Immunology

On this page
🧬 Immunological Memory: The Body's Elite Intelligence Network
Your immune system doesn't just fight infections-it remembers them, building a sophisticated intelligence network that transforms every pathogen encounter into lasting protection. This lesson reveals how your body creates, maintains, and deploys immunological memory through specialized cellular machinery, distributed surveillance networks, and rapid-response protocols that make second infections vastly different from first ones. You'll trace the journey from initial antigen exposure through memory cell formation, long-term maintenance, and the coordinated systems that turn past battles into future victories, ultimately connecting these mechanisms to the vaccines and therapies that harness memory for clinical benefit.

⭐ Clinical Pearl: Secondary immune responses occur 100-1000x faster than primary responses, with antibody levels reaching peak concentrations in 3-5 days versus 10-14 days for initial exposure.
The immune system's memory function relies on specialized long-lived cells that persist for 20-50 years in humans, maintaining surveillance through distinct anatomical compartments. Memory B cells circulate through secondary lymphoid organs, while memory T cells establish residence in both lymphoid tissues and peripheral sites of previous antigen encounter.
📌 Remember: SLIM Memory Characteristics - Specific (antigen-specific recognition), Long-lasting (decades of persistence), Immediate (rapid secondary responses), Magnified (enhanced magnitude compared to primary responses)
| Memory Cell Type | Location | Lifespan | Response Time | Key Markers | Clinical Significance |
|---|---|---|---|---|---|
| Central Memory T cells | Secondary lymphoid organs | 20-30 years | 6-12 hours | CCR7+, CD62L+ | Vaccine efficacy, transplant rejection |
| Effector Memory T cells | Peripheral tissues | 5-10 years | 1-3 hours | CCR7-, CD62L- | Local tissue immunity, autoimmunity |
| Memory B cells | Bone marrow, spleen | 30-50 years | 24-48 hours | CD27+, IgG+ | Antibody recall responses |
| Plasma cells | Bone marrow niches | Months to years | Immediate | CD138+, BLIMP1+ | Baseline antibody maintenance |
| Tissue-Resident Memory | Barrier sites | Years to decades | Minutes | CD69+, CD103+ | Mucosal immunity, skin protection |
💡 Master This: Memory formation occurs in 15-20% of activated lymphocytes during primary responses, with the remainder becoming short-lived effector cells that eliminate the immediate threat.
Connect these foundational memory principles through cellular differentiation mechanisms to understand how immune memory shapes clinical outcomes across diverse disease states.
🧬 Immunological Memory: The Body's Elite Intelligence Network
⚡ Memory Formation Machinery: The Cellular Decision Engine

The asymmetric cell division model explains how single activated T cells generate both effector and memory progeny. During the first division after activation, daughter cells inherit different amounts of key transcription factors, with TCF1-high cells becoming memory precursors and T-BET-high cells becoming effectors.
📌 Remember: TEAM Memory Signals - TCF1 (memory transcription factor), Eomes (memory maintenance), Asymmetric division (fate determination), MTOR inhibition (memory promotion)
-
Memory-Promoting Factors
- IL-7 and IL-15: Homeostatic cytokines supporting memory survival
- Low mTOR activity: Metabolic state favoring memory formation (<30% of effector levels)
- FOXO1 activation: Promotes memory transcriptional programs
- Upregulates BCL2 anti-apoptotic proteins
- Enhances fatty acid oxidation for long-term survival
- Maintains stem-like characteristics in memory precursors
-
Effector-Promoting Factors
- High antigen load: Drives terminal differentiation (>10^6 peptide-MHC complexes)
- Inflammatory cytokines: IL-12, IFN-γ promote effector commitment
- mTOR hyperactivation: Supports rapid proliferation and function
- Increases glycolysis by 5-10 fold
- Drives protein synthesis for cytokine production
- Shortens telomeres through rapid division cycles
⭐ Clinical Pearl: Memory T cells maintain glucose independence, utilizing fatty acid oxidation for energy, explaining their survival during metabolic stress conditions like sepsis or malnutrition.
