Pigmentary Disorders

On this page
🎨 Pigmentary Disorders: The Skin's Color Canvas Mastery
Skin color tells a story written in melanocytes, immune cells, hormones, and environmental exposures-and when that narrative goes awry, pigmentary disorders emerge as visible maps of underlying pathology. You'll master the genetic blueprints that govern pigmentation, trace how inflammation and hormones reshape the skin's palette, and build a systematic approach to diagnosis that distinguishes vitiligo from tinea versicolor at a glance. By integrating pattern recognition with targeted therapies, you'll transform complex presentations into confident clinical decisions that restore both pigment and patient well-being.
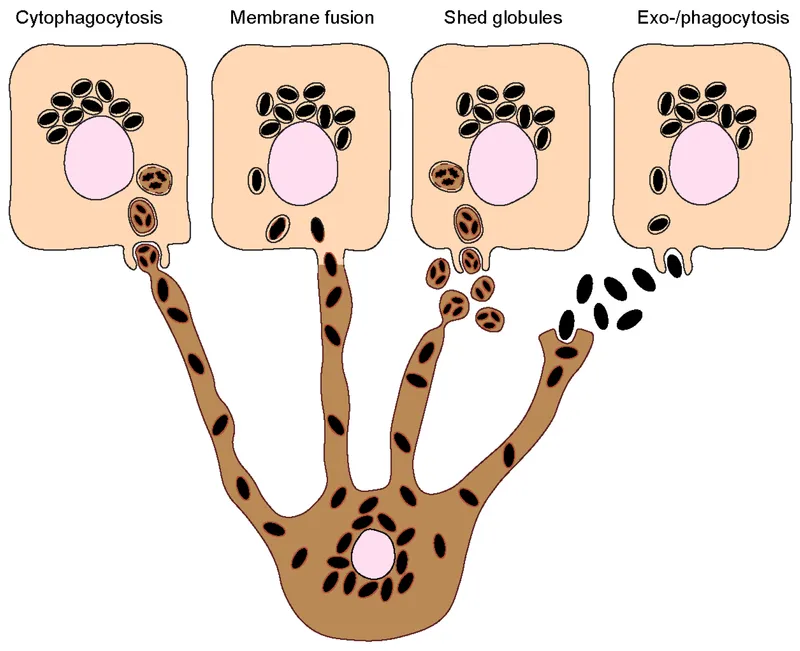
Melanocyte Architecture: The Pigment Production Network
Melanocytes function as specialized pigment factories, comprising only 5-10% of epidermal cells yet controlling skin color through dendritic networks reaching 30-40 keratinocytes each. These cells originate from neural crest migration during embryogenesis, establishing permanent residence in the basal epidermis at densities of 1,000-2,000 cells/mm².
📌 Remember: MELANOCYTE - Migration from neural crest, Epidermal basal layer, Long dendrites, Active tyrosinase, Numerous melanosomes, Octopus-like morphology, Color production, Yield to keratinocytes, Transfer pigment, Essential for protection
The melanin synthesis pathway operates through 4 critical enzymatic steps, with tyrosinase serving as the rate-limiting enzyme. Normal melanin production requires copper availability, pH optimization at 6.8, and adequate substrate supply of tyrosine and cysteine.
-
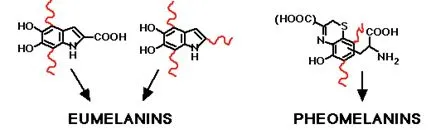
Eumelanin Production (Brown-Black Pigment)
- Tyrosine → DOPA → Dopaquinone → Eumelanin
- Requires functional tyrosinase enzyme (TYR gene)
- Produces photoprotective melanin with UV absorption
- Dominant in darker skin phototypes (IV-VI)
-
Pheomelanin Production (Red-Yellow Pigment)
- Cysteine incorporation pathway
- Less photoprotective capacity
- Predominant in fair skin (phototypes I-II)
- Associated with increased UV sensitivity
| Melanin Type | Color | UV Protection | Phototype | Genetic Control | Clinical Significance |
|---|---|---|---|---|---|
| Eumelanin | Brown-Black | High (90% UV absorption) | IV-VI | MC1R, TYR, TYRP1 | Photoprotection, cancer prevention |
| Pheomelanin | Red-Yellow | Low (30% UV absorption) | I-II | MC1R variants | UV sensitivity, melanoma risk |
| Mixed | Variable | Moderate (60% UV absorption) | III | Balanced expression | Intermediate protection |
💡 Master This: Every pigmentary disorder reflects disruption in melanocyte number, function, or melanin transfer - understanding this triad predicts clinical presentations and guides therapeutic approaches.
