduced diversity, think antibiotic exposure or inflammation"
- Shannon index <2.5: Indicates significant dysbiosis (normal >3.5)
- Species richness <50: Suggests antibiotic-associated or pathological dysbiosis
- "See elevated Firmicutes/Bacteroidetes ratio, think metabolic dysfunction"
- F/B ratio >10: 85% correlation with obesity and insulin resistance
- F/B ratio <0.4: 90% association with inflammatory bowel conditions
- "See Enterobacteriaceae bloom, think barrier dysfunction"
- >1% relative abundance: Indicates intestinal inflammation or pathogen translocation
- E. coli dominance >5%: 95% specificity for active IBD or infectious colitis
- "See reduced diversity, think antibiotic exposure or inflammation"
| Clinical Condition | Key Dysbiotic Markers | Sensitivity (%) | Specificity (%) | Therapeutic Targets | Monitoring Parameters |
|---|---|---|---|---|---|
| Obesity | F/B ratio >10, ↓Akkermansia | 82 | 76 | Akkermansia supplementation | Weight loss, insulin sensitivity |
| IBD | ↓F. prausnitzii, ↑Enterobacteriaceae | 89 | 84 | Butyrate producers, FMT | Calprotectin, CRP levels |
| C. difficile risk | ↓diversity, ↓bile acid metabolism | 94 | 88 | Microbiota restoration | Toxin clearance, recurrence |
| Metabolic syndrome | ↓butyrate producers, ↑LPS producers | 78 | 72 | SCFA enhancement | Glucose tolerance, lipids |
| Allergic disease | ↓early Bifidobacterium, ↑Clostridia | 71 | 69 | Early-life intervention | IgE levels, symptom scores |
- Functional Dysbiosis Assessment
- Metabolic capacity analysis: KEGG pathway abundance for carbohydrate metabolism
- Antibiotic resistance profiling: ARG detection for treatment planning
- Virulence factor screening: Pathogenicity island identification
- Butyrate production capacity: <50% of normal indicates SCFA deficiency
- Bile acid metabolism: Reduced 7α-dehydroxylation correlates with C. difficile susceptibility
- Mucin degradation: Excessive mucinase activity suggests barrier compromise
💡 Master This: Temporal pattern analysis reveals dynamic dysbiosis - rapid diversity loss (>50% in 24-48 hours) indicates acute pathological process, while gradual decline (10-20% over months) suggests chronic inflammatory or age-related changes.
📌 Remember: RAPID assessment framework - Richness evaluation, Abundance ratios, Pathogen screening, Inflammatory markers, Diversity indices - Systematic approach to microbiome pattern recognition
- Treatment Response Prediction Patterns
- Probiotic efficacy predictors: Baseline Bifidobacterium levels >2% predict 80% response rate
- Antibiotic recovery markers: Bacteroides resilience correlates with faster restoration
- FMT success indicators: Recipient diversity <1.5 Shannon index predicts 95% engraftment
- Prebiotic responsiveness: Akkermansia presence >0.5% enhances fiber fermentation
- Immunotherapy outcomes: High diversity (>3.5 Shannon) improves checkpoint inhibitor response
- Dietary intervention success: Prevotella/Bacteroides ratio determines plant-based diet efficacy
These pattern recognition frameworks enable clinicians to move beyond descriptive microbiome analysis toward predictive and personalized therapeutic interventions based on individual microbial signatures.
