Addiction Medicine

On this page
🧠 Addiction's Neural Architecture: The Hijacked Brain
Addiction commandeers the brain's reward pathways, transforming survival circuits into relapse engines through neuroadaptation, tolerance, and withdrawal that persist long after substances leave the body. You'll trace how repeated drug exposure rewires neural architecture, why cravings ambush patients years into recovery, and how genetic variants load the dice before first use. By mastering the neurobiological cascade from dopamine hijacking to receptor downregulation, you'll recognize addiction as a chronic brain disease demanding evidence-based intervention, not moral judgment. This foundation transforms how you diagnose, treat, and support patients trapped in cycles their conscious mind alone cannot break.
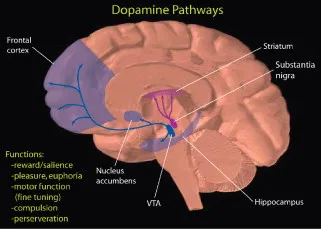
📌 Remember: DRIVE pathway - Dopamine from Reward circuits Initiates Ventral tegmental area Excitation. VTA neurons project to nucleus accumbens (75% of reward processing), prefrontal cortex (executive control), and amygdala (emotional memory).
The mesolimbic dopamine system forms addiction's foundation through three interconnected regions:
-
Ventral Tegmental Area (VTA)
- Primary dopamine cell bodies (65,000 neurons)
- Projects to multiple brain regions
- Responds to reward prediction errors
- Natural rewards: 150-200% baseline dopamine
- Cocaine: 300-400% baseline dopamine
- Amphetamines: 1000% baseline dopamine
-
Nucleus Accumbens (NAc)
- Core and shell subdivisions
- Integrates reward and motivation signals
- Medium spiny neurons comprise 95% of cells
- Express D1 and D2 dopamine receptors
- Gate reward-seeking behavior
- Show 40-60% volume reduction in chronic addiction
-
Prefrontal Cortex (PFC)
- Executive control and decision-making
- Inhibits impulsive behaviors
- Shows 20-30% decreased activity in addiction
- Dorsolateral PFC: working memory and planning
- Orbitofrontal cortex: reward valuation
- Anterior cingulate: conflict monitoring
| Brain Region | Normal Function | Addiction Changes | Recovery Timeline | Clinical Significance |
|---|---|---|---|---|
| VTA | Reward prediction | 300-1000% ↑ firing | 6-12 months | Craving intensity |
| Nucleus Accumbens | Motivation gating | 40-60% ↓ volume | 12-24 months | Anhedonia severity |
| Prefrontal Cortex | Executive control | 20-30% ↓ activity | 6-18 months | Relapse risk |
| Amygdala | Fear/stress response | 50-100% ↑ reactivity | 3-12 months | Stress-induced craving |
| Hippocampus | Memory formation | 15-25% ↓ volume | 12-36 months | Learning impairment |
💡 Master This: The opponent-process theory explains tolerance and withdrawal. Initial drug use creates positive hedonic state (+3), followed by negative opponent process (-1), resulting in net pleasure (+2). With repeated use, opponent process strengthens to (-5), requiring drug just to feel normal (0), while withdrawal creates severe negative state (-5).
Neuroplasticity changes occur within hours to days of first substance exposure, but full circuit hijacking requires weeks to months of repeated use. These changes persist months to years after cessation, explaining why addiction is a chronic relapsing condition requiring long-term management strategies.
The transition from voluntary drug use to compulsive addiction involves measurable shifts in neural control from ventral to dorsal striatum, representing a progression from goal-directed to habitual behavior patterns that form the neurobiological foundation for evidence-based treatment approaches.
🧠 Addiction's Neural Architecture: The Hijacked Brain
⚡ Neuroadaptation Cascade: The Brain's Chemical Rebellion
📌 Remember: ADAPT mechanisms - Axon sprouting, Dendritic remodeling, Astrocyte activation, Protein synthesis changes, Transcriptional modifications. These changes occur within 2-7 days of repeated exposure and persist 6-24 months after cessation.