The melanosome maturation process progresses through 4 distinct stages, from initial protein matrix formation to fully melanized organelles ready for keratinocyte transfer. Stage IV melanosomes contain 60-80% melanin by weight and measure 0.5-0.7 micrometers in diameter.
Connect melanocyte biology through cellular dysfunction patterns to understand how genetic mutations, inflammatory processes, and environmental factors create the diverse spectrum of pigmentary disorders encountered in clinical practice.
🎨 Pigmentary Disorders: The Skin's Color Canvas Mastery
🧬 Genetic Architecture: The Pigmentation Code Decoded
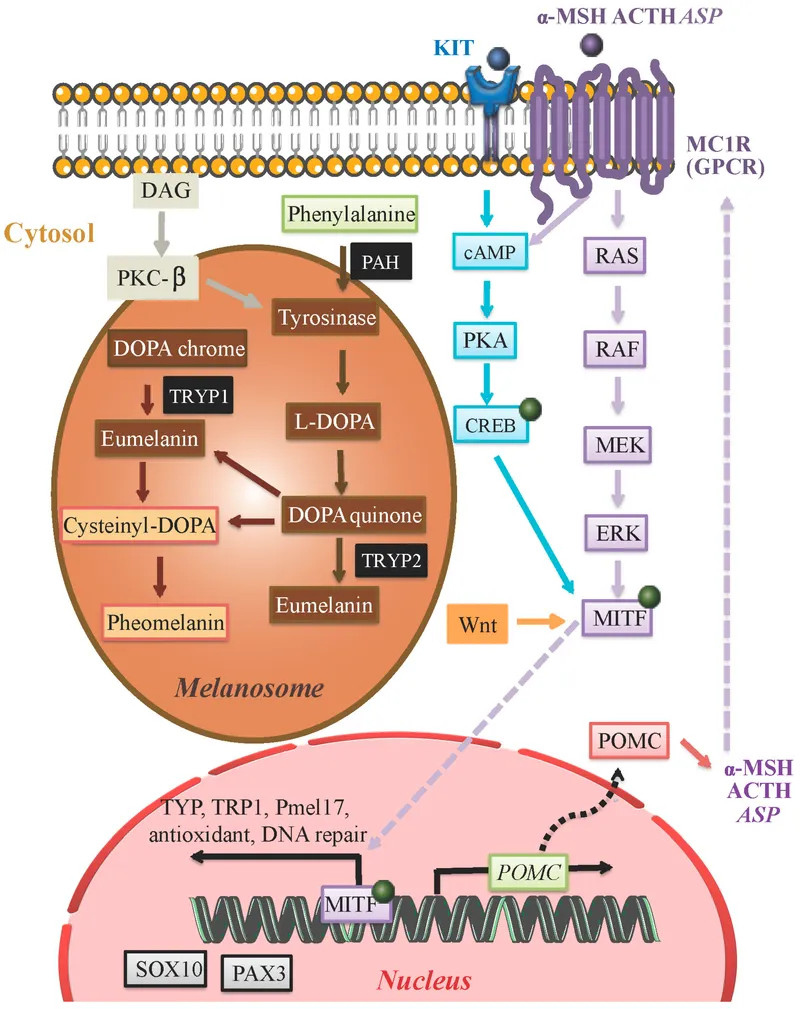
Master Gene Controllers: The Pigmentation Regulatory Network
The MC1R gene functions as the master switch for melanin type determination, with >150 documented variants creating the spectrum from red hair to black hair phenotypes. Functional MC1R variants increase cAMP levels 10-fold, driving eumelanin production through MITF transcription factor activation.
📌 Remember: PIGMENT GENES - PMC1R (receptor), ITYR (tyrosinase), GTYRP1 (related protein), MITF (transcription), EOCA genes (albinism), NASPM (microcephaly), TSLC45A2 (transporter)
- Primary Pigmentation Genes
- MC1R: Melanocortin-1 receptor (16q24.3)
- >300 variants documented globally
- Loss-of-function → pheomelanin predominance
- Gain-of-function → enhanced eumelanin production
- TYR: Tyrosinase enzyme (11q14.3)
- >400 mutations causing OCA1
- Complete loss → OCA1A (no pigment)
- Partial function → OCA1B (minimal pigment)
- OCA2: P protein (15q12-q13)
- Most common albinism worldwide (70% of cases)
- Melanosome pH regulation
- African populations: 1:4,000 carrier frequency
- MC1R: Melanocortin-1 receptor (16q24.3)
| Gene | Chromosome | Function | Mutation Frequency | Associated Disorders | Population Prevalence |
|---|---|---|---|---|---|
| MC1R | 16q24.3 | Melanin type switch | 1:4 Europeans | Red hair, melanoma risk | 4% red hair variants |
| TYR | 11q14.3 | Rate-limiting enzyme | 1:17,000 | OCA1A, OCA1B | 1:40,000 births |
| OCA2 | 15q12-q13 | Melanosome pH | 1:4,000 Africans | OCA2, brown OCA | 1:15,000 globally |
| TYRP1 | 9p23 | Eumelanin stability | 1:8,500 Africans | OCA3, brown albinism | Rare outside Africa |
| SLC45A2 | 5p13.2 | Tyrosine transport | 1:50,000 | OCA4, skin lightening | Asian populations |
Epigenetic Modulation: Environmental Gene Control
Environmental factors modify gene expression through DNA methylation and histone modifications, explaining why identical twins can develop different pigmentation patterns. UV exposure increases MITF expression 5-fold within 24 hours, while hormonal changes during pregnancy activate MSH production leading to melasma development.