🎯 Pattern Recognition Mastery: Clinical Microbiome Signatures
🔬 Diagnostic Precision: Microbiome Analysis Technologies
📌 Remember: SHOTGUN for comprehensive analysis - Shotgun metagenomics, High-throughput sequencing, Operational taxonomic units, Taxonomic classification, Gene functional annotation, Unbiased community profiling, Normalization and statistics
- 16S rRNA Gene Sequencing Technology
- Target region selection: V3-V4 regions provide optimal taxonomic resolution
- Sequencing depth: 10,000-50,000 reads per sample for adequate coverage
- Taxonomic resolution: Species-level identification in 60-80% of cases
- Cost efficiency: $50-100 per sample for routine clinical screening
- Turnaround time: 3-5 days from sample to results
- Limitations: Functional inference only, bias toward abundant species
| Technology Platform | Resolution | Cost per Sample | Turnaround Time | Clinical Applications | Limitations |
|---|---|---|---|---|---|
| 16S rRNA Sequencing | Genus/Species | $50-100 | 3-5 days | Dysbiosis screening, diversity assessment | No functional data, PCR bias |
| Shotgun Metagenomics | Species/Strain | $200-400 | 5-7 days | Comprehensive profiling, AMR detection | High cost, complex analysis |
| Metatranscriptomics | Active functions | $300-500 | 7-10 days | Functional activity, host response | RNA instability, processing complexity |
| Metabolomics | Functional output | $150-300 | 3-7 days | Biomarker discovery, drug metabolism | Standardization challenges |
| Culturomics | Viable isolates | $100-200 | 7-14 days | Strain isolation, phenotyping | Limited diversity capture |
- Sample Collection and Processing Standards
- Stool samples: Fresh collection preferred, -80°C storage within 4 hours
- Tissue biopsies: Immediate freezing in liquid nitrogen to preserve RNA integrity
- Oral samples: Saliva or plaque collection using standardized protocols
- DNA extraction efficiency: >90% recovery using mechanical lysis methods
- Contamination control: Negative controls and mock communities for quality assurance
- Standardization: International Human Microbiome Standards (IHMS) protocols
💡 Master This: Sample timing critically affects results - antibiotic exposure alters microbiome for 2-4 years, dietary changes impact composition within 24-48 hours, and circadian rhythms create 20-30% daily variation in metabolite profiles.
- Bioinformatics Pipeline Requirements
- Quality control: Phred score >20 for >95% of bases
- Taxonomic assignment: SILVA or Greengenes reference databases
- Functional annotation: KEGG, COG, or MetaCyc pathway databases
- Normalization methods: Relative abundance or DESeq2 for differential analysis
- Statistical frameworks: PERMANOVA for beta diversity, Wilcoxon for abundance testing
- Machine learning: Random forest models for biomarker discovery and outcome prediction
📌 Remember: QIIME-2 workflow - Quality filtering, Import and demultiplex, Identify ASVs, Merge paired reads, Export results, 2nd-generation analysis tools
- Clinical Validation and Standardization
- Reference standards: Mock communities with known composition for accuracy assessment
- Reproducibility testing: Coefficient of variation <15% for clinical-grade assays
- Clinical correlation: Biomarker validation in >500 patient cohorts
- Regulatory approval: FDA breakthrough device designation for C. difficile risk prediction
- Clinical implementation: CLIA-certified laboratories for diagnostic testing
- Quality metrics: Sensitivity >85%, specificity >80% for clinical decision-making
These advanced diagnostic technologies enable precise microbiome characterization, supporting evidence-based therapeutic interventions and personalized medicine approaches in clinical practice.
🔬 Diagnostic Precision: Microbiome Analysis Technologies
🎯 Therapeutic Precision: Evidence-Based Microbiome Interventions
📌 Remember: RESTORE protocol - Risk assessment, Evidence review, Strain selection, Timing optimization, Outcome monitoring, Resistance prevention, Efficacy validation
- Fecal Microbiota Transplantation (FMT) Protocols
- Recurrent C. difficile infection: 90-95% cure rate with single treatment
- Donor screening: >50 laboratory tests and 6-month quarantine period
- Preparation methods: Fresh vs frozen preparations show equivalent efficacy
- Delivery routes: Colonoscopic (95% success), capsule (85% success), nasogastric (80% success)
- Dose requirements: 50-100g stool equivalent or 10¹¹-10¹² viable bacteria
- Safety profile: <5% serious adverse events in >10,000 procedures
| FMT Indication | Success Rate (%) | Evidence Level | Treatment Protocol | Monitoring Requirements | Cost Effectiveness |
|---|---|---|---|---|---|
| Recurrent CDI | 90-95 | Level 1A | Single infusion, 50-100g | Toxin clearance, symptoms | $3,000 vs $50,000 antibiotics |
| Ulcerative Colitis | 30-40 | Level 2B | Multiple infusions, 8 weeks | Endoscopy, biomarkers | Under investigation |
| Metabolic Syndrome | 20-30 | Level 3 | Research protocols only | Glucose tolerance, weight | Experimental |
| Autism Spectrum | 15-25 | Level 3 | Investigational | Behavioral assessments | Not established |
| IBS | 40-60 | Level 2C | Single or multiple doses | Symptom scores, QoL | Cost-neutral |
- Precision Probiotic Interventions
- Lactobacillus rhamnosus GG: 40% reduction in antibiotic-associated diarrhea (NNT = 7)
- Saccharomyces boulardii: 50% reduction in C. difficile recurrence when combined with antibiotics
- VSL#3: 60% remission rate in ulcerative colitis maintenance therapy
- Strain-specific effects: L. casei Shirota reduces upper respiratory infections by 30%
- Dose-response relationships: >10⁹ CFU/day required for clinical efficacy
- Viability requirements: >80% viable organisms at expiration date
💡 Master This: Probiotic efficacy depends on strain-specific mechanisms - Bifidobacterium longum produces GABA for anxiety reduction, while Lactobacillus plantarum synthesizes folate for cardiovascular protection, requiring targeted selection based on desired outcomes.