Memory B cell formation follows distinct kinetics, with early memory cells emerging by day 7 and late memory cells developing after 2-3 weeks of germinal center reactions. Early memory cells provide rapid but lower-affinity responses, while late memory cells offer high-affinity, class-switched antibodies.
The selection stringency in germinal centers determines memory quality. High-affinity B cells (Kd < 10^-9 M) preferentially survive selection, while lower-affinity clones undergo apoptosis. This process requires CD40-CD40L interactions and IL-21 signaling for optimal memory generation.
💡 Master This: Germinal center reactions lasting >3 weeks produce higher-quality memory responses, explaining why some vaccines require extended prime-boost intervals for optimal efficacy.
These molecular decision mechanisms connect through metabolic reprogramming pathways to establish the distinct functional properties that define immunological memory.
⚡ Memory Formation Machinery: The Cellular Decision Engine
🎯 Memory Surveillance Networks: The Distributed Defense Grid

-
Central Memory Surveillance Hub
- Location: Secondary lymphoid organs (spleen, lymph nodes)
- Cell density: 10^6-10^7 cells per lymph node
- Response characteristics: High proliferative capacity, systemic mobilization
- Doubling time: 8-12 hours upon reactivation
- Migration speed: 10-15 μm/minute through lymphoid tissues
- Cytokine production: Delayed but sustained (>72 hours)
-
Effector Memory Patrol Units
- Location: Blood, spleen, non-lymphoid tissues
- Circulation patterns: 2-4 hour blood transit times
- Immediate functions: Rapid cytokine release, direct cytotoxicity
- IFN-γ production: Within 2-4 hours of reactivation
- Perforin release: <30 minutes for CD8+ memory cells
- Chemokine secretion: Recruits additional immune cells
📌 Remember: PATROL Memory Distribution - Peripheral (effector memory), Anatomical barriers (tissue-resident), Trafficking (central memory), Rapid response (immediate function), Organized compartments (distinct locations), Long-term positioning (stable residence)
| Memory Subset | Tissue Location | Response Time | Key Function | Maintenance Signals |
|---|---|---|---|---|
| Central Memory (TCM) | Lymph nodes, spleen | 6-12 hours | Proliferation, systemic immunity | IL-7, IL-15, CCR7 |
| Effector Memory (TEM) | Blood, peripheral tissues | 1-3 hours | Immediate effector function | IL-15, antigen-independent |
| Tissue-Resident (TRM) | Skin, lung, gut, brain | <1 hour | Local barrier protection | TGF-β, IL-33, tissue factors |
| Stem Cell Memory (TSCM) | Bone marrow, lymph nodes | 12-24 hours | Self-renewal, long-term maintenance | Wnt signaling, Notch |
| Virtual Memory | Liver, bone marrow | 2-6 hours | Innate-like responses | IL-4, homeostatic proliferation |
⭐ Clinical Pearl: TRM cells in human skin persist for >20 years after smallpox vaccination, providing localized immunity that explains the superior protection of scarification versus injection routes.
The positioning signals that establish memory cell residence involve tissue-specific factors. TGF-β promotes TRM formation in epithelial barriers, while IL-33 maintains memory cells in adipose tissues. S1P receptor downregulation prevents tissue egress, creating stable resident populations.
Memory B cells establish distinct surveillance zones optimized for antigen capture. Marginal zone memory B cells intercept blood-borne antigens with >90% efficiency, while bone marrow memory cells provide long-term antibody production through plasma cell differentiation.
💡 Master This: The anatomical positioning of memory cells determines response kinetics - tissue-resident cells provide immediate local protection, while central memory cells generate sustained systemic responses requiring 6-12 hours for full mobilization.
These surveillance networks connect through chemokine-guided recruitment mechanisms to coordinate rapid and sustained memory responses across multiple tissue compartments.
