EBM Basics - Doctor Detective Toolkit
⭐ "Evidence-based medicine is the conscientious, explicit, and judicious use of current best evidence in making decisions about the care of individual patients." - Sackett
- Core: Integrates best research evidence, clinical expertise, & patient values.
- Importance: ↑Patient outcomes, guides clinical decisions, promotes lifelong learning.
- Process: 📌 The 5 A's
- Ask: Formulate a focused clinical question (e.g., PICO).
- Acquire: Systematically retrieve the best available evidence.
- Appraise: Critically assess evidence for validity and applicability.
- Apply: Integrate evidence with clinical expertise and patient preferences.
- Assess: Evaluate the effectiveness and efficiency of the process.
Study Types - Evidence Pyramid Power
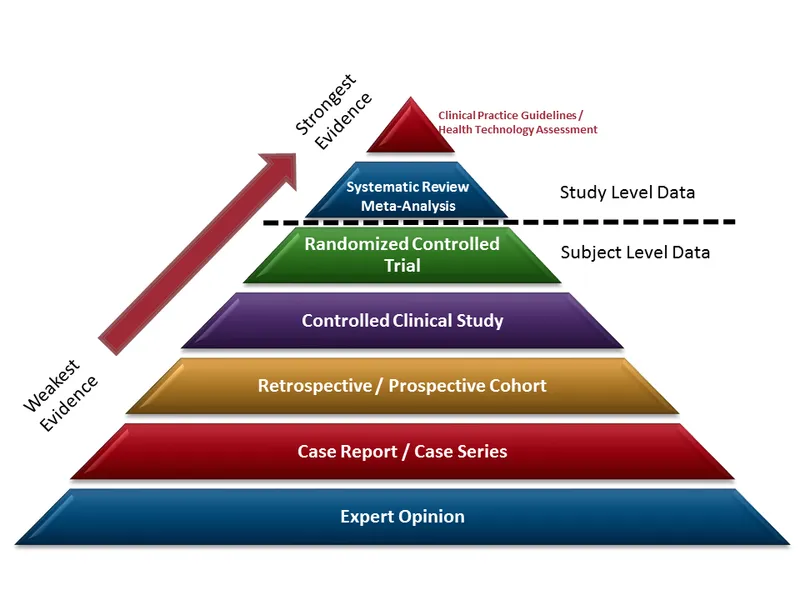
- Evidence Hierarchy (Bottom to Top):
- Expert Opinion, Editorials, Ideas
- In vitro (test tube) research
- Animal research
- Case Reports & Case Series: Describes rare conditions/outcomes.
- Cross-sectional Studies: Prevalence at a point in time (snapshot).
- Case-Control Studies: Retrospective; Odds Ratio (OR). (📌 Case-ConTROl = ReTROspective)
- Cohort Studies: Prospective or retrospective; Relative Risk (RR), Incidence.
- Randomized Controlled Trials (RCTs): Intervention studies; best for cause-effect.
- Systematic Reviews: Structured literature review on a focused question.
- Meta-Analyses: Pools data from multiple studies statistically; highest evidence.
⭐ Gold standard for intervention studies is a well-conducted RCT or meta-analysis of RCTs.
- Key Distinctions:
- Observational (no intervention) vs. Experimental (intervention).
- Descriptive (generate hypotheses) vs. Analytic (test hypotheses).
Biostats in EBM - Numbers Don't Lie
- Diagnostic Accuracy (2x2 Table):
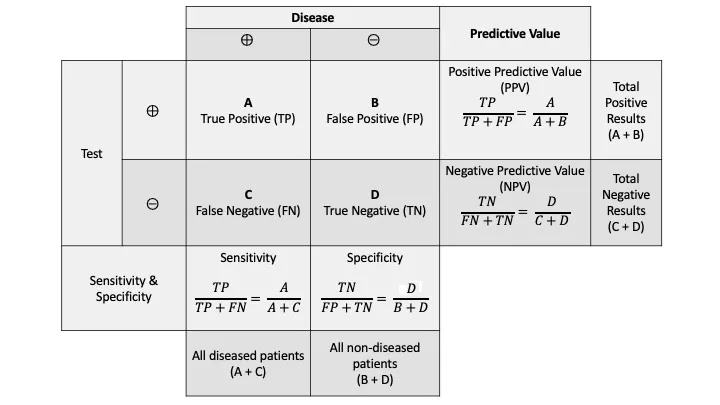
- Sensitivity (Sn): $TP/(TP+FN)$. True Pos rate. 📌 SNOUT (Sensitive test, Negative result, rules OUT disease).
- Specificity (Sp): $TN/(TN+FP)$. True Neg rate. 📌 SPIN (Specific test, Positive result, rules IN disease).
- PPV: $TP/(TP+FP)$. Prob. disease if test Pos.
- NPV: $TN/(TN+FN)$. Prob. no disease if test Neg.
- Association & Effect:
- Relative Risk (RR): $[a/(a+b)]/[c/(c+d)]$. Risk ratio (cohorts).
- Odds Ratio (OR): $(ad)/(bc)$. Odds of exposure (case-control).
- Absolute Risk Reduction (ARR): $|CER - EER|$.
- Number Needed to Treat (NNT): $1/ARR$. Lower = better.
- Significance Testing:
- p-value: If < 0.05, statistically significant.
- Confidence Interval (CI): Range of plausible true values.
⭐ For RR/OR, a 95% CI not crossing 1.0 (e.g., 1.2-3.5 or 0.3-0.7) means statistical significance.
Appraisal & Use - Smart Practice Steps
Integrate evidence into daily practice using these key steps for informed decisions.
⭐ EBM involves integrating best research evidence with clinical expertise and patient values.
- Evidence-Based Practice Cycle:
- Critical Appraisal Focus (📌 VIA):
- Validity: How true are the study results? (Bias, study design)
- Importance: How clinically significant are findings? (Magnitude of effect, NNT)
- Applicability: How relevant to your specific patient? (Context, resources)
High‑Yield Points - ⚡ Biggest Takeaways
- Formulate clinical questions using PICO (Patient, Intervention, Comparison, Outcome).
- Hierarchy of evidence: Systematic reviews & meta-analyses are at the top.
- Recognize and mitigate various types of bias in research.
- Critical appraisal is key to assessing study validity and relevance.
- Calculate NNT (1/ARR) and NNH (1/ARI) for intervention effects.
- Interpret sensitivity, specificity, PPV, and NPV for diagnostic tests.
- Forest plots summarize meta-analysis results, including pooled estimates and CIs (Confidence Intervals).
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

