Endocrinology

On this page
🧬 Hormonal Command Center: The Endocrine Architecture
You'll master endocrinology by understanding how hormones orchestrate every physiological system through elegant feedback loops and intricate signaling networks. This lesson builds your clinical reasoning from molecular architecture through diagnostic interpretation to therapeutic decision-making, transforming abstract endocrine concepts into practical pattern recognition skills. You'll learn to navigate complex hormonal disorders by integrating laboratory data with clinical presentations, then apply precise interventions that restore metabolic balance and improve patient outcomes.
📌 Remember: FLAT PiG - Feedback loops, Lipophilic vs hydrophilic, Axis systems, Target organs, Pituitary control, Gland locations
The endocrine system's precision depends on three fundamental mechanisms: negative feedback loops (maintaining 95% of hormonal stability), receptor sensitivity (varying 1000-fold between tissues), and half-life kinetics (ranging from 3 minutes for insulin to 7 days for thyroid hormones).
- Hormone Classification Systems
- Peptide/Protein Hormones: 60% of all hormones, water-soluble, surface receptors
- Insulin: 6-minute half-life, immediate glucose effects
- Growth hormone: 20-minute half-life, hours for IGF-1 effects
- Steroid Hormones: 25% of hormones, lipophilic, nuclear receptors
- Cortisol: 90-minute half-life, hours for genomic effects
- Testosterone: 10-100 minutes, days for anabolic effects
- Amine Hormones: 15% of hormones, variable properties
- Epinephrine: 2-minute half-life, seconds for response
- T4: 7-day half-life, weeks for steady state
- Peptide/Protein Hormones: 60% of all hormones, water-soluble, surface receptors
| Hormone Class | Receptor Type | Response Time | Half-Life Range | Clinical Examples |
|---|---|---|---|---|
| Peptide | Surface/cAMP | Seconds-Minutes | 3-120 minutes | Insulin, Glucagon, ACTH |
| Steroid | Nuclear/Genomic | Hours-Days | 30-300 minutes | Cortisol, Aldosterone, Estrogen |
| Thyroid | Nuclear/T3 | Hours-Weeks | 1-7 days | T3, T4, rT3 |
| Catecholamine | Surface/G-protein | Seconds | 1-5 minutes | Epinephrine, Norepinephrine |
| Eicosanoid | Multiple | Minutes | 5-30 minutes | Prostaglandins, Leukotrienes |
💡 Master This: Receptor downregulation occurs with chronic exposure - 50% reduction in 24-48 hours explains why continuous hormone therapy often requires dose escalation