🎯 Memory Surveillance Networks: The Distributed Defense Grid
🔍 Memory Response Dynamics: The Accelerated Defense Protocol
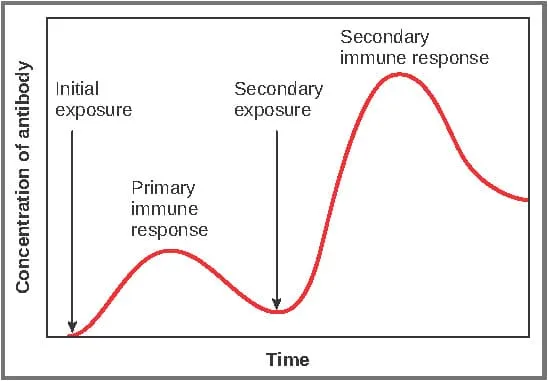
-
Primary Response Timeline
- Days 0-3: Antigen recognition and initial activation
- Days 4-7: Clonal expansion begins (2-3 doublings)
- Days 8-14: Peak effector function and antibody production
- Days 15-21: Contraction phase (90-95% cell death)
- Antibody levels: Peak at 10^2-10^3 μg/ml
- Affinity maturation: 10-100 fold improvement
- Memory formation: 5-15% of activated cells survive
-
Secondary Response Acceleration
- Hours 1-6: Memory cell reactivation and immediate function
- Days 1-3: Rapid proliferation (4-6 doublings)
- Days 3-5: Peak antibody production (10-100x higher levels)
- Days 5-7: Enhanced effector functions and tissue protection
- Response magnitude: 100-1000 fold greater than primary
- Antibody affinity: Pre-selected high-affinity clones
- Tissue penetration: Enhanced vascular permeability
📌 Remember: RAPID Memory Advantages - Recognition (immediate antigen binding), Amplified (100-1000x magnitude), Pre-selected (high affinity), Immediate (hours not days), Durable (sustained protection)
| Response Parameter | Primary Response | Secondary Response | Fold Improvement | Clinical Significance |
|---|---|---|---|---|
| Onset Time | 7-14 days | 1-3 days | 5-10x faster | Prevents disease establishment |
| Peak Magnitude | Baseline | 100-1000x higher | 100-1000x | Sterilizing immunity possible |
| Antibody Affinity | Low to moderate | High (pre-selected) | 10-100x | Neutralizes variants |
| Duration | Weeks to months | Months to years | 5-50x longer | Long-term protection |
| Tissue Penetration | Limited | Enhanced | 10-50x better | Mucosal immunity |
⭐ Clinical Pearl: Memory T cells produce IFN-γ within 2-4 hours of reactivation compared to 24-48 hours for naive cells, explaining the rapid viral clearance observed in breakthrough infections among vaccinated individuals.
Antibody recall responses demonstrate the most dramatic kinetic improvements. Memory B cells bypass the germinal center reaction, directly differentiating into plasma cells within 24-48 hours. Pre-existing bone marrow plasma cells immediately increase antibody production upon cytokine stimulation.
The quality differences in memory responses extend beyond speed and magnitude. Memory-derived antibodies show broader cross-reactivity due to somatic hypermutation, enhanced complement fixation through class switching, and improved tissue penetration via transcytosis mechanisms.
💡 Master This: Heterologous immunity allows memory cells specific for one pathogen to provide partial protection against related organisms, explaining why seasonal influenza vaccination reduces COVID-19 severity in some populations.
- Cross-Protective Mechanisms
- Molecular mimicry: Shared epitopes between pathogens (20-30% sequence homology)
- Bystander activation: Cytokine-mediated enhancement of unrelated memory cells
- Trained immunity: Epigenetic reprogramming of innate immune cells
- Duration: 3-12 months for monocyte/macrophage training
- Magnitude: 2-5 fold enhanced cytokine production
- Specificity: Broad-spectrum antimicrobial enhancement
These accelerated response dynamics connect through metabolic reprogramming mechanisms to sustain enhanced immune function during memory recall responses.
