Pulmonology

On this page
🫁 The Respiratory Arsenal: Mastering Pulmonary Medicine
You'll master the complete architecture of respiratory medicine-from airway anatomy through gas exchange mechanics to pattern recognition of clinical presentations. This lesson builds your diagnostic reasoning by connecting structural foundations with pathophysiology, then equips you with evidence-based investigation strategies and therapeutic protocols. You'll learn to recognize how pulmonary disease intersects with cardiac, renal, and metabolic systems, developing the rapid assessment frameworks essential for clinical decision-making. By integrating anatomy, physiology, and clinical reasoning, you'll gain the confidence to approach any respiratory case systematically and effectively.

Pulmonary medicine encompasses the intricate network of airways, gas exchange surfaces, and vascular systems that sustain life through every breath. Master these respiratory territories, and you possess the foundation for diagnosing everything from simple bronchitis to complex interstitial lung diseases.
⭐ Clinical Pearl: The respiratory system processes >20,000 breaths daily, moving >10,000 liters of air through 300 million alveoli with a combined surface area of 70 square meters - equivalent to a tennis court.
Understanding pulmonary pathophysiology requires systematic integration of anatomy, physiology, and clinical correlation. Every respiratory symptom follows predictable patterns based on anatomical involvement and physiological disruption.
📌 Remember: BREATHE for respiratory assessment - Breathing pattern, Respiratory rate, Expansion symmetry, Accessory muscle use, Tactile fremitus, Heart sounds, Exercise tolerance
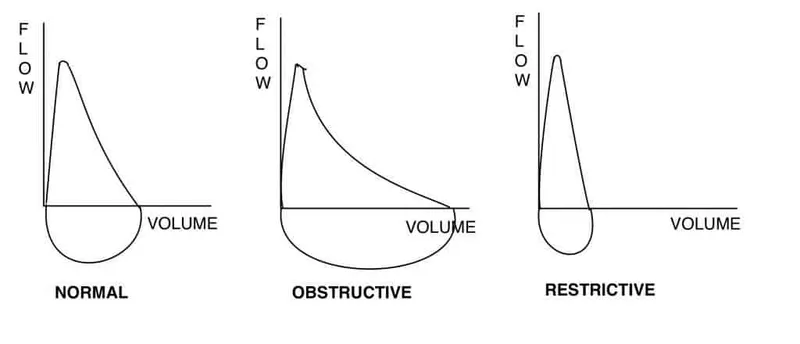
The respiratory system's complexity demands structured approaches to diagnosis and management. From obstructive patterns with FEV1/FVC <70% to restrictive diseases with TLC <80% predicted, quantitative thresholds guide clinical decision-making.
💡 Master This: Respiratory failure occurs when PaO2 <60 mmHg or PaCO2 >50 mmHg with pH <7.35, but clinical context determines intervention urgency more than absolute numbers.
Connect respiratory fundamentals through systematic disease patterns to understand how anatomical disruption creates predictable clinical presentations across the pulmonary medicine spectrum.
🫁 The Respiratory Arsenal: Mastering Pulmonary Medicine
🌬️ The Airway Architecture: Foundation of Respiratory Function

The conducting airways (generations 0-16) provide airflow pathways without gas exchange, while respiratory zones (generations 17-23) facilitate oxygen and carbon dioxide transfer. Understanding this anatomical hierarchy explains disease patterns and treatment targeting.
📌 Remember: CONDUCTING airways - Cartilage support, Only air movement, No gas exchange, Deadspace volume 150 mL, Upper 16 generations, Ciliary clearance, Trachea to terminal bronchioles, Innervated autonomically, Narrow progressively, Goblet cells present
| Airway Generation | Structure | Diameter (mm) | Function | Clinical Relevance |
|---|---|---|---|---|
| 0-4 | Trachea-Main bronchi | 18-12 | Air conduction | Central obstruction causes stridor |
| 5-11 | Lobar-Segmental bronchi | 8-3 | Air distribution | Bronchiectasis commonly affects |
| 12-16 | Terminal bronchioles | 1-0.5 | Final conduction | Asthma primarily targets |
| 17-19 | Respiratory bronchioles | 0.4-0.3 | Initial gas exchange | Emphysema destroys walls |
| 20-23 | Alveolar ducts/sacs | 0.2-0.1 | Primary gas exchange | Pneumonia fills spaces |
- Upper Airways (Generations 0-8)
- Cartilaginous support maintains patency
- Pseudostratified ciliated epithelium with goblet cells
- Mucociliary escalator clears 1-2 cm/minute
- Normal mucus production: 100 mL/day
- Increased in disease: >500 mL/day
- Lower Airways (Generations 9-16)
- Smooth muscle increases proportionally
- Clara cells produce surfactant precursors
- Autonomic innervation predominates
- Sympathetic: β2-receptors → bronchodilation
- Parasympathetic: M3-receptors → bronchoconstriction
⭐ Clinical Pearl: Airway resistance follows Poiseuille's law - resistance ∝ 1/radius⁴, so 50% diameter reduction increases resistance 16-fold, explaining why small airway inflammation causes dramatic symptoms.