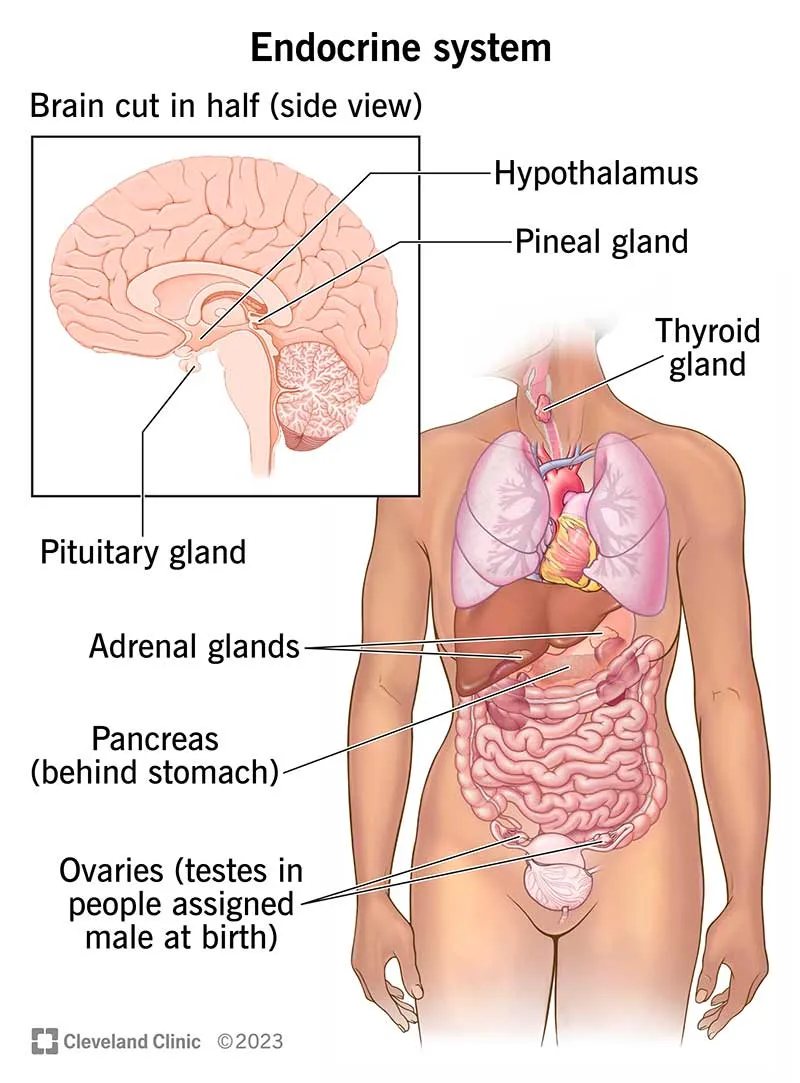
The hypothalamic-pituitary axis serves as the master control center, with the hypothalamus producing 9 releasing hormones that regulate anterior pituitary secretion of 6 major hormones, each controlling peripheral endocrine glands with precision feedback loops maintaining ±5% variation from set points.
Understanding endocrine temporal patterns reveals diagnostic clues: cortisol peaks at 8 AM (15-25 μg/dL), growth hormone surges during deep sleep (>10 ng/mL), and testosterone reaches maximum at 7 AM (300-1000 ng/dL in males). These circadian rhythms guide optimal sampling times and explain symptom patterns in endocrine disorders.
Connect this foundational architecture through feedback mechanisms to understand how hormonal imbalances create the clinical syndromes that define endocrine medicine.
🧬 Hormonal Command Center: The Endocrine Architecture
⚖️ Feedback Mastery: The Regulatory Symphony
📌 Remember: SHARP Feedback - Sensitivity varies, Hierarchical control, Amplification cascades, Rapid vs slow loops, Positive exceptions
Negative feedback governs 95% of endocrine regulation, with sensitivity varying 1000-fold between systems. The thyroid axis maintains TSH within 0.4-4.0 mIU/L through log-linear feedback, where 2-fold T4 changes produce 10-fold TSH changes, explaining why TSH serves as the most sensitive thyroid function marker.
- Feedback Loop Classifications
- Ultra-short loops: Seconds to minutes, local tissue regulation
- Insulin-glucose: 3-5 minute response time, ±20 mg/dL precision
- Parathyroid-calcium: 1-2 minute response, ±0.2 mg/dL control
- Short loops: Minutes to hours, pituitary self-regulation
- Growth hormone: 20-minute pulses, 3-hour cycles
- Prolactin: 90-minute oscillations, circadian modulation
- Long loops: Hours to days, hypothalamic-pituitary-target organ
- HPA axis: 24-hour cortisol rhythm, stress responsiveness
- HPG axis: 28-day cycles in females, seasonal variations
- Ultra-short loops: Seconds to minutes, local tissue regulation
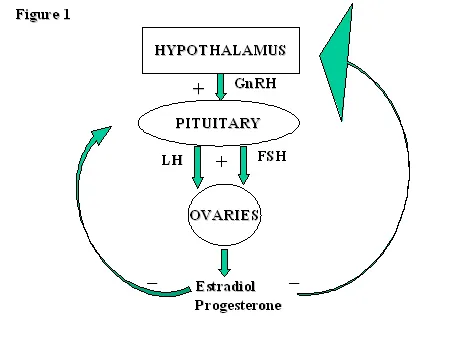
Positive feedback occurs in <5% of endocrine situations but creates dramatic clinical events: LH surge (10-fold increase) triggers ovulation, oxytocin amplification drives labor contractions, and parathyroid crisis can escalate calcium to >15 mg/dL without intervention.
