General Medicine

On this page
🩺 The Cardiovascular Command Center: Mastering Heart Failure Pathophysiology
Heart failure affects over 6 million Americans and remains one of medicine's most complex clinical challenges, demanding you master not just isolated facts but integrated systems thinking. You'll build a complete mental framework from cellular mechanics through hemodynamic principles to bedside diagnosis, learning how the failing heart triggers cascading multi-organ effects and how evidence-based therapies target specific pathophysiologic nodes. By connecting molecular mechanisms to clinical patterns and treatment algorithms, you'll develop the diagnostic precision and therapeutic reasoning that transforms you from memorizer to master clinician capable of rapid, accurate decision-making when patients need it most.
The heart failure spectrum encompasses two primary phenotypes: Heart Failure with Reduced Ejection Fraction (HFrEF) with EF <40% and Heart Failure with Preserved Ejection Fraction (HFpEF) with EF ≥50%. Understanding these distinct pathophysiological patterns unlocks the logic behind every therapeutic intervention.
📌 Remember: FAILURES mnemonic for heart failure etiology - Failure of contractility, Afterload excess, Ischemia, Load volume excess, Uncontrolled hypertension, Rhythm disorders, Endocrine disorders, Structural abnormalities
-
Systolic Heart Failure (HFrEF)
- Primary mechanism: Impaired contractility with EF <40%
- Compensatory responses: Neurohormonal activation within 24-48 hours
- Frank-Starling mechanism: ↑ preload to maintain stroke volume
- Sympathetic activation: ↑ heart rate and contractility
- RAAS activation: Sodium retention and vasoconstriction
- Progressive remodeling: Chamber dilation and wall thinning
-
Diastolic Heart Failure (HFpEF)
- Primary mechanism: Impaired relaxation with preserved EF ≥50%
- Pathophysiology: ↑ filling pressures despite normal systolic function
- Left ventricular stiffness: ↑ end-diastolic pressure >16 mmHg
- Impaired lusitropy: Delayed relaxation and ↓ compliance
- Pulmonary congestion: Elevated PCWP >15 mmHg at rest
| Parameter | HFrEF | HFpEF | Mixed HF | Acute HF | Chronic HF |
|---|---|---|---|---|---|
| Ejection Fraction | <40% | ≥50% | 41-49% | Variable | Variable |
| LV End-Diastolic Pressure | ↑↑ >18 mmHg | ↑ 16-20 mmHg | ↑ 15-18 mmHg | ↑↑↑ >25 mmHg | ↑ 12-18 mmHg |
| Cardiac Index | <2.2 L/min/m² | 2.2-2.8 L/min/m² | 2.0-2.5 L/min/m² | <2.0 L/min/m² | 2.0-2.5 L/min/m² |
| BNP Levels | >400 pg/mL | >200 pg/mL | >300 pg/mL | >900 pg/mL | >150 pg/mL |
| Mortality (5-year) | 65% | 45% | 55% | 30% (in-hospital) | 50% |
The neurohormonal cascade begins within hours of cardiac dysfunction, triggering compensatory mechanisms that ultimately become maladaptive. Sympathetic nervous system activation increases norepinephrine levels 3-5 fold, while RAAS activation elevates angiotensin II and aldosterone, promoting sodium retention and vasoconstriction.
💡 Master This: The transition from compensated to decompensated heart failure occurs when compensatory mechanisms become pathological drivers. Sympathetic activation initially maintains cardiac output but eventually causes downregulation of β1-receptors and myocyte apoptosis, while RAAS activation preserves perfusion pressure but promotes fibrosis and remodeling.
Understanding these foundational concepts creates the framework for recognizing the hemodynamic patterns that distinguish different heart failure phenotypes and guide targeted therapeutic interventions.