🔍 Memory Response Dynamics: The Accelerated Defense Protocol
⚖️ Memory Maintenance Systems: The Longevity Infrastructure

-
Homeostatic Maintenance Signals
- IL-7 signaling: Essential for central memory survival
- Receptor expression: IL-7Rα (CD127) high on memory cells
- Survival duration: >6 months without antigen stimulation
- Metabolic support: Maintains glucose uptake and fatty acid oxidation
- IL-15 signaling: Critical for effector memory and TRM maintenance
- Trans-presentation: Dendritic cells provide IL-15/IL-15Rα complexes
- Proliferation rate: Slow homeostatic division every 30-60 days
- Tissue residence: Supports TRM longevity in barrier organs
- IL-7 signaling: Essential for central memory survival
-
Self-Renewal Mechanisms
- Asymmetric division: Maintains stem-like memory precursors
- Telomere maintenance: Telomerase activity in memory subsets
- Autophagy programs: mTOR-independent survival pathways
- Mitochondrial quality: Enhanced mitophagy removes damaged organelles
- Protein homeostasis: Chaperone upregulation prevents aggregation
- Stress resistance: Heat shock proteins maintain function
📌 Remember: SURVIVE Memory Maintenance - Signals (IL-7, IL-15), Upregulated autophagy, Renewal (asymmetric division), Viable metabolism (fatty acids), Independent (antigen-free), Very long-lived (decades), Environmental adaptation
| Maintenance Factor | Memory Subset | Half-life | Mechanism | Clinical Relevance |
|---|---|---|---|---|
| IL-7 | Central Memory | 20-30 years | JAK/STAT signaling, Bcl-2 upregulation | Vaccine longevity |
| IL-15 | Effector Memory, TRM | 5-15 years | mTOR activation, metabolic support | Tissue immunity |
| Antigen persistence | All subsets | Variable | Low-level TCR stimulation | Chronic infections |
| Cross-reactivity | Heterologous memory | 10-20 years | Molecular mimicry, bystander activation | Broad protection |
| Stem cell renewal | Memory precursors | >50 years | Wnt signaling, self-renewal | Long-term immunity |
⭐ Clinical Pearl: Memory T cell numbers decline by approximately 5-8% per decade after age 40, explaining increased susceptibility to infections and reduced vaccine responses in elderly populations.
Memory B cell maintenance involves distinct mechanisms centered on bone marrow niches and splenic architecture. Long-lived plasma cells occupy specialized survival niches with CXCL12-producing stromal cells, IL-6 availability, and contact-dependent survival signals.

- Plasma Cell Longevity Factors
- Niche competition: Limited survival slots (~10^6 cells in human bone marrow)
- Affinity-based selection: High-affinity clones preferentially survive
- Metabolic adaptation: Unfolded protein response manages antibody production
- ER stress tolerance: XBP1 and ATF6 activation
- Protein folding: Enhanced chaperone expression
- Secretory capacity: >1000 antibodies per second per cell
The competition model explains memory maintenance dynamics. New memory cells must compete with existing populations for survival niches and maintenance signals. High-affinity, recently activated memory cells can displace older, lower-affinity populations.
💡 Master This: Original antigenic sin occurs when pre-existing memory cells outcompete new responses to variant antigens, explaining suboptimal immunity to influenza variants and the need for updated vaccine formulations.
These maintenance systems connect through epigenetic stability mechanisms to preserve memory cell identity and function across decades of environmental exposure.