| Feedback Type | Response Time | Sensitivity | Clinical Example | Pathology Pattern |
|---|---|---|---|---|
| Ultra-short | 1-5 minutes | High (±5%) | Glucose-Insulin | Diabetes, Hypoglycemia |
| Short | 20-120 minutes | Moderate (±15%) | GH Pulses | Acromegaly, Deficiency |
| Long | 6-48 hours | Variable (±25%) | Thyroid Axis | Hyper/Hypothyroidism |
| Positive | Minutes-Hours | Explosive | LH Surge | PCOS, Anovulation |
| Circadian | 24 hours | Rhythmic | Cortisol Pattern | Cushing's, Addison's |
💡 Master This: Set point alterations explain chronic diseases - insulin resistance shifts glucose set point upward, leptin resistance elevates weight set point, aging reduces GH and sex hormone set points
Amplification cascades multiply hypothalamic signals by 1000-10,000 fold through the pituitary-target organ pathway. TRH (nanogram quantities) stimulates TSH (microgram amounts) to produce T4 (milligram daily production), explaining how small hypothalamic lesions cause profound endocrine deficiencies.
Feedback sensitivity varies with physiological state: pregnancy reduces insulin sensitivity by 50%, stress increases cortisol set point 3-5 fold, aging decreases GH responsiveness 10% per decade after age 30. Understanding these adaptive changes distinguishes pathological from physiological hormone variations.
Connect these regulatory principles through clinical correlation patterns to understand how feedback disruption creates the diagnostic signatures of endocrine diseases.
⚖️ Feedback Mastery: The Regulatory Symphony
🎯 Pattern Recognition: The Clinical Detective Framework
📌 Remember: DETECT Patterns - Demographics matter, Exam findings cluster, Timing reveals clues, End-organ effects, Comorbidity patterns, Test interpretation
Demographic clustering provides powerful diagnostic clues: thyroid disorders affect women 8:1, type 1 diabetes peaks at ages 5-15, Cushing's occurs 3:1 in women aged 25-40, and pheochromocytoma follows the "rule of 10s" (10% bilateral, 10% malignant, 10% extra-adrenal).
- High-Yield Pattern Recognition Frameworks
- "See Fatigue, Think Thyroid": 80% of hypothyroidism presents with fatigue
- TSH >10 mIU/L: Overt hypothyroidism, treat immediately
- TSH 4-10 mIU/L: Subclinical, treat if symptomatic or pregnant
- Free T4 <0.8 ng/dL: Confirms tissue hypothyroidism
- "See Weight Gain + Purple Striae, Think Cushing's": 95% specificity
- Midnight cortisol >7.5 μg/dL: Abnormal circadian rhythm
- 24-hour urine cortisol >3x normal: Suggests Cushing's syndrome
- Dexamethasone suppression failure: Confirms autonomous cortisol
- "See Polyuria + Polydipsia, Think Diabetes": Classic triad in 60%
- Random glucose >200 mg/dL: Diagnostic with symptoms
- HbA1c >6.5%: Reflects 3-month average glucose
- Fasting glucose >126 mg/dL: Requires confirmation on separate day
- "See Fatigue, Think Thyroid": 80% of hypothyroidism presents with fatigue
Temporal patterns reveal diagnostic clues: morning stiffness suggests adrenal insufficiency, afternoon fatigue indicates cortisol deficiency, nocturnal symptoms point to pheochromocytoma, and menstrual irregularities signal reproductive endocrine dysfunction.