💡 Master This: The "equal pressure point" during forced expiration moves peripherally in COPD, causing dynamic airway collapse and flow limitation independent of expiratory effort - explaining why COPD patients cannot "blow harder" to improve flow.
Understanding airway anatomy reveals why different diseases target specific generations and respond to particular interventions, forming the foundation for systematic respiratory disease management.
🌬️ The Airway Architecture: Foundation of Respiratory Function
⚙️ The Gas Exchange Engine: Alveolar-Capillary Interface Mastery
📌 Remember: MEMBRANE components - Microscopic thickness 0.5μm, Epithelial type I cells, Membrane basement fusion, Blood-gas barrier, Red cell transit 0.25s, Alveolar surface 70m², Normal diffusion capacity, Endothelial capillary lining
Gas exchange efficiency depends on ventilation-perfusion (V/Q) matching, with normal ratio 0.8-1.0. Mismatching creates physiological shunting and dead space ventilation, fundamental concepts in respiratory pathophysiology.
| Gas Exchange Parameter | Normal Value | Clinical Significance | Disease Correlation |
|---|---|---|---|
| Alveolar surface area | 70 m² | Determines diffusion capacity | ↓ in emphysema, fibrosis |
| Membrane thickness | 0.5 μm | Affects diffusion rate | ↑ in pulmonary edema |
| Capillary transit time | 0.25 seconds | Limits gas equilibration | ↓ in exercise, disease |
| V/Q ratio | 0.8-1.0 | Optimal gas exchange | Altered in most lung diseases |
| DLCO | 25-30 mL/min/mmHg | Overall diffusion capacity | ↓ in ILD, emphysema |
- Ultra-thin cytoplasm enables gas diffusion
- Tight junctions prevent fluid leak
- Cannot regenerate after injury
- Damage leads to type II cell proliferation
- Repair may cause fibrosis
- Type II Pneumocytes (5% surface area)
- Produce surfactant (phosphatidylcholine 70%)
- Stem cell function for alveolar repair
- Metabolically active with lamellar bodies
- Surfactant reduces surface tension from 70 to 25 dynes/cm
- Prevents alveolar collapse at low volumes
⭐ Clinical Pearl: Fick's law governs gas diffusion: Rate ∝ (Surface Area × Pressure Gradient) / (Membrane Thickness × Molecular Weight). This explains why CO diffusion (MW 28) tests membrane integrity better than O₂ (MW 32).
Ventilation-perfusion relationships determine gas exchange efficiency through gravitational and pathological influences. In upright position, V/Q ratio varies from 3.0 at apex to 0.6 at base, creating physiological dead space and shunt.
💡 Master This: The A-a gradient normally <15 mmHg in young adults, increasing 2.5 mmHg per decade. Elevation indicates V/Q mismatch, diffusion limitation, or right-to-left shunting - distinguishing pulmonary from cardiac causes of hypoxemia.
Connect gas exchange principles through pathophysiological disruptions to understand how diseases create predictable patterns of hypoxemia, hypercapnia, and respiratory failure.