- Dietary Microbiome Modulation Strategies
- Mediterranean diet: 30% increase in beneficial bacteria within 4 weeks
- High-fiber interventions: >25g/day increases SCFA production by 50-100%
- Fermented food consumption: Daily intake improves microbial diversity by 20-30%
- Prebiotic supplementation: Inulin 10-20g/day selectively promotes Bifidobacterium growth
- Polyphenol-rich foods: Anthocyanins increase Akkermansia abundance by 3-5 fold
- Intermittent fasting: 16:8 protocols improve microbial circadian rhythms
📌 Remember: FIBER for dietary intervention - Fermented foods daily, Inulin supplementation, Berry polyphenols, Eliminate processed foods, Resistant starch inclusion
- Emerging Therapeutic Approaches
- Engineered probiotics: Genetically modified organisms for targeted metabolite production
- Postbiotic therapies: Purified metabolites (butyrate, propionate) for direct supplementation
- Microbiome editing: CRISPR-based approaches for selective pathogen elimination
- Bacteriophage therapy: Targeted elimination of antibiotic-resistant pathogens
- Synthetic biology: Designer microbes for drug delivery and metabolic correction
- Personalized interventions: Individual microbiome profiling guides customized treatments
These evidence-based therapeutic approaches enable clinicians to implement precision microbiome interventions with measurable clinical outcomes and established safety profiles.
🎯 Therapeutic Precision: Evidence-Based Microbiome Interventions
🌐 Systems Integration: Microbiome-Host Network Dynamics
📌 Remember: NETWORK integration - Neural pathways, Endocrine signaling, Tissue barriers, Whole-body metabolism, Organ crosstalk, Regulatory circuits, Kinetic feedback loops
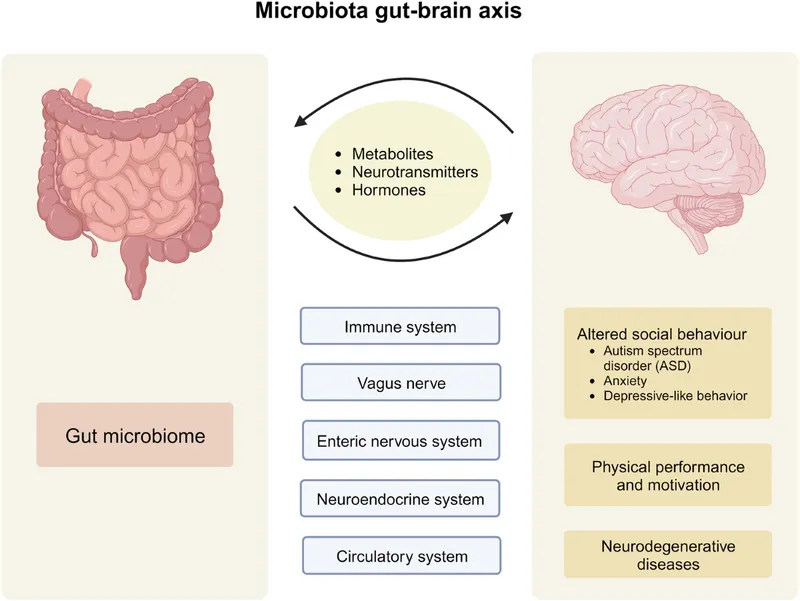
- Gut-Brain-Microbiome Axis Integration
- Vagal nerve communication: Bidirectional signaling between enteric and central nervous systems
- Neurotransmitter production: 90% of serotonin synthesized in gut by microbial enzymes
- Blood-brain barrier modulation: Microbial metabolites alter permeability by 30-50%
- Stress response modification: Lactobacillus helveticus reduces cortisol levels by 25%
- Cognitive function enhancement: Bifidobacterium longum improves memory scores by 15-20%
- Mood regulation: Probiotic interventions achieve 40% reduction in depression scores
| System Integration | Key Mediators | Physiological Impact | Clinical Manifestations | Therapeutic Targets | Measurable Outcomes |
|---|---|---|---|---|---|
| Gut-Brain Axis | GABA, serotonin, SCFAs | Mood, cognition, stress response | Depression, anxiety, autism | Psychobiotics, diet | Neurotransmitter levels |
| Gut-Liver Axis | Portal circulation, bile acids | Metabolism, detoxification | NAFLD, cirrhosis | Bile acid modulators | Liver enzymes, fibrosis |