Tolerance develops through multiple mechanisms operating on different timelines:
-
Acute Tolerance (Minutes-Hours)
- Receptor desensitization
- 50-70% reduced response within 30 minutes
- Involves protein kinase phosphorylation
- cAMP-dependent protein kinase activation
- Receptor internalization and degradation
- Immediate functional tolerance
-
Chronic Tolerance (Days-Weeks)
- Receptor downregulation: 40-80% reduction
- Compensatory upregulation of opposing systems
- Structural synaptic changes
- Dendritic spine density ↓ 20-40%
- Synaptic strength ↓ 30-60%
- New protein synthesis required
-
Behavioral Tolerance (Weeks-Months)
- Learned compensatory responses
- Environmental conditioning effects
- Context-dependent tolerance development
- Same dose less effective in familiar environment
- Higher overdose risk in novel settings
- Conditioned compensatory responses
| Adaptation Type | Timeline | Mechanism | Recovery Period | Clinical Impact |
|---|---|---|---|---|
| Receptor Desensitization | Minutes | Phosphorylation | Hours | Acute tolerance |
| Receptor Downregulation | Days | Protein degradation | Weeks | Chronic tolerance |
| Synaptic Remodeling | Weeks | Structural changes | Months | Persistent vulnerability |
| Epigenetic Changes | Months | Gene expression | Years | Transgenerational risk |
| Circuit Reorganization | Years | Network plasticity | Decades | Lifelong susceptibility |
Physical dependence emerges through opponent-process mechanisms involving multiple neurotransmitter systems:
-
Dopamine System Adaptations
- Baseline dopamine ↓ 40-60% below normal
- D2 receptor density ↓ 20-30%
- Dopamine transporter ↑ 50-100%
- Faster dopamine clearance
- Reduced reward sensitivity (anhedonia)
- Requires drug to achieve normal mood
-
GABA System Changes
- GABA receptor downregulation 30-50%
- Compensatory glutamate upregulation
- Explains alcohol/benzodiazepine withdrawal seizures
- Loss of inhibitory control
- Hyperexcitability and anxiety
- Life-threatening complications possible
-
Stress System Activation
- HPA axis dysregulation
- Cortisol levels ↑ 200-400% during withdrawal
- CRF (corticotropin-releasing factor) ↑ 300-500%
- Drives negative emotional states
- Triggers stress-induced relapse
- Persists 6-12 months in recovery
💡 Master This: Allostatic load concept explains addiction chronicity. Brain attempts to maintain stability (homeostasis) through adaptations that ultimately create new "abnormal normal" state (allostasis). Recovery requires 12-24 months for partial normalization, with some changes potentially permanent.
Withdrawal syndromes reflect the unmasking of these neuroadaptive changes when drug levels drop, creating predictable symptom clusters that guide evidence-based medical management protocols rather than symptomatic treatment approaches.
⚡ Neuroadaptation Cascade: The Brain's Chemical Rebellion
🎯 Craving Circuitry: The Relapse Command Center
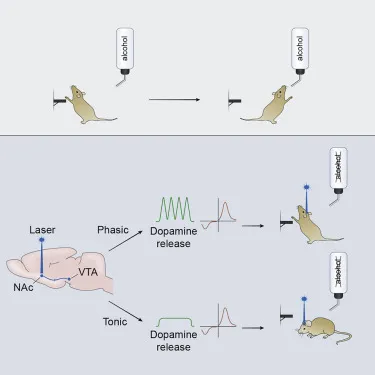
Craving emerges through three primary neural circuits operating in parallel:
-
Incentive Salience Circuit
- Attributes motivational significance to drug cues
- Involves nucleus accumbens and dorsal striatum
- "Wanting" without "liking" phenomenon
- Dopamine drives motivation, not pleasure
- Explains continued use despite negative consequences
- Can persist years after cessation
-
Executive Control Circuit
- Prefrontal cortex regulation of impulses
- Shows 20-40% decreased activation during craving
- Cognitive control vs. automatic responses
- Working memory deficits during craving episodes
- Impaired decision-making capacity
- Response inhibition ↓ 30-50%
-
Stress/Negative Affect Circuit
- Amygdala-hypothalamic activation
- Drives negative reinforcement seeking
- Stress-induced craving most common relapse trigger
- 60-80% of relapses follow stressful events
- Cortisol elevation predicts craving intensity
- Explains higher relapse rates during life transitions
📌 Remember: CRAVE triggers - Cues (environmental), Restlessness (internal states), Anxiety (negative emotions), Vulnerability (HALT: Hungry, Angry, Lonely, Tired), Exposure (social situations). Each trigger activates different neural circuits requiring specific intervention strategies.
| Craving Type | Neural Substrate | Trigger | Duration | Intervention Target |
|---|---|---|---|---|
| Cue-Induced | Amygdala-NAc | Environmental | 5-20 minutes | Cue exposure therapy |
| Stress-Induced | HPA axis | Life events | Hours-days | Stress management |
| Protracted | PFC-striatum | Abstinence | Weeks-months | Medication support |
| Priming | VTA-dopamine | Drug exposure | Minutes-hours | Relapse prevention |
| Mood-Induced | Limbic system | Depression/anxiety | Variable | Dual diagnosis treatment |
Conditioned craving responses develop through classical conditioning mechanisms:
-
Cue-Craving Associations
- Form within 5-10 exposures to drug-environment pairings
- Strengthen over months to years of use
- Persist indefinitely without extinction training
- Environmental cues trigger automatic responses
- Pavlovian-to-instrumental transfer drives behavior
- Explains sudden relapse after prolonged sobriety
-
Context-Dependent Learning
- Location-specific craving responses
- Social cues particularly powerful triggers
- Time-of-day conditioning effects
- Morning coffee → cocaine craving
- Weekend socializing → alcohol craving
- Work stress → prescription drug craving
-
Interoceptive Conditioning
- Internal bodily states become triggers
- Mood states, fatigue, physical sensations
- Negative emotional states most potent
- Depression increases craving 300-500%
- Anxiety triggers craving in 70-90% of patients
- Sleep deprivation amplifies all craving types
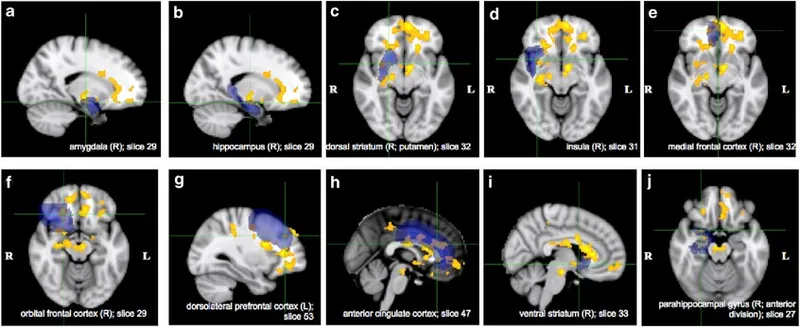
💡 Master This: Incubation of craving phenomenon shows craving intensity actually increases during early abstinence, peaking at 2-6 weeks before gradually declining. This explains why relapse risk remains high despite initial motivation and why intensive support during early recovery is critical.