⚙️ The Gas Exchange Engine: Alveolar-Capillary Interface Mastery
🎯 The Pattern Recognition Matrix: Clinical Presentation Frameworks
📌 Remember: DYSPNEA pattern analysis - Duration (acute vs chronic), Young vs elderly, Smoking history, Positional changes, Nocturnal symptoms, Exercise limitation, Associated symptoms, Auscultatory findings
| Clinical Pattern | Key Features | Diagnostic Tests | Sensitivity/Specificity |
|---|---|---|---|
| Obstructive | Expiratory wheeze, prolonged expiration | FEV1/FVC <70% | 95%/90% |
| Restrictive | Inspiratory crackles, reduced expansion | TLC <80% predicted | 90%/95% |
| Vascular | Sudden onset, pleuritic pain | D-dimer >500 ng/mL | 85%/50% |
| Infectious | Fever, purulent sputum | Chest X-ray infiltrates | 80%/70% |
| Malignant | Weight loss, hemoptysis | CT chest abnormalities | 90%/85% |
- Pulmonary embolism: sudden onset, pleuritic pain, tachycardia >100
- Pneumothorax: sharp chest pain, reduced breath sounds unilaterally
- Acute asthma: expiratory wheeze, accessory muscle use, peak flow <50%
- Severe: cannot speak full sentences
- Life-threatening: silent chest, pulsus paradoxus >25 mmHg
- Chronic Dyspnea Patterns (Weeks to Months)
- COPD: smoking history, barrel chest, FEV1/FVC <70%
- Interstitial lung disease: dry cough, fine crackles, DLCO <80%
- Heart failure: orthopnea, S3 gallop, BNP >400 pg/mL
- Pulmonary edema: bilateral crackles, frothy sputum
- Right heart failure: peripheral edema, elevated JVP
⭐ Clinical Pearl: The "orthopnea grade" quantifies heart failure severity: Grade 1 = 3+ pillows, Grade 2 = 2 pillows, Grade 3 = 1 pillow, Grade 4 = cannot lie flat. Each grade correlates with NYHA functional class and ejection fraction ranges.
Physical examination findings provide immediate diagnostic clues with high specificity when properly interpreted. Combining multiple findings increases diagnostic accuracy exponentially.
- High-Yield Auscultatory Patterns
- Fine crackles: Interstitial disease, early CHF (sensitivity 80%)
- Coarse crackles: Pneumonia, advanced CHF (specificity 85%)
- Expiratory wheeze: Asthma, COPD (sensitivity 90% for obstruction)
- Inspiratory wheeze: Upper airway obstruction, severe asthma
- Stridor: Central airway obstruction (emergency evaluation)
- Pleural friction rub: Pleuritis, malignancy (specificity 95%)
💡 Master This: Pulsus paradoxus >10 mmHg indicates severe airway obstruction or cardiac tamponade. In asthma, >25 mmHg suggests life-threatening attack requiring immediate intervention, while >40 mmHg may indicate impending respiratory arrest.
Understanding clinical patterns enables rapid triage and appropriate diagnostic testing, forming the foundation for efficient respiratory medicine practice and optimal patient outcomes.
🎯 The Pattern Recognition Matrix: Clinical Presentation Frameworks
🔬 The Diagnostic Arsenal: Evidence-Based Investigation Strategies
📌 Remember: IMAGING sequence - Initial chest X-ray, Modality selection (CT/MRI), Angiography if vascular, Guided biopsy procedures, Interventional options, Nuclear medicine studies, Genetic testing when indicated
| Diagnostic Test | Sensitivity | Specificity | Best Clinical Use | Cost-Effectiveness |
|---|---|---|---|---|
| Chest X-ray | 70-80% | 85-90% | Initial screening | High |
| CT chest | 90-95% | 80-85% | Detailed evaluation | Moderate |
| PFTs | 85-90% | 90-95% | Functional assessment | High |
| Bronchoscopy | 80-90% | 95-98% | Tissue diagnosis | Low |
| CTPA | 95-98% | 90-95% | Pulmonary embolism | Moderate |
- Chest X-ray: $50-100, identifies 70% of significant pathology
- Pulse oximetry: $5-10, screens for hypoxemia >95% accuracy
- Basic metabolic panel: $25-50, assesses acid-base status
- Anion gap >12: Suggests metabolic acidosis
- HCO3 <22: Compensatory or primary metabolic acidosis
- Advanced Imaging Strategies
- High-resolution CT (HRCT): $500-800, >95% sensitivity for ILD
- CT pulmonary angiogram (CTPA): $800-1200, 98% sensitivity for PE
- Ventilation-perfusion scan: $400-600, preferred in pregnancy, renal failure
- High probability: >80% chance of PE
- Low probability: <20% chance of PE

Pulmonary function testing provides objective assessment of respiratory mechanics and gas exchange, with specific patterns indicating disease categories and severity.