⚙️ The Hemodynamic Engine: Cardiac Performance Mechanics
-
Preload Mechanisms
- Frank-Starling Law: ↑ venous return → ↑ fiber stretch → ↑ contractile force
- Optimal preload: PCWP 15-18 mmHg for maximum stroke volume
- Underfilled: PCWP <12 mmHg → ↓ stroke volume
- Overfilled: PCWP >20 mmHg → pulmonary edema risk
- Volume overload: ↑ end-diastolic volume → chamber dilation
-
Afterload Dynamics
- Systemic vascular resistance: Normal 800-1200 dynes·sec/cm⁵
- Heart failure: SVR >1500 dynes·sec/cm⁵ → ↓ stroke volume
- Laplace's Law: Wall tension = (Pressure × Radius) / (2 × Wall thickness)
- Dilated ventricle: ↑ radius → ↑ wall tension → ↑ oxygen demand
- Arterial elastance: ↑ stiffness → ↑ afterload → ↓ efficiency
📌 Remember: SOAP for hemodynamic optimization - SVR reduction, Optimal preload, Augmented contractility, Perfusion pressure maintenance. Target SVR 800-1200, PCWP 15-18 mmHg, CI >2.2 L/min/m², MAP >65 mmHg.
| Hemodynamic Profile | PCWP | Cardiac Index | SVR | Clinical Presentation | Mortality Risk |
|---|---|---|---|---|---|
| Warm and Dry | <18 mmHg | >2.2 L/min/m² | <1200 | Compensated | Low (5%) |
| Warm and Wet | >18 mmHg | >2.2 L/min/m² | <1200 | Volume overload | Moderate (15%) |
| Cold and Dry | <18 mmHg | <2.2 L/min/m² | >1200 | Low output | High (25%) |
| Cold and Wet | >18 mmHg | <2.2 L/min/m² | >1200 | Cardiogenic shock | Very High (40%) |
Contractility assessment requires understanding the pressure-volume relationship and load-independent measures. The end-systolic pressure-volume relationship (ESPVR) provides the gold standard for contractility evaluation, with slope reduction indicating systolic dysfunction.
⭐ Clinical Pearl: dP/dt max >1200 mmHg/sec indicates normal contractility, while <800 mmHg/sec suggests significant systolic dysfunction. Ejection fraction remains load-dependent and may appear normal despite impaired contractility in acute mitral regurgitation.
The ventricular-arterial coupling concept describes the interaction between cardiac function and vascular load. Optimal coupling occurs when Ea/Ees ratio = 0.5-1.0, maximizing mechanical efficiency while maintaining adequate stroke work.
💡 Master This: Hemodynamic optimization requires simultaneous assessment of all four determinants. Isolated preload reduction may worsen cardiac output in preload-dependent patients, while afterload reduction without adequate preload can cause hypotension. The goal is hemodynamic coherence - adequate perfusion with minimal congestion.
These hemodynamic principles form the foundation for understanding how therapeutic interventions restore the balance between cardiac performance and metabolic demands.
🎯 The Clinical Recognition Matrix: Pattern-Based Diagnosis
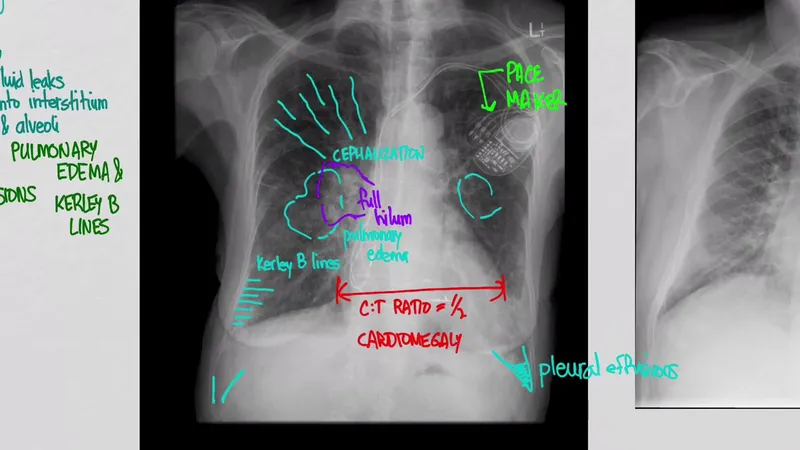
Clinical presentation patterns follow predictable hemodynamic profiles that guide both diagnosis and treatment selection. The "warm/cold" and "wet/dry" classification system provides a systematic framework for rapid assessment and therapeutic targeting.