⚖️ Memory Maintenance Systems: The Longevity Infrastructure
🔗 Memory Integration Networks: The Immune Coordination Matrix
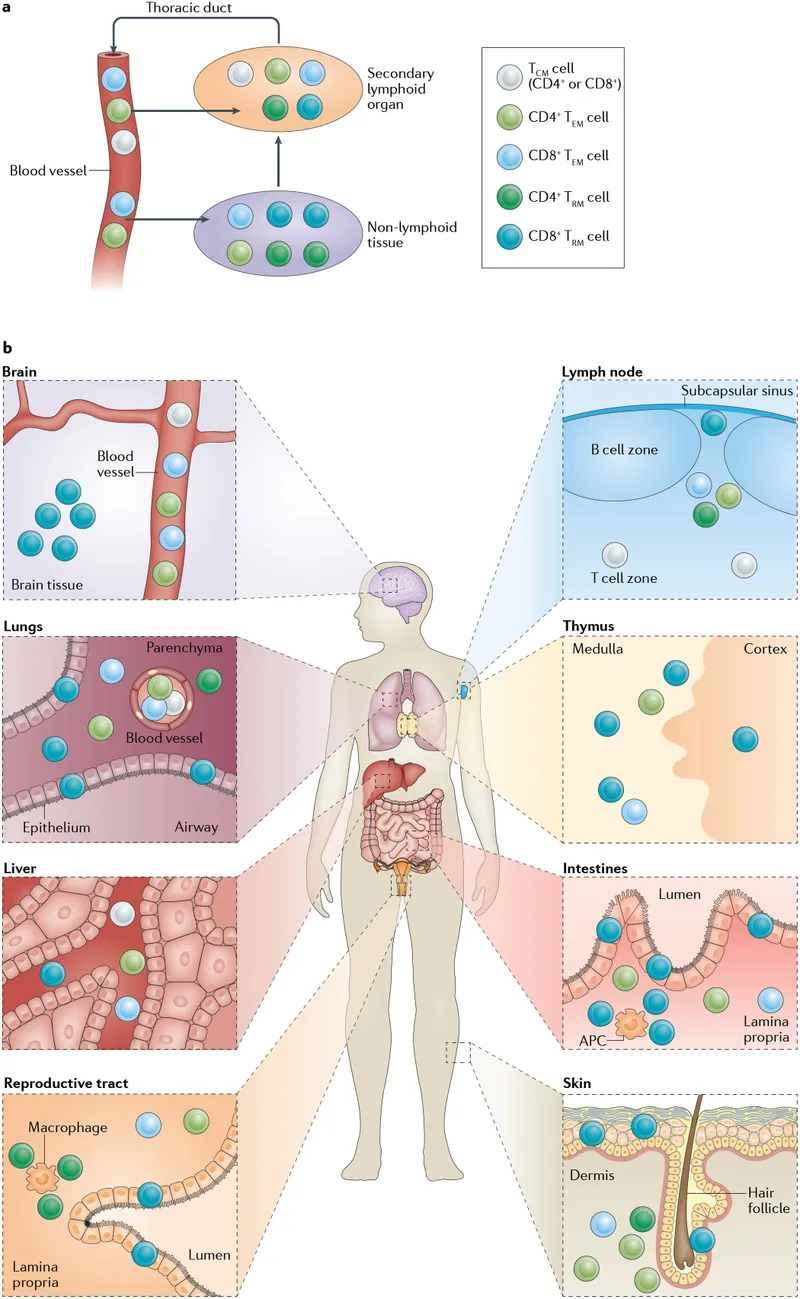
-
Multi-Compartment Integration
- Tissue-to-Lymphoid Communication
- Chemokine gradients: CCL19/CCL21 guide cell trafficking
- Antigen transport: Dendritic cells carry tissue antigens to lymph nodes
- Cytokine networks: Type I interferons coordinate distant responses
- Lymphoid-to-Tissue Mobilization
- Activation signals: S1P receptor modulation controls egress
- Homing receptors: Tissue-specific integrin and selectin expression
- Recruitment kinetics: Peak mobilization within 12-24 hours
- Tissue-to-Lymphoid Communication
-
Cross-Memory Interactions
- B-T Cell Cooperation: Memory helper T cells enhance antibody recall
- CD4-CD8 Coordination: Helper cells optimize cytotoxic memory responses
- Innate-Adaptive Integration: Memory cells enhance innate immune training
- Cytokine priming: Memory-derived IFN-γ activates macrophages
- Complement enhancement: Antibodies improve C3b deposition
- NK cell education: MHC-I interactions refine NK specificity
📌 Remember: NETWORK Memory Integration - Nodes (anatomical compartments), Effector coordination (multiple cell types), Trafficking (guided migration), Whole-body (systemic responses), Orchestrated (timed activation), Reciprocal (bidirectional communication), Kinetic synchronization
| Integration Level | Components | Coordination Signals | Response Time | Clinical Impact |
|---|---|---|---|---|
| Local Tissue | TRM, resident APCs | Chemokines, alarmins | <1 hour | Immediate protection |
| Regional Lymphoid | Central memory, B cells | CCR7, S1P | 6-12 hours | Amplified responses |
| Systemic Circulation | Effector memory, antibodies | Selectins, integrins | 12-24 hours | Body-wide immunity |
| Cross-Tissue | Multiple compartments | Cytokine networks | 24-48 hours | Coordinated protection |
| Memory-Innate | Trained immunity, memory cells | Type I IFNs, IL-1β | 1-6 hours | Enhanced surveillance |