| Clinical Pattern | Key Demographics | Diagnostic Threshold | Confirmatory Test | Sensitivity/Specificity |
|---|---|---|---|---|
| Thyroid Fatigue | Female, 30-60 years | TSH >4.0 mIU/L | Free T4, Anti-TPO | 95%/85% |
| Cushing's Obesity | Female, 25-45 years | UFC >3x normal | Dexamethasone test | 90%/95% |
| Diabetes Polyuria | Any age, family Hx | Glucose >200 mg/dL | HbA1c, OGTT | 85%/98% |
| PCOS Hirsutism | Female, 15-35 years | Testosterone >70 ng/dL | Free androgen index | 80%/90% |
| Addison's Fatigue | Any age, autoimmune | Cortisol <5 μg/dL | ACTH stimulation | 95%/100% |
End-organ effect patterns provide objective evidence: thyroid eye disease confirms Graves' hyperthyroidism, acanthosis nigricans suggests insulin resistance, nephrolithiasis indicates hyperparathyroidism, and osteoporosis in young patients signals hypogonadism or hypercortisolism.
💡 Master This: Laboratory pattern recognition trumps individual values - suppressed TSH with elevated T4 suggests hyperthyroidism, elevated glucose with low C-peptide indicates type 1 diabetes, high calcium with high PTH confirms hyperparathyroidism
Comorbidity clustering reveals underlying endocrine disorders: diabetes + hypertension + obesity suggests metabolic syndrome, osteoporosis + kidney stones + depression indicates hyperparathyroidism, infertility + hirsutism + obesity points to PCOS.
Red flag patterns demand immediate evaluation: severe hyponatremia (<120 mEq/L) with hypotension suggests adrenal crisis, calcium >14 mg/dL indicates parathyroid crisis, glucose >600 mg/dL with altered mental status signals hyperosmolar coma.
Connect these recognition patterns through systematic diagnostic approaches to understand how clinical clustering guides efficient testing strategies and accurate diagnoses.
🎯 Pattern Recognition: The Clinical Detective Framework
🔬 Diagnostic Precision: The Laboratory Navigation System
📌 Remember: LABS Logic - Location of sampling, Assay limitations, Biological variation, Suppression/stimulation tests
Reference range interpretation requires understanding population statistics: 95% of healthy individuals fall within 2 standard deviations, but 5% of normal people have "abnormal" results. Age-specific ranges are crucial - testosterone declines 1-2% annually after age 30, TSH increases 0.02 mIU/L per decade, IGF-1 decreases 14% per decade.
- Critical Laboratory Interpretation Principles
- Biological Variation: Intra-individual variation often <inter-individual
- Cortisol: 50% daily variation, morning peak essential
- Testosterone: 20% day-to-day variation, morning sampling required
- TSH: 15% variation, consistent timing improves accuracy
- Assay Interference: 10-15% of hormone assays have interference
- Biotin supplementation: Falsely low TSH, high T4
- Heterophile antibodies: Falsely elevated multiple hormones
- Macro-hormones: Falsely elevated prolactin, TSH, CK
- Physiological States: Pregnancy, illness, medications alter ranges
- Pregnancy: TSH decreases first trimester, cortisol increases 3-fold
- Critical illness: Low T3 syndrome, suppressed reproductive hormones
- Medications: Biotin, steroids, dopamine affect multiple assays
- Biological Variation: Intra-individual variation often <inter-individual
Dynamic testing reveals functional capacity when baseline values are equivocal. Stimulation tests assess glandular reserve, suppression tests evaluate autonomous function, and provocative tests unmask subtle deficiencies.
| Test Type | Clinical Application | Normal Response | Abnormal Pattern | Diagnostic Accuracy |
|---|---|---|---|---|
| ACTH Stimulation | Adrenal insufficiency | Cortisol >18 μg/dL | <18 μg/dL at 60 min | 95% sensitivity |
| Dexamethasone Suppression | Cushing's syndrome | Cortisol <1.8 μg/dL | >1.8 μg/dL morning | 95% specificity |
| Oral Glucose Tolerance | Diabetes, IGT | 2-hr glucose <140 | 140-199 (IGT), >200 (DM) | 85% sensitivity |
| GnRH Stimulation | Hypogonadotropic hypogonadism | LH rise >3x baseline | Blunted response | 90% specificity |
| TRH Stimulation | TSH reserve | TSH rise 2-5x baseline | Absent/excessive rise | 80% diagnostic |
Timing considerations are crucial for accurate interpretation: cortisol must be drawn before 9 AM, growth hormone requires fasting or stimulation, reproductive hormones depend on menstrual cycle phase, and parathyroid hormone needs simultaneous calcium measurement.