Craving episodes follow predictable patterns with identifiable phases: trigger exposure → attention bias → physiological arousal → cognitive preoccupation → behavioral urges. Understanding this sequence enables targeted interventions at each phase, transforming overwhelming urges into manageable clinical symptoms with specific evidence-based treatments.
🎯 Craving Circuitry: The Relapse Command Center
🔬 Tolerance Mechanisms: The Diminishing Returns Dilemma
Tolerance develops through four distinct mechanisms operating simultaneously:
-
Pharmacokinetic Tolerance
- Enhanced drug metabolism and elimination
- Liver enzyme induction (CYP450 system)
- Metabolic tolerance develops within 3-7 days
- Alcohol: 30-50% faster metabolism after chronic use
- Barbiturates: 200-400% increased clearance
- Requires higher doses to achieve same blood levels
-
Pharmacodynamic Tolerance
- Receptor-level adaptations at target sites
- Receptor downregulation: 40-80% reduction in density
- Desensitization: 50-70% reduced responsiveness
- G-protein uncoupling from receptors
- Altered second messenger systems
- Changes in ion channel function
-
Behavioral Tolerance
- Learned compensatory responses
- Practice effects and environmental conditioning
- Functional tolerance through adaptation
- Motor skills compensation during intoxication
- Cognitive strategies to mask impairment
- Context-dependent tolerance effects
-
Cross-Tolerance
- Shared tolerance between similar drugs
- Same receptor system involvement
- Clinical significance for treatment planning
- Benzodiazepines and alcohol (GABA receptors)
- Opioids share 70-90% cross-tolerance
- Stimulants show partial cross-tolerance
📌 Remember: TAPER principles - Tolerance develops rapidly, Abrupt cessation dangerous, Protracted withdrawal possible, Escalation inevitable, Receptor changes persist. Medical supervision required for high-tolerance patients discontinuing CNS depressants.
| Substance Class | Tolerance Timeline | Mechanism | Peak Tolerance | Recovery Period |
|---|---|---|---|---|
| Alcohol | 3-7 days | GABA downregulation | 5-10x baseline | 2-4 weeks |
| Opioids | 1-3 days | Receptor desensitization | 10-100x baseline | 4-12 weeks |
| Benzodiazepines | 2-4 weeks | GABA receptor changes | 3-5x baseline | 6-18 months |
| Stimulants | Hours-days | Dopamine depletion | 2-5x baseline | 1-4 weeks |
| Nicotine | Hours | Receptor upregulation | 2-3x baseline | 2-4 weeks |
| %%{init: {'flowchart': {'htmlLabels': true}}}%% | ||||
| flowchart TD |
Start["💊 Drug Exposure
• Chronic dosing• Multiple doses"]
Act["🧪 Activation
• Receptor binding• Signal cascade"]
Comp["⚖️ Compensation
• Body adjustment• Homeostasis"]
Type{"📋 Tolerance?