-
Congestion Assessment ("Wet" vs "Dry")
- Wet signs: JVD >8 cm, S3 gallop, rales, peripheral edema
- Orthopnea: Sensitivity 84% for elevated filling pressures
- Paroxysmal nocturnal dyspnea: Specificity 91% for heart failure
- Hepatojugular reflux: >3 cm JVD rise with 95% specificity
- Dry signs: Absence of congestion with normal JVP and clear lungs
- BNP <100 pg/mL: 98% negative predictive value for heart failure
- Normal chest X-ray: Excludes acute pulmonary edema with >95% certainty
- Wet signs: JVD >8 cm, S3 gallop, rales, peripheral edema
-
Perfusion Assessment ("Warm" vs "Cold")
- Cold signs: Cool extremities, delayed capillary refill >3 seconds, altered mental status
- Narrow pulse pressure <25 mmHg: Indicates low stroke volume
- Proportional pulse pressure <0.25: Suggests reduced cardiac output
- Lactate >2.0 mmol/L: Tissue hypoperfusion marker
- Warm signs: Adequate perfusion with normal pulse pressure and mentation
- Cold signs: Cool extremities, delayed capillary refill >3 seconds, altered mental status
📌 Remember: FACES for heart failure recognition - Fatigue and weakness, Activity limitation, Congestion signs, Edema formation, Shortness of breath. Each component has specific sensitivity/specificity profiles for diagnostic accuracy.
| Clinical Sign | Sensitivity | Specificity | Positive LR | Negative LR | Clinical Utility |
|---|---|---|---|---|---|
| Dyspnea on exertion | 84% | 17% | 1.0 | 0.9 | Screening |
| Orthopnea | 50% | 91% | 5.6 | 0.5 | Rule-in |
| S3 gallop | 31% | 95% | 6.2 | 0.7 | Rule-in |
| JVD >8 cm | 39% | 92% | 4.9 | 0.7 | Rule-in |
| Peripheral edema | 67% | 68% | 2.1 | 0.5 | Moderate |
- BNP Diagnostic Thresholds
- Acute setting: BNP >400 pg/mL or NT-proBNP >2000 pg/mL
- Chronic setting: BNP >150 pg/mL or NT-proBNP >600 pg/mL
- Age adjustment: NT-proBNP thresholds - <50 years: >450, 50-75 years: >900, >75 years: >1800
- Confounding factors: Renal dysfunction (↑ levels), obesity (↓ levels), atrial fibrillation (↑ levels)
⭐ Clinical Pearl: BNP/NT-proBNP ratio >0.5 suggests acute heart failure, while ratio <0.2 indicates chronic stable disease. Serial measurements provide more value than single values for monitoring treatment response.
Echocardiographic patterns complete the diagnostic framework by providing structural and functional assessment. The comprehensive echo evaluation includes systolic function, diastolic parameters, valve assessment, and hemodynamic estimation.
💡 Master This: Clinical pattern recognition requires integration of history, physical examination, biomarkers, and imaging. No single finding confirms or excludes heart failure - the diagnostic probability emerges from the constellation of findings. High pretest probability patients with negative BNP may still have heart failure, particularly HFpEF or chronic stable disease.
These recognition patterns enable rapid triage and appropriate diagnostic workup, forming the foundation for evidence-based treatment decisions.
🔬 The Diagnostic Discrimination Engine: Systematic Differentiation
-
HFrEF Characteristics
- Ejection fraction: <40% with global hypokinesis
- LV geometry: Eccentric remodeling with chamber dilation
- LV end-diastolic diameter: >5.6 cm (normal <5.2 cm)
- LV end-systolic diameter: >4.0 cm (normal <3.6 cm)
- Relative wall thickness: <0.42 indicating eccentric pattern
- Hemodynamics: ↓ stroke volume, ↑ filling pressures, compensatory tachycardia
- Biomarkers: BNP >400 pg/mL, troponin elevation in 70% of cases
-
HFpEF Characteristics
- Ejection fraction: ≥50% with preserved systolic function
- LV geometry: Concentric remodeling with normal chamber size
- LV mass index: >95 g/m² (women), >115 g/m² (men)
- Relative wall thickness: >0.42 indicating concentric pattern
- Left atrial volume index: >34 mL/m² from chronic pressure elevation
- Diastolic dysfunction: E/e' ratio >14 indicates ↑ filling pressures
- Comorbidities: Hypertension (85%), diabetes (45%), obesity (70%)
📌 Remember: RELAX for HFpEF diagnosis - Reduced exercise capacity, Elevated natriuretic peptides, Left atrial enlargement, Abnormal diastolic function, X-ray showing pulmonary congestion. All components must be present with preserved EF.