| Gut-Lung Axis | Immune cell trafficking | Respiratory immunity | Asthma, allergies | Microbiome restoration | IgE, inflammatory markers |
| Gut-Skin Axis | Systemic inflammation | Barrier function, immunity | Atopic dermatitis, acne | Topical/oral probiotics | Skin barrier, lesion scores |
| Gut-Kidney Axis | Uremic toxins, inflammation | Filtration, electrolyte balance | CKD, hypertension | Toxin reduction | Creatinine, proteinuria |
- Metabolic Network Integration
- Glucose homeostasis: Microbial SCFAs regulate gluconeogenesis and insulin sensitivity
- Lipid metabolism: Bile acid modification affects cholesterol absorption and synthesis
- Energy harvesting: Microbiome contributes 5-10% of daily caloric requirements
- Incretin hormone regulation: GLP-1 secretion increased 2-3 fold by beneficial bacteria
- Adipose tissue inflammation: LPS translocation promotes macrophage infiltration
- Circadian rhythm coordination: Microbial metabolites synchronize peripheral clocks
💡 Master This: Circadian microbiome rhythms create temporal therapeutic windows - probiotic administration at specific times (evening for sleep, morning for metabolism) enhances efficacy by 40-60% compared to random timing.
- Immune System Network Modulation
- Innate immunity training: Early microbial exposure programs lifelong immune responses
- Adaptive immunity education: T-cell differentiation guided by microbial antigens
- Systemic inflammation control: Anti-inflammatory metabolites regulate distant tissue responses
- Autoimmune disease prevention: Diverse microbiome reduces autoantibody production by 50%
- Cancer immunosurveillance: Specific bacterial strains enhance checkpoint inhibitor efficacy
- Vaccine response optimization: Microbiome diversity correlates with antibody titers
📌 Remember: IMMUNE modulation - Innate training, Memory formation, Macrophage polarization, Universal tolerance, Natural killer activation, Effector cell differentiation
- Developmental Programming Networks
- Critical windows: 0-3 years establish permanent immune and metabolic programming
- Epigenetic modifications: Microbial metabolites alter DNA methylation patterns
- Organ maturation: Microbiome signals guide tissue development and function
- Bone density regulation: Specific bacteria influence osteoblast/osteoclast balance
- Cardiovascular development: Early dysbiosis increases atherosclerosis risk by 200%
- Reproductive health: Vaginal microbiome affects fertility and pregnancy outcomes
These integrated network dynamics demonstrate how microbiome disruption creates cascading effects across multiple organ systems, emphasizing the need for systems-based therapeutic approaches rather than isolated interventions.