• Classify logic• Pick pathway"}
MetInd["🧬 Enzymes
• Enzyme induction• Liver metabolic"]
MetClear["⚡ Clearance
• Faster removal• ⬇️ Half-life"]
RecDown["⬇️ Receptors
• Downregulation• ⬇️ Number bound"]
RecSens["📉 Sensitivity
• Reduced effect• ⬇️ Response"]
BehLearn["🧠 Learning
• Adaptation• Habituation"]
BehComp["🚶 Behavior
• Compensation• Skill learning"]
Dose["⚠️ Escalation
• ⬆️ Dose needed• Clinical need"]
Start --> Act Act --> Comp Comp --> Type
Type -->|Metabolic| MetInd MetInd --> MetClear MetClear --> Dose
Type -->|Receptor| RecDown RecDown --> RecSens RecSens --> Dose
Type -->|Behavioral| BehLearn BehLearn --> BehComp BehComp --> Dose
style Start fill:#F1FCF5, stroke:#BEF4D8, stroke-width:1.5px, rx:12, ry:12, color:#166534 style Act fill:#FFF7ED, stroke:#FFEED5, stroke-width:1.5px, rx:12, ry:12, color:#C2410C style Comp fill:#F7F5FD, stroke:#F0EDFA, stroke-width:1.5px, rx:12, ry:12, color:#6B21A8 style Type fill:#FEF8EC, stroke:#FBECCA, stroke-width:1.5px, rx:12, ry:12, color:#854D0E style MetInd fill:#F1FCF5, stroke:#BEF4D8, stroke-width:1.5px, rx:12, ry:12, color:#166534 style MetClear fill:#F1FCF5, stroke:#BEF4D8, stroke-width:1.5px, rx:12, ry:12, color:#166534 style RecDown fill:#F7F5FD, stroke:#F0EDFA, stroke-width:1.5px, rx:12, ry:12, color:#6B21A8 style RecSens fill:#F7F5FD, stroke:#F0EDFA, stroke-width:1.5px, rx:12, ry:12, color:#6B21A8 style BehLearn fill:#EEFAFF, stroke:#DAF3FF, stroke-width:1.5px, rx:12, ry:12, color:#0369A1 style BehComp fill:#EEFAFF, stroke:#DAF3FF, stroke-width:1.5px, rx:12, ry:12, color:#0369A1 style Dose fill:#FDF4F3, stroke:#FCE6E4, stroke-width:1.5px, rx:12, ry:12, color:#B91C1C
> ⭐ **Clinical Pearl**: **Acute vs. Chronic Tolerance** distinction is critical. Acute tolerance develops within **single dosing session** (**30 minutes** for alcohol), while chronic tolerance requires **repeated exposures** over **days to weeks**. This explains why binge patterns can be more dangerous than steady use.
**Reverse tolerance (sensitization)** occurs with some substances and effects:
* **Stimulant Sensitization**
- **Opposite** of tolerance for some effects
- **Locomotor activity** and **psychosis risk** increase
- **Kindling-like phenomenon**
+ Lower doses trigger stronger responses
+ Explains cocaine-induced seizures
+ Persists **months to years** after cessation
* **Alcohol Kindling**
- **Withdrawal severity** increases with repeated episodes
- **Seizure threshold** progressively lowers
- **Cognitive impairment** accumulates
+ Each withdrawal episode causes brain damage
+ **30-40%** increased seizure risk per episode
+ Medical supervision increasingly critical
> 💡 **Master This**: **Protracted tolerance** can persist **months to years** after cessation, explaining why former users remain vulnerable to overdose if they relapse using previous tolerance-level doses. **Tolerance loss** occurs **faster** than tolerance development, creating dangerous overdose window during early relapse.
Understanding tolerance mechanisms guides **medication-assisted treatment** strategies, explaining why **gradual dose reduction** prevents dangerous withdrawal while **cross-tolerant medications** enable safe detoxification from high-tolerance states that would otherwise require life-threatening abrupt cessation.
🔬 Tolerance Mechanisms: The Diminishing Returns Dilemma
💊 Withdrawal Neurobiology: The Brain's Revolt
Withdrawal involves three overlapping phases with distinct neurobiological signatures:
-
Acute Withdrawal (Hours-Days)
- Rebound hyperexcitability from suppressed systems
- Life-threatening complications possible
- Medical emergency requiring monitoring
- Alcohol: seizures within 6-48 hours
- Benzodiazepines: seizures 1-14 days
- Opioids: peak symptoms 48-72 hours
-
Protracted Withdrawal (Weeks-Months)
- Persistent neuroadaptive changes
- Subtle but significant functional impairments
- High relapse risk period
- Sleep disturbances 3-6 months
- Mood dysregulation 6-12 months
- Cognitive deficits 12-24 months
-
Post-Acute Withdrawal (Months-Years)
- Episodic symptom recurrence
- Stress-triggered symptom flares
- Lifelong vulnerability to certain triggers
- Environmental cue reactivity
- Stress-induced craving episodes
- Sleep and mood instability
📌 Remember: WITHDRAWAL severity predictors - Weekly consumption amount, Intoxication frequency, Tolerance level, History of prior withdrawals, Duration of use, Rapid cessation, Age and health status, Warning signs present, Alcohol blood level, Last use timing. Each factor increases complication risk exponentially.