| Discriminating Feature | HFrEF | HFpEF | HFmrEF | Acute HF | Right HF |
|---|---|---|---|---|---|
| Ejection Fraction | <40% | ≥50% | 40-49% | Variable | Variable |
| LV End-Diastolic Diameter | >5.6 cm | <5.2 cm | 5.2-5.6 cm | Variable | Normal |
| E/e' Ratio | >14 | >14 | >14 | >20 | <8 |
| RVSP | 35-45 mmHg | 40-50 mmHg | 35-45 mmHg | >50 mmHg | >40 mmHg |
| Treatment Response | ACE-I/ARB +++ | Limited | Moderate | Diuretics +++ | RV-specific |

-
Ischemic Cardiomyopathy
- Coronary anatomy: >70% stenosis in major epicardial vessels
- Wall motion: Regional abnormalities in coronary territories
- Viability assessment: >10% viable myocardium predicts revascularization benefit
- Biomarkers: Persistent troponin elevation suggests ongoing ischemia
-
Non-ischemic Cardiomyopathy
- Coronary anatomy: <50% stenosis in all vessels
- Wall motion: Global hypokinesis without territorial pattern
- Specific etiologies: Viral (30%), idiopathic (50%), toxic (10%), genetic (10%)
- Reversibility: Higher potential for functional recovery
⭐ Clinical Pearl: Coronary angiography remains the gold standard for ischemic versus non-ischemic discrimination. Cardiac MRI with late gadolinium enhancement can identify myocardial scar patterns - subendocardial suggests ischemic, while mid-wall or epicardial suggests non-ischemic etiology.
Advanced discrimination includes infiltrative, inflammatory, and genetic cardiomyopathies that require specific diagnostic approaches and targeted therapies.
💡 Master This: Systematic discrimination prevents therapeutic errors and missed opportunities. HFpEF patients do not benefit from ACE inhibitors or beta-blockers for mortality reduction, while ischemic cardiomyopathy patients may benefit from revascularization even with severe LV dysfunction. Accurate phenotyping determines evidence-based treatment selection.
This discrimination framework ensures appropriate therapeutic targeting and optimal patient outcomes through precision diagnosis.
⚕️ The Therapeutic Algorithm: Evidence-Based Treatment Pathways
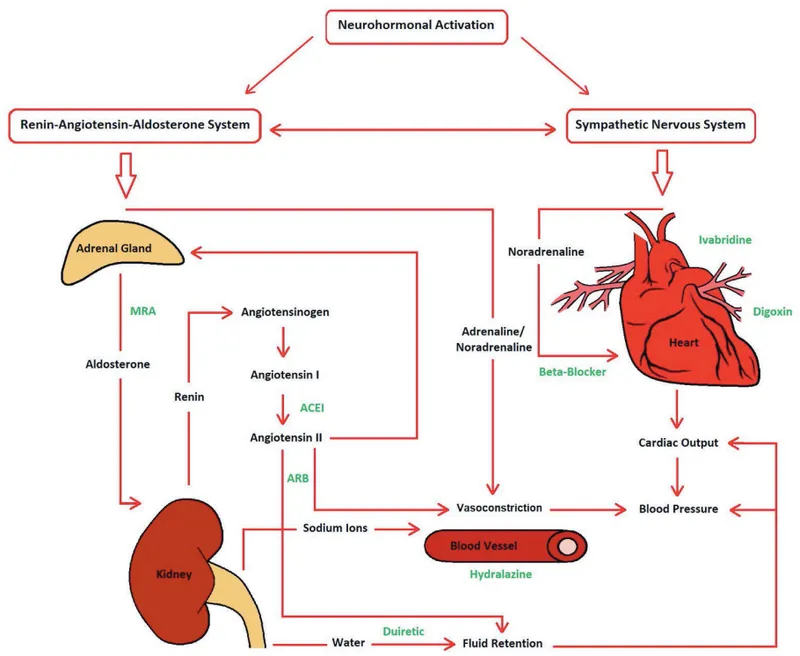
HFrEF treatment follows a stepwise approach with proven mortality benefits from neurohormonal blockade. The "fantastic four" medications form the cornerstone therapy with specific titration protocols and outcome targets.