💡 Master This: Pattern recognition in laboratory clusters - high calcium + high PTH = hyperparathyroidism, low sodium + high potassium = adrenal insufficiency, high glucose + low C-peptide = type 1 diabetes
Critical values requiring immediate action: glucose <40 or >600 mg/dL, sodium <120 or >160 mEq/L, calcium >14 mg/dL, cortisol <3 μg/dL with stress, TSH >50 mIU/L suggest life-threatening endocrine emergencies.
Quality assurance principles improve diagnostic accuracy: pre-analytical factors cause 60% of laboratory errors, proper collection techniques prevent hemolysis and contamination, appropriate storage maintains hormone stability, and clinical correlation identifies implausible results.
Connect these laboratory principles through treatment monitoring strategies to understand how serial testing guides therapeutic adjustments and outcome assessment.
🔬 Diagnostic Precision: The Laboratory Navigation System
💊 Treatment Mastery: The Therapeutic Command Center
📌 Remember: TREAT Smart - Target goals, Regular monitoring, Efficacy assessment, Adverse effects, Titration protocols
Hormone replacement requires physiological dosing that mimics normal patterns: levothyroxine 1.6 μg/kg daily achieves euthyroidism in >90%, hydrocortisone 15-25 mg daily in divided doses replaces cortisol, testosterone 100-200 mg weekly maintains normal levels.
- Evidence-Based Treatment Targets
- Diabetes Management: Individualized HbA1c goals based on patient factors
- Young, healthy: HbA1c <7.0% (<53 mmol/mol)
- Elderly, comorbid: HbA1c <8.0% (<64 mmol/mol)
- Pregnancy: HbA1c <6.0% (<42 mmol/mol)
- Thyroid Replacement: TSH normalization with symptom resolution
- Primary hypothyroidism: TSH 0.4-4.0 mIU/L
- Pregnancy: Trimester-specific TSH targets
- Elderly: TSH 4-6 mIU/L may be appropriate
- Lipid Management: Risk-based LDL targets
- Very high risk: LDL <55 mg/dL (<1.4 mmol/L)
- High risk: LDL <70 mg/dL (<1.8 mmol/L)
- Moderate risk: LDL <100 mg/dL (<2.6 mmol/L)
- Diabetes Management: Individualized HbA1c goals based on patient factors
Monitoring protocols ensure therapeutic efficacy and safety: HbA1c every 3 months for diabetes, TSH every 6-8 weeks during thyroid dose adjustment, lipid panels every 6-12 weeks after statin initiation.
| Condition | First-line Treatment | Target Goal | Monitoring Frequency | Adjustment Criteria |
|---|---|---|---|---|
| Type 2 Diabetes | Metformin 500-2000 mg | HbA1c <7% | Every 3 months | >0.5% from target |
| Hypothyroidism | Levothyroxine 1.6 μg/kg | TSH 0.4-4.0 mIU/L | 6-8 weeks | Outside normal range |
| Hyperlipidemia | Statin (moderate intensity) | LDL <70 mg/dL | 6-12 weeks | >20% from target |
| Osteoporosis | Bisphosphonate | T-score >-2.5 | Annual DXA | Continued bone loss |
| PCOS | Metformin + OCP | Regular cycles | Every 6 months | Persistent symptoms |
Combination therapy becomes necessary when monotherapy fails to achieve targets: diabetes often requires 2-3 medications for HbA1c <7%, resistant hypertension needs 3-4 agents, severe hyperlipidemia may require statin + ezetimibe + PCSK9 inhibitor.