| Substance | Onset | Peak | Duration | Life-Threatening Complications | Mortality Risk |
|---|---|---|---|---|---|
| Alcohol | 6-24 hours | 24-72 hours | 5-7 days | Seizures, DTs, arrhythmias | 5-15% untreated |
| Benzodiazepines | 1-7 days | 7-14 days | 2-8 weeks | Seizures, hyperthermia | 1-4% untreated |
| Opioids | 6-12 hours | 48-72 hours | 7-14 days | Dehydration, cardiac stress | <1% with support |
| Stimulants | Hours | 1-3 days | 1-2 weeks | Depression, suicidality | Variable |
| Barbiturates | 12-24 hours | 2-4 days | 2-3 weeks | Seizures, hyperthermia | 10-20% untreated |
| %%{init: {'flowchart': {'htmlLabels': true}}}%% | |||||
| flowchart TD |
Start["🛑 Drug Cessation
• Stop drug intake• Trigger response"]
Comp["🧠 Compensatory
• Systems unmasked• Loss of balance"]
Type{"❓ Withdrawal Type
• Identify class• Path physiologic"}
CNS["⚡ Hyperexcitability
• CNS Depressant• GABA/Glutamate"]
Seize["⚠️ Seizure Risk
• High risk state• Neural storm"]
Stim["😴 Depression
• Stimulant crash• Severe fatigue"]
Suicide["⚠️ Suicide Risk
• Mood instability• Mental health"]
Opioid["💓 Autonomic
• Opioid effect• Vital sign changes"]
Cardio["🫀 Cardio Stress
• High HR/BP• Vascular strain"]
Med["💊 Management
• Medical therapy• Clinical care"]
Start --> Comp Comp --> Type
Type -->|CNS Depressant| CNS CNS --> Seize Seize --> Med
Type -->|Stimulant| Stim Stim --> Suicide Suicide --> Med
Type -->|Opioid| Opioid Opioid --> Cardio Cardio --> Med
style Start fill:#F1FCF5, stroke:#BEF4D8, stroke-width:1.5px, rx:12, ry:12, color:#166534 style Comp fill:#F7F5FD, stroke:#F0EDFA, stroke-width:1.5px, rx:12, ry:12, color:#6B21A8 style Type fill:#FEF8EC, stroke:#FBECCA, stroke-width:1.5px, rx:12, ry:12, color:#854D0E style CNS fill:#F7F5FD, stroke:#F0EDFA, stroke-width:1.5px, rx:12, ry:12, color:#6B21A8 style Stim fill:#F7F5FD, stroke:#F0EDFA, stroke-width:1.5px, rx:12, ry:12, color:#6B21A8 style Opioid fill:#F7F5FD, stroke:#F0EDFA, stroke-width:1.5px, rx:12, ry:12, color:#6B21A8 style Seize fill:#FDF4F3, stroke:#FCE6E4, stroke-width:1.5px, rx:12, ry:12, color:#B91C1C style Suicide fill:#FDF4F3, stroke:#FCE6E4, stroke-width:1.5px, rx:12, ry:12, color:#B91C1C style Cardio fill:#FDF4F3, stroke:#FCE6E4, stroke-width:1.5px, rx:12, ry:12, color:#B91C1C style Med fill:#F1FCF5, stroke:#BEF4D8, stroke-width:1.5px, rx:12, ry:12, color:#166534
> ⭐ **Clinical Pearl**: **Delirium Tremens (DTs)** occurs in **5-10%** of alcohol withdrawal cases but carries **15-20%** mortality if untreated. **Predictors** include: prior DTs (**50%** recurrence), seizure history, **>10 drinks/day**, concurrent illness, and **rapid cessation**. **CIWA-Ar scores >15** indicate high-risk withdrawal.
**Alcohol Withdrawal Neurobiology** involves multiple neurotransmitter systems:
* **GABA System Dysfunction**