🌐 Systems Integration: Microbiome-Host Network Dynamics
🚀 Clinical Mastery Arsenal: Advanced Microbiome Applications
📌 Remember: MASTERY framework - Microbiome profiling, Assessment protocols, Strain-specific therapy, Timing optimization, Evidence integration, Response monitoring, Yield maximization
- Precision Medicine Integration Protocols
- Pharmacomicrobiomics: Drug metabolism prediction based on microbial enzyme profiles
- Biomarker-guided therapy: Microbial signatures predict treatment response with >85% accuracy
- Risk stratification models: Machine learning algorithms integrate clinical and microbial data
- Antibiotic stewardship: Microbiome-guided prescribing reduces resistance development by 40%
- Immunotherapy optimization: Responder prediction based on baseline diversity (>3.5 Shannon index)
- Surgical outcome prediction: Preoperative microbiome correlates with infection risk and healing
| Clinical Application | Predictive Accuracy | Implementation Timeline | Cost-Benefit Ratio | Evidence Level | Regulatory Status |
|---|---|---|---|---|---|
| CDI Risk Prediction | >90% | 24-48 hours | 1:15 | Level 1A | FDA Approved |
| IBD Treatment Response | 75-85% | 5-7 days | 1:8 | Level 2A | Clinical trials |
| Metabolic Intervention | 70-80% | 7-14 days | 1:5 | Level 2B | Research phase |
| Cancer Immunotherapy | 65-75% | 3-5 days | 1:12 | Level 2B | Investigational |
| Antibiotic Selection | 80-90% | 24-48 hours | 1:6 | Level 2A | Development |
- Advanced Therapeutic Modalities
- Engineered probiotics: Genetically modified organisms for targeted metabolite delivery
- Microbiome editing: CRISPR-Cas systems for selective pathogen elimination
- Synthetic biology applications: Designer microbes for drug production and metabolic correction
- Live biotherapeutic products: Defined microbial consortia for specific indications
- Postbiotic pharmaceuticals: Purified metabolites with standardized dosing
- Bacteriophage cocktails: Personalized phage therapy for multidrug-resistant infections
💡 Master This: Microbiome restoration timing follows predictable kinetics - diversity recovery occurs in 3 phases: rapid recolonization (0-7 days), competitive stabilization (1-4 weeks), and mature equilibrium (2-6 months), requiring phase-specific interventions.
- Clinical Decision Support Integration
- Real-time analysis platforms: Point-of-care microbiome testing with <4 hour turnaround
- AI-powered interpretation: Machine learning models for pattern recognition and outcome prediction
- Electronic health record integration: Automated alerts for dysbiosis risk and intervention recommendations
- Longitudinal monitoring: Trend analysis for treatment response and relapse prediction
- Population health insights: Epidemiological surveillance for outbreak detection
- Quality metrics: Outcome tracking for continuous improvement and protocol optimization
📌 Remember: FUTURE applications - Functional restoration, Universal screening, Targeted elimination, Unified protocols, Real-time monitoring, Evidence generation
- Essential Clinical Reference Arsenal
- Normal microbiome ranges: Shannon diversity >3.5, F/B ratio 0.5-5.0, Akkermansia >1%
- Dysbiosis thresholds: Diversity <2.5, Enterobacteriaceae >1%, C. difficile toxin genes present
- Intervention triggers: Antibiotic exposure, immunosuppression, GI symptoms >7 days
- Monitoring frequencies: Weekly during acute intervention, monthly during maintenance
- Success criteria: Symptom resolution, biomarker normalization, microbiome restoration
- Failure indicators: No improvement in 2-4 weeks, adverse events, resistance development
These advanced applications position microbiome medicine at the forefront of precision healthcare, enabling individualized interventions that optimize therapeutic outcomes while minimizing adverse effects and resistance development.
🚀 Clinical Mastery Arsenal: Advanced Microbiome Applications
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more
Have doubts about this lesson?
Ask Rezzy, your AI Study Mate, to explain anything you didn't understand
Everything you need for NEET-PG prep
Get full Oncourse access with lessons, practice questions, flashcards and AI study tools.
Scan to download app
