⭐ Clinical Pearl: Heterologous prime-boost vaccination strategies exploit memory integration networks, with different vaccine platforms activating complementary memory pathways to achieve >95% protective efficacy against complex pathogens.
Memory B-T cell cooperation demonstrates the most sophisticated integration mechanisms. Memory helper T cells provide cognate help to memory B cells through CD40L-CD40 interactions, IL-21 secretion, and germinal center reformation for affinity maturation.
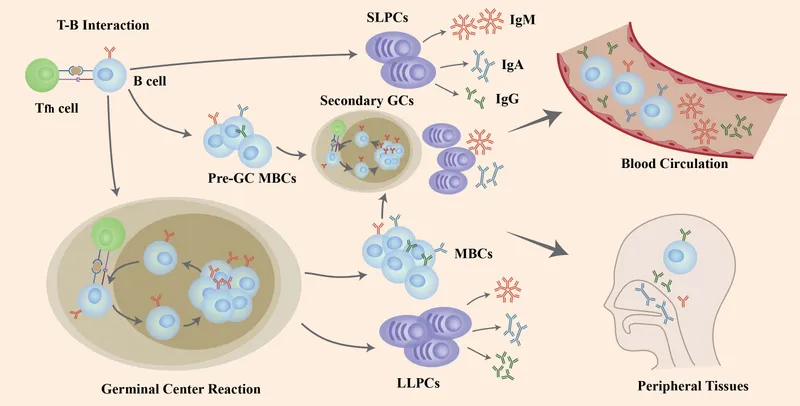
- Cooperative Enhancement Mechanisms
- Antigen presentation: Memory B cells present antigen to memory T cells
- Cytokine amplification: IL-4, IL-21 enhance antibody production
- Affinity maturation: Secondary germinal centers improve antibody quality
- Mutation rate: 10^-3 per base pair per division
- Selection pressure: 100-1000 fold affinity improvement
- Class switching: IgG, IgA production for tissue protection
The feedback regulation of memory responses prevents excessive activation while maintaining protective capacity. PD-1/PD-L1 interactions limit memory T cell activation, while FcγRIIB provides negative feedback for memory B cells.
💡 Master This: Memory inflation occurs during chronic viral infections, where persistent antigen exposure drives continuous memory cell expansion, potentially comprising >50% of total T cell populations in CMV-infected individuals.
- Integration Challenges and Solutions
- Autoimmune Prevention: Regulatory T cells control memory activation
- Tissue Damage Limitation: IL-10 and TGF-β resolve inflammation
- Resource Competition: Niche availability limits memory expansion
- Survival signals: Limited IL-7 and IL-15 availability
- Metabolic constraints: Glucose and amino acid competition
- Physical space: Anatomical niches restrict cell numbers
These integration networks connect through epigenetic coordination mechanisms to maintain stable memory programs while enabling rapid functional adaptation during recall responses.