- **Chronic alcohol** enhances GABA (inhibitory)
- **Compensatory downregulation** of GABA receptors
- **Withdrawal** unmasks **hyperexcitability**
+ Seizure threshold ↓ **60-80%**
+ Anxiety and agitation
+ Autonomic instability
* **Glutamate System Hyperactivity**
- **Compensatory upregulation** during chronic use
- **Withdrawal** creates **excitotoxicity**
- **NMDA receptor** hyperactivation
+ Contributes to seizures and DTs
+ Cognitive impairment
+ Potential brain damage
* **Noradrenergic Hyperactivity**
- **Sympathetic nervous system** overdrive
- **Catecholamine surge** during withdrawal
- **Clinical manifestations**
+ Hypertension (**180/110** common)
+ Tachycardia (**>120 bpm**)
+ Hyperthermia and diaphoresis
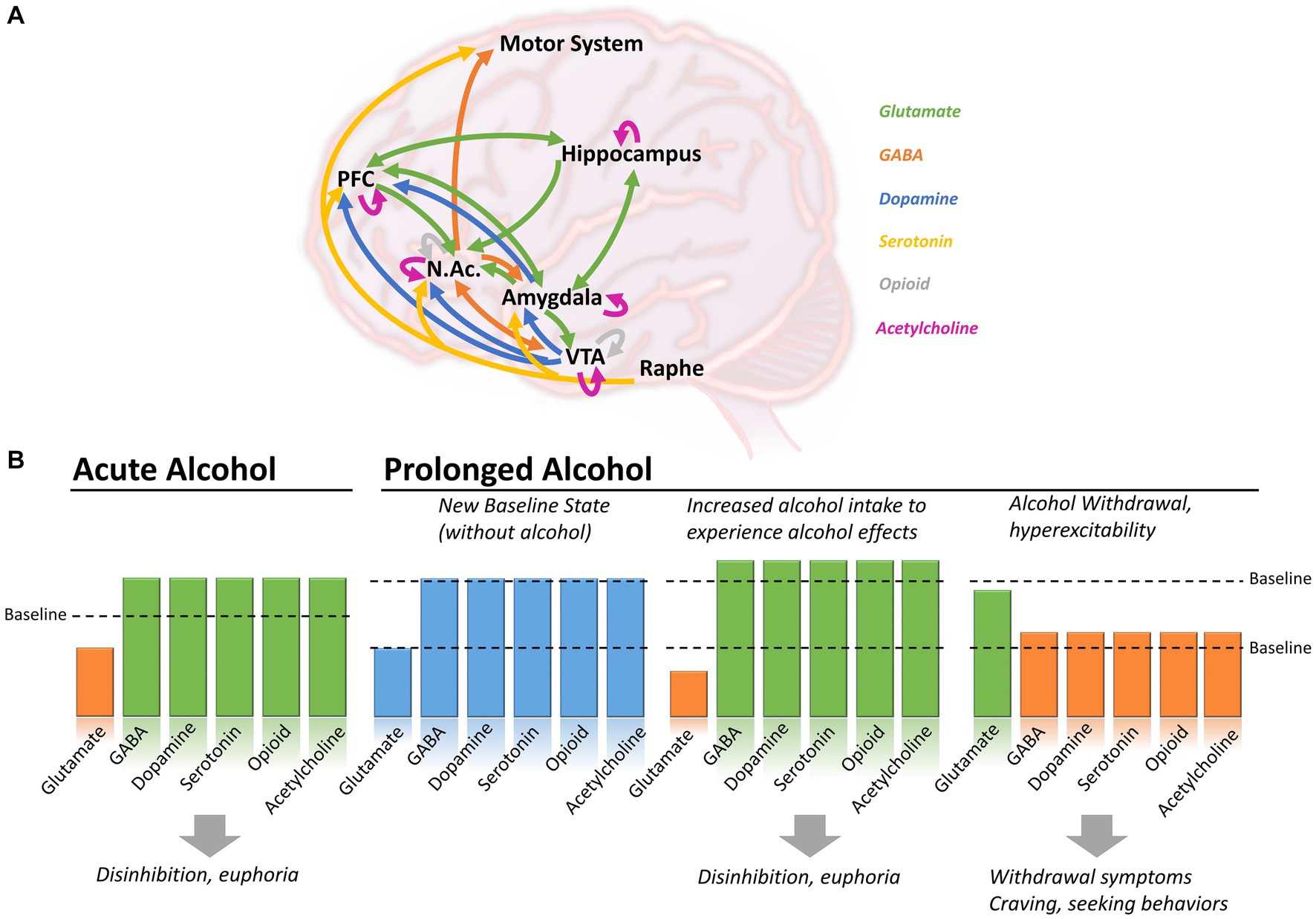
> 💡 **Master This**: **Kindling phenomenon** in withdrawal means each episode becomes **progressively more severe**. **Seizure risk** increases **30-40%** with each prior withdrawal, and **cognitive recovery** becomes **increasingly incomplete**. This explains why **medical detoxification** becomes more critical with repeated attempts.
**Protracted Withdrawal Syndrome** involves **persistent neuroadaptive changes**:
* **Dopamine System Dysfunction**
- **Baseline dopamine** ↓ **40-60%** for **months**
- **Anhedonia** and **depression** common
- **Relapse vulnerability** remains high
+ Normal activities provide insufficient reward
+ Drug cues trigger intense craving
+ Stress sensitivity markedly increased
* **HPA Axis Dysregulation**
- **Cortisol** levels remain **elevated** for **weeks**
- **Stress response** exaggerated and prolonged
- **Sleep architecture** disrupted for **months**
+ REM sleep ↓ **30-50%**
+ Sleep efficiency ↓ **20-40%**
+ Insomnia perpetuates relapse risk
Understanding withdrawal as **medical emergency** rather than **character weakness** enables **evidence-based protocols** that reduce **mortality from 15-20%** to **<1%** while providing foundation for **long-term recovery** through **neurobiologically-informed** treatment approaches.
💊 Withdrawal Neurobiology: The Brain's Revolt
🧬 Genetic Vulnerability: The Hereditary Hijack
Heritability estimates demonstrate strong genetic components across substance classes:
- Alcohol Use Disorder: 50-60% heritable
- Opioid Use Disorder: 40-60% heritable
- Cocaine Use Disorder: 65-79% heritable
- Nicotine Dependence: 40-70% heritable
- Cannabis Use Disorder: 45-75% heritable
📌 Remember: GENES influence addiction - Genetic variants affect Enzyme function, Neurotransmitter systems, Environmental sensitivity, Stress response. Twin studies show identical twins have 2-3x higher concordance than fraternal twins, confirming genetic contribution independent of shared environment.