-
ACE Inhibitors/ARBs
- Target doses: Lisinopril 20-40 mg daily, Enalapril 10-20 mg BID
- Mortality benefit: 20-25% reduction in cardiovascular death
- Titration: Start low, double dose every 2 weeks to maximum tolerated
- Monitor: Creatinine increase <30%, potassium <5.5 mEq/L
- Contraindications: Bilateral renal artery stenosis, angioedema history
- ARNi (Sacubitril/Valsartan): Superior to ACE-I with 16% additional mortality reduction
-
Beta-Blockers
- Evidence-based agents: Metoprolol succinate, carvedilol, bisoprolol
- Target doses: Metoprolol XL 200 mg daily, Carvedilol 25 mg BID
- Mortality benefit: 30-35% reduction in sudden cardiac death
- Start low: Metoprolol 12.5 mg BID, double every 2 weeks
- Monitor: Heart rate >60 bpm, SBP >90 mmHg, no decompensation
- Contraindications: Severe asthma, high-grade AV block, cardiogenic shock
📌 Remember: BEAM for HFrEF therapy - Beta-blockers, Enalapril/ARB, Aldosterone antagonists, Mineral corticoid receptor antagonists. Each provides independent mortality benefit with additive effects when combined appropriately.
| Medication Class | Mortality Reduction | Target Dose Achievement | Monitoring Parameters | Major Contraindications |
|---|---|---|---|---|
| ACE-I/ARB | 20-25% | <50% patients | Creatinine, K+ | Angioedema, pregnancy |
| Beta-blockers | 30-35% | <40% patients | HR, BP, symptoms | Severe asthma, AV block |
| MRA | 15-20% | <60% patients | K+, creatinine | K+ >5.0, CrCl <30 |
| SGLT2-I | 13-18% | <30% patients | eGFR, ketones | Type 1 DM, dialysis |
- Loop Diuretics
- Furosemide equivalents: Furosemide 40 mg = Bumetanide 1 mg = Torsemide 20 mg
- Dosing strategy: Continuous infusion superior to bolus dosing for acute decompensation
- Initial dose: 2.5x home oral dose as IV bolus
- Continuous infusion: 5-10 mg/hour furosemide with titration
- Monitoring: Daily weights, fluid balance, electrolytes, renal function
- Diuretic resistance: Combination therapy with thiazides or acetazolamide
⭐ Clinical Pearl: Torsemide shows superior bioavailability (80-90%) compared to furosemide (50-60%) and may provide better outcomes in chronic heart failure. Spironolactone 25 mg daily reduces mortality by 30% in NYHA Class III-IV patients with EF <35%.
Advanced therapies target patients with persistent symptoms despite optimal medical therapy. Device therapy and mechanical support provide additional survival benefits in selected populations.
💡 Master This: Guideline-directed medical therapy requires systematic titration to maximum tolerated doses before considering inadequate response. Combination therapy provides additive benefits - patients on ACE-I + beta-blocker + MRA show 50-60% mortality reduction compared to placebo. Early initiation and aggressive titration maximize clinical benefits.
These evidence-based algorithms ensure optimal therapeutic outcomes while minimizing adverse effects through systematic monitoring and dose optimization.