- Spirometry Interpretation
- FEV1/FVC <70%: Obstructive pattern (COPD, asthma)
- FVC <80% predicted: Restrictive pattern (ILD, chest wall disease)
- Bronchodilator response >12% and 200 mL: Suggests reversible obstruction
- Asthma: Usually >15% improvement
- COPD: Typically <12% improvement
- Advanced PFT Parameters
- DLCO <80% predicted: Impaired gas exchange (emphysema, ILD)
- Total lung capacity (TLC): Distinguishes restriction from poor effort
- Residual volume (RV): Elevated in air trapping (>120% predicted)
⭐ Clinical Pearl: D-dimer has >95% negative predictive value for PE when <500 ng/mL in low-risk patients, but positive predictive value <50% due to elevation in multiple conditions including infection, malignancy, and pregnancy.
Invasive diagnostic procedures require careful risk-benefit analysis and appropriate patient selection to maximize diagnostic yield while minimizing complications.
💡 Master This: Bronchoscopy diagnostic yield varies by indication: >90% for endobronchial lesions, 70-80% for peripheral nodules >2 cm, <50% for nodules <1 cm. Transbronchial biopsy carries 5-10% pneumothorax risk, requiring post-procedure chest X-ray.
Connect diagnostic strategies through evidence-based algorithms to develop systematic approaches that optimize accuracy, efficiency, and patient safety in respiratory medicine practice.
🔬 The Diagnostic Arsenal: Evidence-Based Investigation Strategies
🎛️ The Treatment Command Center: Therapeutic Mastery Protocols
📌 Remember: THERAPY principles - Targeted mechanism, Hierarchical approach, Evidence-based dosing, Response monitoring, Adverse effect surveillance, Patient education, Yield optimization
| Therapeutic Class | Mechanism | Onset Time | Duration | Clinical Efficacy |
|---|---|---|---|---|
| SABA (Albuterol) | β2-agonist | 5-15 minutes | 4-6 hours | 85% bronchodilation |
| LABA (Salmeterol) | β2-agonist | 30-60 minutes | 12 hours | 70% symptom control |
| ICS (Fluticasone) | Anti-inflammatory | 2-4 weeks | 12-24 hours | 60% exacerbation reduction |
| LAMA (Tiotropium) | Anticholinergic | 30 minutes | 24 hours | 75% COPD control |
| Systemic steroids | Anti-inflammatory | 4-6 hours | Variable | 90% acute response |
- Short-acting β2-agonists (SABA): Albuterol 2.5-5 mg nebulized
- Onset: 5-15 minutes, peak: 30-60 minutes
- Rescue therapy: 2-4 puffs q4-6h PRN
- Acute exacerbation: Continuous nebulization if severe
- Long-acting β2-agonists (LABA): Never monotherapy in asthma
- Salmeterol 50 μg BID or Formoterol 12 μg BID
- Combination with ICS: Reduces exacerbations 40-50%
- Anti-inflammatory Strategies
- Inhaled corticosteroids (ICS): First-line controller therapy
- Low dose: Fluticasone 100-250 μg/day
- Medium dose: 250-500 μg/day
- High dose: >500 μg/day (monitor for systemic effects)
- Systemic corticosteroids: Prednisolone 0.5-1 mg/kg/day
- Acute asthma: 40-60 mg daily × 5-7 days
- COPD exacerbation: 30-40 mg daily × 5 days
- Inhaled corticosteroids (ICS): First-line controller therapy
Oxygen therapy requires precise titration based on underlying pathophysiology, with different targets for acute versus chronic conditions and specific monitoring parameters.
- Oxygen Therapy Protocols
- Acute hypoxemia: Target SpO2 94-98% (normal lungs)
- COPD patients: Target SpO2 88-92% (avoid CO2 retention)
- Delivery systems: Nasal cannula 1-6 L/min, Venturi mask 24-60%
- High-flow nasal cannula: Up to 60 L/min, 21-100% FiO2
- Non-invasive ventilation: BiPAP for hypercapnic failure
- Advanced Respiratory Support
- Mechanical ventilation indications: pH <7.25, PaCO2 >60 mmHg with acidosis
- Lung-protective ventilation: Tidal volume 6-8 mL/kg ideal body weight
- PEEP titration: 5-15 cmH2O based on oxygenation and compliance
⭐ Clinical Pearl: Inhaler technique errors occur in >70% of patients, reducing drug delivery by 50-90%. Spacer devices improve lung deposition from 10-15% to 20-30% and reduce oropharyngeal side effects.