Key genetic variants with established clinical significance:
| Gene | Variant | Function | Population Frequency | Clinical Impact |
|---|---|---|---|---|
| ALDH2 | *2 allele | Alcohol metabolism | 40% East Asian | 90% ↓ alcoholism risk |
| ADH1B | His47Arg | Alcohol metabolism | 70% East Asian | 3-5x ↓ alcoholism risk |
| OPRM1 | A118G | Opioid receptor | 15-30% European | 2x ↑ opioid addiction |
| DRD2 | TaqIA | Dopamine receptor | 30-35% global | 1.5-2x ↑ addiction risk |
| COMT | Val158Met | Dopamine metabolism | 25% Met/Met | Stress sensitivity ↑ |
Dopamine system genetics significantly influence addiction vulnerability:
-
DRD2 Gene Variants
- TaqIA polymorphism reduces D2 receptor density by 30-40%
- Reward deficiency syndrome hypothesis
- Higher addiction risk across multiple substances
- 2x increased alcohol dependence risk
- 1.5x increased cocaine addiction risk
- Earlier age of onset by 2-3 years
-
DAT1 Gene (Dopamine Transporter)
- VNTR polymorphisms affect dopamine clearance
- 9-repeat allele associated with ADHD and stimulant response
- 10-repeat allele linked to cocaine addiction vulnerability
- Faster dopamine clearance
- Reduced reward sensitivity
- Higher stimulant self-administration
-
COMT Gene (Catechol-O-Methyltransferase)
- Val158Met polymorphism affects dopamine metabolism
- Met/Met genotype: slower dopamine clearance
- Val/Val genotype: faster dopamine clearance
- Met/Met: stress-sensitive, anxiety-prone
- Val/Val: stress-resilient, executive function
- Influences treatment response patterns
💡 Master This: Gene-environment interactions are critical - genetic variants create vulnerability, but environmental factors determine expression. High-risk genotypes may show no addiction in protective environments, while low-risk genotypes can develop severe addiction with extreme exposure.
Pharmacogenomics applications in addiction treatment:
-
Opioid Metabolism Variants
- CYP2D6 polymorphisms affect codeine and tramadol conversion
- Poor metabolizers (7% European): no analgesic effect
- Ultra-rapid metabolizers (1-2% European): overdose risk
- 29% Ethiopian population ultra-rapid
- Genetic testing guides opioid selection
- Morphine preferred for poor metabolizers
-
Alcohol Treatment Response
- OPRM1 A118G variant affects naltrexone response
- G-allele carriers show better naltrexone response
- 2-3x greater relapse prevention efficacy
- Personalized treatment selection
- Pharmacogenetic testing cost-effective
- Precision medicine approach emerging
-
Nicotine Metabolism
- CYP2A6 variants affect nicotine clearance
- Slow metabolizers: lower addiction risk, better quit rates
- Fast metabolizers: higher dependence, more intensive treatment
- Nicotine replacement dosing adjustments
- Bupropion more effective in fast metabolizers
- Varenicline response genotype-independent
Understanding genetic contributions enables risk stratification, early intervention for high-risk individuals, and personalized treatment selection based on individual genetic profiles rather than trial-and-error approaches that delay effective care.
🧬 Genetic Vulnerability: The Hereditary Hijack
🎯 Addiction Mastery: Clinical Command Arsenal
📌 Remember: ASSESS framework - Acute intoxication level, Suicidality and safety, Substance use history, Emergent medical issues, Severity of dependence, Support systems available. Complete assessment within first 30 minutes determines immediate management priorities.
Essential Clinical Thresholds for immediate decision-making:
-
Alcohol Withdrawal Risk Stratification
- CIWA-Ar >15: High-risk withdrawal, medical monitoring required
- CIWA-Ar 8-15: Moderate risk, frequent assessment needed
- CIWA-Ar <8: Low risk, supportive care appropriate
- Seizure history: Automatic high-risk category
- DT history: ICU-level monitoring consideration
- >10 drinks/day: Medical detox recommended
-
Opioid Overdose Recognition
- Respiratory rate <12: Naloxone indicated
- Oxygen saturation <90%: Immediate airway management
- Pinpoint pupils + CNS depression: Classic triad
- Naloxone 0.4-2mg IV/IM/IN initial dose
- Repeat q2-3 minutes if no response
- Duration 30-90 minutes - re-dosing often needed
-
Stimulant Intoxication Severity
- Temperature >101°F: Hyperthermia risk
- BP >180/110: Hypertensive emergency
- Heart rate >120: Cardiac monitoring required
- Benzodiazepines first-line for agitation
- Avoid beta-blockers (unopposed alpha stimulation)
- Cooling measures for hyperthermia
| Assessment Tool | Timeframe | Threshold Scores | Clinical Action | Sensitivity/Specificity |
|---|---|---|---|---|
| CIWA-Ar | Q4-8 hours | >15 high-risk | Medical detox | 90%/85% |
| COWS | Q4-6 hours | >24 moderate | Comfort meds | 85%/80% |
| AUDIT | Screening | >8 hazardous | Brief intervention | 92%/94% |
| DAST-10 | Screening | >3 problematic | Further assessment | 86%/84% |
| PHQ-9 | Baseline | >15 severe | Suicide assessment | 88%/88% |
| %%{init: {'flowchart': {'htmlLabels': true}}}%% | ||||
| flowchart TD |
Start["👤 Patient Presentation
• Initial intake• Screen symptoms"]
Intox["❓ Acute Intoxication?
• Assess sensorium• Vitals check"]
Stabilize["⚠️ Stabilize Medically
• Airway protection• IV fluids/support"]
Safety["👁️ Safety Monitoring
• Close observation• Fall precautions"]
Withdrawal["📋 Withdrawal Assess
• CIWA or COWS• History of risk"]
Risk["❓ High-Risk?