🔗 Memory Integration Networks: The Immune Coordination Matrix
🎯 Memory Mastery Arsenal: The Clinical Command Center
📌 Remember: MASTER Clinical Memory - Measure (quantify responses), Assess (memory status), Stratify (patient risk), Time (optimal intervals), Evaluate (response quality), Respond (adjust protocols)
-
Essential Memory Metrics
- Memory Cell Frequencies: >1:1000 antigen-specific cells for protection
- Antibody Titers: >1:40 HAI for influenza, >10 IU/ml for hepatitis B
- Response Kinetics: <5 days to peak secondary responses
- Cross-Reactivity: >70% homology for heterologous protection
- Neutralizing capacity: IC50 <100 ng/ml for viral protection
- Avidity index: >60% for high-quality antibody responses
- Memory persistence: >10 years for live attenuated vaccines
-
Clinical Assessment Framework
- Memory Status Evaluation
- Vaccination history: Document all previous immunizations
- Infection exposure: Natural immunity assessment
- Immunocompromise: Evaluate memory formation capacity
- Response Prediction Models
- Age factors: 5-8% decline per decade after age 40
- Comorbidities: Diabetes reduces responses by 30-50%
- Medications: Immunosuppressants impair memory by 60-90%
- Memory Status Evaluation
| Patient Category | Memory Capacity | Vaccine Response | Booster Strategy | Monitoring Frequency |
|---|---|---|---|---|
| Healthy Adults | 100% baseline | >90% seroconversion | Standard intervals | Every 5-10 years |
| Elderly (>65) | 60-80% reduced | 60-80% response rate | Enhanced dosing | Every 2-3 years |
| Immunocompromised | 20-60% impaired | 30-70% variable | Frequent boosters | Every 6-12 months |
| Chronic Disease | 40-80% reduced | 50-80% response | Adjuvanted vaccines | Every 1-2 years |
| Pediatric | Developing | >95% robust | Series completion | Per schedule |
Optimization Strategies for memory-based interventions require understanding individual and population-level factors:
-
Vaccine Timing Optimization
- Prime-boost intervals: 4-8 weeks for rapid protection, 6-12 months for optimal memory
- Seasonal considerations: Fall vaccination for winter respiratory viruses
- Travel medicine: >2 weeks before exposure for memory development
- Yellow fever: Single dose provides >30 years protection
- Hepatitis A: >95% protection after 2-dose series
- Meningococcal: 3-5 years duration requiring boosters
-
Special Population Considerations
- Pregnancy: Enhanced antibody transfer to fetus during third trimester
- Immunosuppression: Live vaccines contraindicated, inactivated preferred
- Cancer patients: Timing around chemotherapy for optimal responses
- Pre-chemotherapy: >2 weeks before treatment initiation
- During treatment: Avoid live vaccines, monitor responses
- Post-treatment: >3 months after immune reconstitution
💡 Master This: Serological correlates of protection vary by pathogen - neutralizing antibodies for viruses, opsonizing antibodies for bacteria, cellular immunity for intracellular pathogens, requiring pathogen-specific memory assessment strategies.
- Advanced Memory Applications
- Therapeutic Vaccination: Cancer immunotherapy using memory enhancement
- Adoptive Transfer: Memory cell infusion for immunodeficiency
- Memory Modulation: Checkpoint inhibitors enhance memory function
- PD-1 blockade: Restores exhausted memory in chronic infections
- CTLA-4 inhibition: Enhances memory formation during priming
- Combination therapy: >60% response rates in cancer treatment
The clinical mastery of immunological memory transforms theoretical knowledge into practical tools for optimizing patient outcomes across diverse clinical scenarios, from routine vaccination to complex immunotherapy protocols.
🎯 Memory Mastery Arsenal: The Clinical Command Center
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more
Have doubts about this lesson?
Ask Rezzy, your AI Study Mate, to explain anything you didn't understand
Everything you need for NEET-PG prep
Get full Oncourse access with lessons, practice questions, flashcards and AI study tools.
Scan to download app