Combination therapy strategies follow evidence-based guidelines with specific sequencing and monitoring requirements to optimize outcomes while minimizing polypharmacy risks.
💡 Master This: Step-up therapy in asthma follows GINA guidelines: Step 1 (SABA PRN) → Step 2 (Low-dose ICS) → Step 3 (ICS/LABA) → Step 4 (Medium/high-dose ICS/LABA) → Step 5 (Add LAMA, biologics, or oral steroids).
Understanding therapeutic protocols enables systematic treatment approaches that maximize efficacy, minimize adverse effects, and improve long-term respiratory outcomes across diverse pulmonary conditions.
🎛️ The Treatment Command Center: Therapeutic Mastery Protocols
🧠 The Integration Network: Multi-System Respiratory Connections
📌 Remember: SYSTEMS integration - Systemic circulation coupling, Young vs elderly responses, Sensory chemoreceptor feedback, Tissue oxygen delivery, Endocrine hormone interactions, Metabolic acid-base balance, Sympathetic nervous system activation
| System Integration | Mechanism | Clinical Manifestation | Quantitative Threshold |
|---|---|---|---|
| Cardiovascular | Hypoxic pulmonary vasoconstriction | Cor pulmonale | PaO2 <60 mmHg chronically |
| Neurological | Central chemoreceptor response | Altered mental status | PaCO2 >50 mmHg acutely |
| Renal | Bicarbonate retention/excretion | Metabolic compensation | pH 7.35-7.45 maintenance |
| Hematologic | Erythropoietin stimulation | Secondary polycythemia | Hgb >16 g/dL (men), >14 g/dL (women) |
| Metabolic | Oxygen consumption changes | Exercise intolerance | VO2 max <85% predicted |
- Hypoxic pulmonary vasoconstriction: PaO2 <60 mmHg triggers vasoconstriction
- Acute response: Optimizes V/Q matching
- Chronic response: Leads to pulmonary hypertension (mPAP >25 mmHg)
- Right heart adaptation: Cor pulmonale develops with sustained pressure elevation
- Compensated: Normal cardiac output at rest
- Decompensated: Elevated JVP, peripheral edema, hepatomegaly
- Neurological-Respiratory Integration
- Central chemoreceptors (medulla): Respond to CSF pH changes from CO2
- Primary drive: 80% of respiratory stimulus at rest
- Blunted response: In chronic hypercapnia (COPD)
- Peripheral chemoreceptors (carotid/aortic bodies): Respond to PaO2 <60 mmHg
- Hypoxic drive: Becomes primary in chronic CO2 retention
- Oxygen therapy: Can suppress drive in COPD patients
- Central chemoreceptors (medulla): Respond to CSF pH changes from CO2
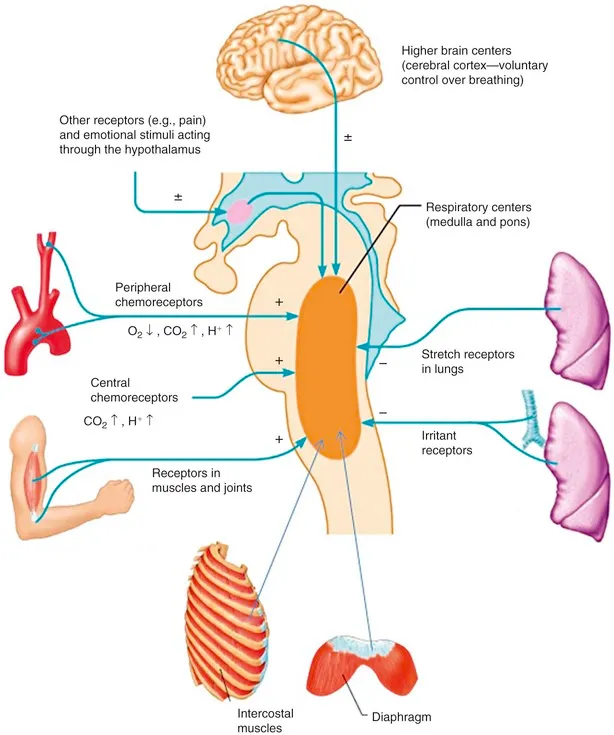
Acid-base integration demonstrates sophisticated physiological compensation mechanisms that maintain pH homeostasis through respiratory and renal adjustments with specific time courses and quantitative relationships.