• Seizure history• Liver status"]
Detox["💊 Medical Detox
• Inpatient care• Pharm support"]
Outpatient["✅ Outpatient Mgmt
• Social support• Clinic follow up"]
Planning["📋 Tx Planning
• Long-term care• Psych referral"]
Start --> Intox Intox -->|Yes| Stabilize Intox -->|No| Withdrawal Stabilize --> Safety Safety --> Planning Withdrawal --> Risk Risk -->|Yes| Detox Risk -->|No| Outpatient Detox --> Planning Outpatient --> Planning
style Start fill:#F7F5FD, stroke:#F0EDFA, stroke-width:1.5px, rx:12, ry:12, color:#6B21A8 style Intox fill:#FEF8EC, stroke:#FBECCA, stroke-width:1.5px, rx:12, ry:12, color:#854D0E style Stabilize fill:#FDF4F3, stroke:#FCE6E4, stroke-width:1.5px, rx:12, ry:12, color:#B91C1C style Safety fill:#EEFAFF, stroke:#DAF3FF, stroke-width:1.5px, rx:12, ry:12, color:#0369A1 style Withdrawal fill:#FEF8EC, stroke:#FBECCA, stroke-width:1.5px, rx:12, ry:12, color:#854D0E style Risk fill:#FEF8EC, stroke:#FBECCA, stroke-width:1.5px, rx:12, ry:12, color:#854D0E style Detox fill:#F1FCF5, stroke:#BEF4D8, stroke-width:1.5px, rx:12, ry:12, color:#166534 style Outpatient fill:#F6F5F5, stroke:#E7E6E6, stroke-width:1.5px, rx:12, ry:12, color:#525252 style Planning fill:#F7F5FD, stroke:#F0EDFA, stroke-width:1.5px, rx:12, ry:12, color:#6B21A8
> ⭐ **Clinical Pearl**: **"Rule of 3s"** for withdrawal timing - **Alcohol**: symptoms **3-6 hours**, peak **24-72 hours**, duration **3-7 days**. **Opioids**: symptoms **6-12 hours**, peak **48-72 hours**, duration **7-14 days**. **Benzodiazepines**: symptoms **1-7 days**, peak **7-14 days**, duration **2-8 weeks**.
**Rapid Treatment Selection Matrix**:
* **Alcohol Use Disorder**
- **Naltrexone**: **50mg daily**, **craving reduction** **25-30%**
- **Acamprosate**: **666mg TID**, **abstinence maintenance**
- **Disulfiram**: **250mg daily**, **supervised administration**
+ **Naltrexone** for **craving-dominant** patients
+ **Acamprosate** for **anxiety-prone** patients
+ **Disulfiram** for **motivated** patients with **supervision**
* **Opioid Use Disorder**
- **Buprenorphine**: **8-24mg daily**, **office-based** treatment
- **Methadone**: **60-120mg daily**, **clinic-based** treatment
- **Naltrexone XR**: **380mg monthly**, **abstinence-based**
+ **Buprenorphine** for **most patients** (**first-line**)
+ **Methadone** for **high-dose** users or **buprenorphine failures**
+ **Naltrexone** for **motivated** patients **post-detox**
* **Tobacco Dependence**
- **Varenicline**: **1mg BID**, **highest efficacy** (**33%** quit rate)
- **Bupropion**: **150mg BID**, **depression comorbidity**
- **NRT**: **Variable dosing**, **combination therapy**
+ **Varenicline** first-line unless **contraindicated**
+ **Combination NRT** (patch + gum) **more effective**
+ **Bupropion** for **weight-concerned** patients
> 💡 **Master This**: **Medication-Assisted Treatment (MAT)** reduces **overdose mortality** by **50-70%** and **criminal activity** by **40-60%**. **Retention rates**: **Methadone 60-80%**, **Buprenorphine 40-60%**, **Naltrexone 20-40%**. **Higher retention** = **better outcomes** across all measures.
**Crisis Intervention Protocols** for immediate implementation:
* **Overdose Response Checklist**
- **Call 911** immediately
- **Naloxone administration** if opioid suspected
- **Recovery position** if breathing adequate
- **Rescue breathing** if respiratory depression
- **Stay until EMS arrives** - **re-sedation possible**
* **Withdrawal Complications**
- **Seizure**: **Lorazepam 2-4mg IV**, **thiamine 100mg**
- **DTs**: **ICU transfer**, **high-dose benzodiazepines**
- **Hyperthermia**: **Cooling**, **IV fluids**, **electrolyte monitoring**
* **Suicide Risk Management**
- **PHQ-9 item 9 >0**: **Immediate safety assessment**
- **Means restriction** counseling
- **Safety planning** with **specific coping strategies**
- **Follow-up within 24-48 hours**
This clinical arsenal transforms addiction medicine from **overwhelming complexity** to **systematic expertise**, enabling **confident practice** with **measurable outcomes** and **evidence-based interventions** that **save lives** and **restore function** through **neurobiologically-informed** medical care.
🎯 Addiction Mastery: Clinical Command Arsenal
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more
Have doubts about this lesson?
Ask Rezzy, your AI Study Mate, to explain anything you didn't understand
Everything you need for NEET-PG prep
Get full Oncourse access with lessons, practice questions, flashcards and AI study tools.
Scan to download app






