🌐 The Systems Integration Network: Multi-Organ Interactions
-
Type 1 Cardiorenal Syndrome (Acute)
- Mechanism: Acute cardiac dysfunction → ↓ renal perfusion → AKI
- Hemodynamics: ↓ cardiac output + ↑ venous pressure → ↓ renal blood flow
- Renal perfusion pressure: MAP - CVP determines glomerular filtration
- Critical threshold: MAP <65 mmHg → autoregulation failure
- Biomarkers: Creatinine rise >0.3 mg/dL within 48 hours
- Management: Optimize hemodynamics before aggressive diuresis
-
Type 2 Cardiorenal Syndrome (Chronic)
- Mechanism: Chronic heart failure → neurohormonal activation → progressive CKD
- Pathophysiology: RAAS activation → glomerular sclerosis + interstitial fibrosis
- eGFR decline: 2-4 mL/min/1.73m² annually in stable heart failure
- Proteinuria: >300 mg/day indicates glomerular damage
- Therapeutic challenge: ACE-I/ARB may worsen renal function initially
- Monitoring: eGFR, proteinuria, electrolytes every 3-6 months
📌 Remember: RIFLE criteria for cardiorenal syndrome - Risk (Cr ↑ 1.5x), Injury (Cr ↑ 2x), Failure (Cr ↑ 3x), Loss (persistent failure >4 weeks), End-stage (>3 months). Each stage carries increasing mortality risk from 15% to 60%.
| System | Normal Function | Heart Failure Impact | Compensatory Response | Therapeutic Target |
|---|---|---|---|---|
| Renal | eGFR >90 | ↓ perfusion, ↑ congestion | Na+ retention, ↑ RAAS | Optimize hemodynamics |
| Pulmonary | PCWP <12 mmHg | ↑ filling pressures | ↑ respiratory rate | Preload reduction |
| Hepatic | Normal synthesis | Congestion, ↓ perfusion | ↓ albumin, ↑ bilirubin | Venous decongestion |
| Neurohormonal | Balanced RAAS/SNS | Hyperactivation | Vasoconstriction, ↑ HR | Neurohormonal blockade |
-
Acute Pulmonary Edema
- Mechanism: PCWP >25 mmHg → alveolar flooding → impaired gas exchange
- Clinical presentation: Severe dyspnea, pink frothy sputum, O2 sat <90%
- Treatment: Immediate preload reduction with IV diuretics + vasodilators
- Monitoring: Arterial blood gas, chest X-ray, hemodynamic parameters
-
Chronic Pulmonary Hypertension
- Prevalence: 60-70% of advanced heart failure patients
- Mechanism: Chronic ↑ LA pressure → pulmonary vascular remodeling
- Passive component: Elevated PCWP → ↑ PA pressures
- Active component: Pulmonary vasoconstriction + vascular remodeling
- Prognosis: RVSP >50 mmHg → 2-fold ↑ mortality risk
- Treatment: Optimize left-sided filling pressures before pulmonary vasodilators
Hepatic congestion develops from elevated right-sided pressures and affects synthetic function, drug metabolism, and nutritional status. Cardiac hepatopathy can mimic primary liver disease and complicate medication dosing.
⭐ Clinical Pearl: Hepatojugular reflux indicates elevated right atrial pressure and hepatic congestion. ALT/AST elevation in acute heart failure suggests hepatic hypoperfusion, while chronic elevation indicates congestive hepatopathy. Albumin <3.0 g/dL predicts poor outcomes and increased readmission risk.
Neurohormonal integration involves complex feedback loops between sympathetic nervous system, RAAS, natriuretic peptides, and vasopressin. Therapeutic interventions must account for system interactions and compensatory responses.
💡 Master This: Multi-system optimization requires sequential intervention rather than simultaneous manipulation. Hemodynamic stabilization precedes aggressive neurohormonal blockade, while renal function preservation guides diuretic dosing. System interactions can create therapeutic conflicts - ACE inhibitors improve cardiac function but may worsen renal function in volume-depleted patients.
Understanding these complex interactions enables comprehensive management that addresses the full spectrum of heart failure pathophysiology.
🎯 The Clinical Mastery Arsenal: Rapid Assessment Tools
Rapid Clinical Assessment Framework enables bedside evaluation within 5-10 minutes using high-yield clinical indicators and point-of-care testing. This systematic approach provides immediate risk stratification and treatment prioritization.