💡 Master This: Treatment failure patterns guide next steps - primary failure suggests wrong diagnosis or severe disease, secondary failure indicates progression or non-adherence, partial response requires dose optimization
Adverse effect monitoring prevents serious complications: metformin requires annual creatinine monitoring, statins need baseline and follow-up liver enzymes, thiazolidinediones require heart failure surveillance, GLP-1 agonists need pancreatitis awareness.
Special populations require modified approaches: pregnancy demands tight glucose control (<95 mg/dL fasting, <140 mg/dL 1-hour post-meal), elderly patients need less aggressive targets to prevent hypoglycemia, kidney disease requires dose adjustments for renally cleared medications.
Connect these treatment principles through advanced integration concepts to understand how multi-system approaches and personalized medicine optimize long-term outcomes.
💊 Treatment Mastery: The Therapeutic Command Center
🌐 Systems Integration: The Endocrine Network Architecture
📌 Remember: NETWORK Integration - Neural connections, Endocrine axes, Tissue cross-talk, Whole-body effects, Organ interactions, Regulatory cascades, Kinetic relationships
Multi-system integration explains complex clinical presentations: metabolic syndrome involves insulin resistance affecting adipose tissue, liver metabolism, vascular function, and reproductive hormones. Polycystic ovary syndrome demonstrates reproductive-metabolic cross-talk where insulin resistance drives androgen excess and ovarian dysfunction.
- Critical Integration Patterns
- Hypothalamic-Pituitary-Adrenal-Thyroid Cross-talk
- Cortisol excess suppresses TSH by 30-50%, creating functional hypothyroidism
- Severe hypothyroidism impairs cortisol metabolism, requiring careful replacement
- Stress activates HPA axis while suppressing reproductive and growth axes
- Insulin-Growth Hormone-IGF-1 Network
- Insulin resistance reduces IGF-1 bioavailability by 40%
- Growth hormone excess causes insulin resistance in >80% of patients
- IGF-1 deficiency impairs glucose metabolism and protein synthesis
- Reproductive-Metabolic Integration
- Testosterone deficiency increases insulin resistance by 25%
- PCOS involves insulin-androgen positive feedback loops
- Menopause alters lipid metabolism and glucose homeostasis
- Hypothalamic-Pituitary-Adrenal-Thyroid Cross-talk
Tissue-specific hormone sensitivity creates differential responses: muscle shows high insulin sensitivity, liver responds strongly to glucagon, adipose tissue is cortisol-responsive, and bone integrates parathyroid, vitamin D, and sex hormone signals.
| Integration Pattern | Primary Hormones | Clinical Manifestation | Therapeutic Approach | Success Rate |
|---|---|---|---|---|
| Metabolic Syndrome | Insulin, Cortisol, Leptin | Diabetes + HTN + Obesity | Multi-drug, lifestyle | 70% target achievement |
| PCOS | Insulin, Androgens, LH | Hirsutism + Infertility | Metformin + Anti-androgen | 80% cycle regulation |
| Osteoporosis | PTH, Vitamin D, Estrogen | Fractures + Bone loss | Combination therapy | 85% fracture reduction |
| Thyroid-Adrenal | T3/T4, Cortisol | Fatigue + Cold intolerance | Sequential replacement | 90% symptom resolution |
| Growth-Metabolic | GH, IGF-1, Insulin | Short stature + Hypoglycemia | GH + Nutritional support | 75% catch-up growth |
Circadian integration coordinates multiple hormonal rhythms: cortisol peaks drive glucose production, melatonin onset promotes insulin sensitivity, growth hormone surges enhance protein synthesis, and reproductive hormones follow monthly or seasonal cycles.
💡 Master This: Systems thinking improves treatment outcomes - addressing insulin resistance in PCOS improves reproductive function, treating vitamin D deficiency enhances calcium absorption, correcting sleep disorders optimizes multiple hormone rhythms
Aging effects demonstrate system-wide integration: declining growth hormone (50% reduction by age 60) affects muscle mass, bone density, and metabolic rate. Reproductive hormone decline influences cardiovascular risk, bone health, and cognitive function. Insulin sensitivity decreases 6% per decade, affecting multiple metabolic pathways.