- Respiratory Acid-Base Compensation
- Metabolic acidosis: Hyperventilation within minutes
- Expected PaCO2 = 1.5 × [HCO3] + 8 ± 2
- Winter's formula: Predicts appropriate compensation
- Metabolic alkalosis: Hypoventilation limited by hypoxemia
- PaCO2 rarely exceeds 55 mmHg due to hypoxic drive
- Compensation: 0.7 mmHg rise per 1 mEq/L HCO3 increase
- Metabolic acidosis: Hyperventilation within minutes
- Renal Respiratory Compensation
- Respiratory acidosis: HCO3 retention over days
- Acute: 1 mEq/L rise per 10 mmHg PaCO2 increase
- Chronic: 3.5 mEq/L rise per 10 mmHg PaCO2 increase
- Respiratory alkalosis: HCO3 excretion over days
- Acute: 2 mEq/L fall per 10 mmHg PaCO2 decrease
- Chronic: 5 mEq/L fall per 10 mmHg PaCO2 decrease
- Respiratory acidosis: HCO3 retention over days

⭐ Clinical Pearl: Sleep-disordered breathing affects >30% of heart failure patients, creating bidirectional pathophysiology where OSA worsens heart failure through increased afterload and heart failure worsens OSA through fluid shifts and central apneas.
Hematologic adaptations to chronic hypoxemia involve erythropoietin-mediated responses that can become maladaptive, requiring careful monitoring and intervention strategies.
💡 Master This: Secondary polycythemia from chronic hypoxemia improves oxygen delivery initially but becomes detrimental when hematocrit >55% due to increased viscosity. Phlebotomy may be indicated when symptoms occur with hematocrit >60%.
Understanding multi-system integration enables comprehensive management approaches that address respiratory diseases within the context of overall physiological homeostasis and comorbid conditions.
🧠 The Integration Network: Multi-System Respiratory Connections
🎯 The Clinical Mastery Toolkit: Rapid Assessment and Decision Frameworks
📌 Remember: MASTERY framework - Multi-system assessment, Algorithmic thinking, Severity stratification, Treatment prioritization, Evidence integration, Risk-benefit analysis, Yield optimization
| Clinical Scenario | Rapid Assessment Tools | Critical Thresholds | Immediate Actions |
|---|---|---|---|
| Acute dyspnea | CURB-65, Wells score | SpO2 <90%, RR >30 | Oxygen, imaging, labs |
| Asthma exacerbation | Peak flow, accessory muscles | PEF <50%, silent chest | Bronchodilators, steroids |
| COPD exacerbation | ABG, mental status | pH <7.35, altered LOC | NIV, antibiotics |
| Suspected PE | Wells score, D-dimer | High probability >6 | Anticoagulation, CTPA |
| Respiratory failure | ABG, work of breathing | pH <7.25, fatigue | Ventilatory support |
- Normal values: RR 12-20, SpO2 >95%, PaO2 >80 mmHg, PaCO2 35-45 mmHg
- Critical thresholds: SpO2 <90%, PaO2 <60 mmHg, pH <7.30, RR >30
- Severity markers: Accessory muscle use, inability to speak, altered mental status
- Mild distress: Speaks full sentences, RR <25
- Moderate distress: Speaks phrases, RR 25-35
- Severe distress: Single words only, RR >35
⭐ Clinical Pearl: The "Rule of 30s" identifies respiratory failure: RR >30, PaO2 <30 mmHg on room air, A-a gradient >30 mmHg, or FiO2 requirement >30% to maintain adequate oxygenation.
💡 Master This: Pattern recognition mastery combines anatomical knowledge + physiological understanding + clinical experience to achieve diagnostic accuracy >95% in respiratory medicine. Systematic approaches prevent cognitive errors and optimize patient outcomes across diverse presentations.
🎯 The Clinical Mastery Toolkit: Rapid Assessment and Decision Frameworks
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more
Have doubts about this lesson?
Ask Rezzy, your AI Study Mate, to explain anything you didn't understand
Everything you need for NEET-PG prep
Get full Oncourse access with lessons, practice questions, flashcards and AI study tools.
Scan to download app