-
60-Second Hemodynamic Assessment
- Perfusion status: Pulse pressure, capillary refill, mental status
- Adequate perfusion: Pulse pressure >25 mmHg, warm extremities
- Hypoperfusion: Pulse pressure <25 mmHg, cool extremities, altered mentation
- Volume status: JVP, hepatojugular reflux, peripheral edema
- Euvolemic: JVP <8 cm, no HJR, minimal edema
- Volume overloaded: JVP >10 cm, positive HJR, pitting edema
- Severity markers: Heart rate >100 bpm, SBP <90 mmHg, respiratory rate >20
- Perfusion status: Pulse pressure, capillary refill, mental status
-
Essential Laboratory Panel
- BNP/NT-proBNP: Rule-out if <100 pg/mL, confirm if >400 pg/mL
- Comprehensive metabolic panel: Creatinine, eGFR, electrolytes
- Troponin: Elevated in 70% of acute heart failure cases
- Complete blood count: Anemia present in 40% of patients
📌 Remember: CHAMP for rapid heart failure assessment - Congestion signs, Hemodynamic status, Activity tolerance, Medication compliance, Precipitating factors. Each component provides immediate clinical insight for treatment planning.
| Assessment Tool | Time Required | Sensitivity | Specificity | Clinical Application |
|---|---|---|---|---|
| Clinical examination | 2-3 minutes | 70% | 85% | Initial screening |
| BNP/NT-proBNP | 15-30 minutes | 95% | 60% | Diagnostic confirmation |
| Chest X-ray | 5-10 minutes | 80% | 85% | Congestion assessment |
| Echocardiogram | 15-20 minutes | 95% | 95% | Definitive diagnosis |
| Point-of-care US | 2-5 minutes | 90% | 90% | Rapid assessment |
-
MAGGIC Risk Score (Meta-Analysis Global Group in Chronic Heart Failure)
- Variables: Age, EF, NYHA class, creatinine, diabetes, COPD
- Score interpretation: <15 points = low risk, 15-25 = intermediate, >25 = high risk
- 1-year mortality: Low risk 2%, intermediate 8%, high risk 20%
- 3-year mortality: Low risk 7%, intermediate 20%, high risk 45%
-
ADHERE Risk Score (Acute Decompensated Heart Failure National Registry)
- Variables: BUN >43 mg/dL, creatinine >2.75 mg/dL, SBP <115 mmHg
- In-hospital mortality: 0 factors = 2.1%, 1 factor = 6.4%, 2-3 factors = 21.9%
- Clinical utility: Immediate risk stratification in emergency department
Treatment Response Monitoring requires systematic tracking of clinical indicators, biomarkers, and functional parameters. Early identification of treatment failure enables prompt intervention and prevents decompensation.
-
Clinical Response Indicators
- Weight loss: Target 1-2 kg daily during acute treatment
- Symptom improvement: Dyspnea relief within 24-48 hours
- Hemodynamic improvement: ↓ JVP, ↓ edema, improved perfusion
- Biomarker response: BNP reduction >30% indicates effective treatment
-
Treatment Failure Indicators
- Persistent symptoms: No improvement after 48-72 hours
- Worsening renal function: Creatinine ↑ >0.3 mg/dL
- Electrolyte disturbances: Hyponatremia <130 mEq/L, hyperkalemia >5.5 mEq/L
- Hemodynamic deterioration: Hypotension, worsening perfusion
⭐ Clinical Pearl: Serial BNP measurements provide superior prognostic information compared to single values. BNP reduction <30% during hospitalization predicts increased readmission risk and worse outcomes. Target BNP <400 pg/mL at discharge when possible.
Discharge Readiness Criteria ensure safe transition to outpatient management while minimizing readmission risk. Systematic evaluation of clinical stability guides discharge timing and follow-up planning.
💡 Master This: Clinical mastery combines rapid assessment skills with systematic monitoring and evidence-based decision-making. Pattern recognition develops through deliberate practice with immediate feedback. Prognostic awareness enables appropriate goal-setting and resource utilization while maintaining therapeutic optimism and patient-centered care.
These rapid assessment tools transform complex clinical scenarios into manageable, systematic evaluations that optimize patient outcomes through precision medicine approaches.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more
Have doubts about this lesson?
Ask Rezzy, your AI Study Mate, to explain anything you didn't understand
Everything you need for NEET-PG prep
Get full Oncourse access with lessons, practice questions, flashcards and AI study tools.
Scan to download app