Stress integration reveals adaptive responses: acute stress mobilizes glucose and fatty acids while suppressing non-essential functions like reproduction and growth. Chronic stress leads to maladaptive changes including insulin resistance, immune suppression, and reproductive dysfunction.
Pharmacological integration requires understanding drug interactions: glucocorticoids antagonize insulin action, beta-blockers mask hypoglycemic symptoms, thiazides worsen glucose tolerance, and phenytoin accelerates vitamin D metabolism.
Connect these integration concepts through clinical mastery frameworks to understand how comprehensive endocrine assessment and personalized treatment strategies achieve optimal patient outcomes.
🌐 Systems Integration: The Endocrine Network Architecture
🎯 Clinical Mastery: The Endocrine Excellence Toolkit
📌 Remember: MASTER Endocrinology - Multi-system thinking, Accurate diagnosis, Systematic treatment, Target monitoring, Evidence-based care, Risk stratification
Essential Clinical Arsenal for immediate application:
- Critical Diagnostic Thresholds
- Diabetes: Fasting glucose ≥126 mg/dL, HbA1c ≥6.5%, Random glucose ≥200 mg/dL
- Thyroid: TSH 0.4-4.0 mIU/L, Free T4 0.8-1.8 ng/dL, Anti-TPO >35 IU/mL
- Adrenal: Morning cortisol <5 μg/dL (deficiency), >25 μg/dL (excess)
- Parathyroid: Calcium >10.5 mg/dL + PTH >65 pg/mL = hyperparathyroidism
- Reproductive: Testosterone <300 ng/dL (male hypogonadism), LH >25 IU/L (menopause)
| Clinical Scenario | Key Decision Point | Immediate Action | Target Timeline | Success Metric |
|---|---|---|---|---|
| DKA | pH <7.3, Glucose >250 | IV insulin + fluids | 12-24 hours | pH >7.3, Anion gap <12 |
| Thyroid Storm | HR >140, Temp >102°F | Beta-blocker + PTU | 24-48 hours | HR <100, Temp normal |
| Adrenal Crisis | BP <90, Na <130 | IV hydrocortisone 100mg | 1-2 hours | BP >100, Na >135 |
| Severe Hypoglycemia | Glucose <40 mg/dL | IV dextrose/glucagon | 15-30 minutes | Glucose >70 mg/dL |
| Hypercalcemic Crisis | Ca >14 mg/dL | IV saline + bisphosphonate | 24-72 hours | Ca <12 mg/dL |
Pattern Recognition Mastery: "When you see X, immediately consider Y"
- Fatigue + Weight gain + Cold intolerance = Hypothyroidism (TSH first)
- Polyuria + Polydipsia + Weight loss = Diabetes (glucose + ketones)
- Hypertension + Hypokalemia + Alkalosis = Primary aldosteronism (aldosterone/renin ratio)
- Amenorrhea + Galactorrhea + Headache = Prolactinoma (prolactin + MRI)
💡 Master This: Systematic approach prevents missed diagnoses - always consider endocrine causes in unexplained fatigue, weight changes, mood disorders, reproductive dysfunction, and metabolic abnormalities
Treatment Optimization Strategies:
- Start low, go slow for hormone replacement in elderly or cardiac patients
- Aggressive targets for young, healthy patients with diabetes or lipid disorders
- Combination therapy when monotherapy fails to achieve 50% of target improvement
- Regular monitoring every 3-6 months during dose adjustments
Long-term Outcome Predictors: HbA1c <7% reduces diabetic complications by 40%, TSH normalization improves cardiovascular outcomes by 25%, lipid targets decrease cardiac events by 30%, bone density improvement reduces fracture risk by 50%.
🎯 Clinical Mastery: The Endocrine Excellence Toolkit
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more
Have doubts about this lesson?
Ask Rezzy, your AI Study Mate, to explain anything you didn't understand
Everything you need for NEET-PG prep
Get full Oncourse access with lessons, practice questions, flashcards and AI study tools.
Scan to download app
