💡 Master This: Genetic variants create susceptibility patterns, but environmental triggers determine clinical expression - this principle guides both prevention strategies and treatment timing for optimal outcomes.
The ASIP gene (agouti signaling protein) acts as a natural MC1R antagonist, with gain-of-function mutations causing generalized hyperpigmentation and loss-of-function contributing to localized darkening patterns seen in café-au-lait macules.
Connect genetic architecture through inflammatory cascade activation to understand how immune system dysfunction creates acquired pigmentary disorders like vitiligo and post-inflammatory changes.
🔥 Inflammatory Cascade: The Immune-Pigment Battleground

Autoimmune Destruction Patterns: The Vitiligo Paradigm
Vitiligo represents CD8+ T-cell mediated melanocyte destruction through IFN-γ and TNF-α cytokine release, with CXCL10 chemokine levels correlating directly with disease activity and progression rate. Active vitiligo demonstrates >10-fold elevated inflammatory markers compared to stable disease.
📌 Remember: VITILIGO CASCADE - Viral mimicry triggers, IFN-γ activation, T-cell infiltration, IL-15 amplification, Loss of melanocytes, Immune memory formation, Genetic susceptibility, Oxidative stress
-
Phase 1: Initiation (Weeks 1-4)
- Melanocyte stress signals (HSP70 release)
- Dendritic cell activation and antigen presentation
- CD4+ T-helper cell priming and differentiation
- Th1/Th17 cytokine profile establishment
-
Phase 2: Amplification (Months 1-6)
- CD8+ cytotoxic T-cell recruitment and activation
- IFN-γ production increases >50-fold
- CXCL9/10/11 chemokine gradient formation
- Progressive melanocyte apoptosis (>80% loss)
-
Phase 3: Maintenance (Years)
- Resident memory T-cell establishment
- Tissue-resident immune surveillance
- Recurrence potential remains >70%
- Repigmentation resistance in established lesions
⭐ Clinical Pearl: CXCL10 serum levels >150 pg/mL predict active vitiligo progression with 85% sensitivity and 92% specificity, enabling early intervention timing.
Post-Inflammatory Responses: The Pigmentation Pendulum
Post-inflammatory hyperpigmentation (PIH) develops through melanocyte hyperactivation rather than destruction, with inflammatory mediators increasing tyrosinase activity 3-5 fold and melanosome transfer efficiency by 200%. Darker skin types (IV-VI) demonstrate >90% PIH development following inflammation.
| Inflammatory Trigger | Mechanism | Onset Time | Duration | Skin Type Risk | Treatment Response |
|---|---|---|---|---|---|
| Acne | Sebaceous inflammation | 2-4 weeks | 6-24 months | Types IV-VI: 95% | Moderate (60% improvement) |
| Eczema | Th2-mediated inflammation | 1-2 weeks | 3-12 months | All types: 70% | Good (80% improvement) |
| Trauma | Direct tissue damage | Immediate | 2-18 months | Types III-VI: 85% | Variable (40-90%) |
| Infection | Pathogen-induced | 1-3 weeks | 4-36 months | Types IV-VI: 90% | Poor (30% improvement) |
| Chemical | Irritant contact | Hours-days | 1-6 months | All types: 60% | Excellent (90% improvement) |
The prostaglandin E2 pathway plays a crucial role in inflammation-induced pigmentation, with COX-2 inhibitors reducing PIH development by 60% when initiated within 72 hours of inflammatory onset.
Connect inflammatory cascade patterns through hormonal modulation systems to understand how endocrine factors create pregnancy-related pigmentation changes and melasma development.
🔥 Inflammatory Cascade: The Immune-Pigment Battleground
🌙 Hormonal Orchestration: The Endocrine Pigment Symphony
Pregnancy Pigmentation: The Gestational Color Shift
Pregnancy triggers dramatic hormonal changes with estrogen levels increasing 100-fold and progesterone rising 10-fold by the third trimester, directly stimulating melanocyte activity and creating the characteristic linea nigra in >90% of pregnancies and melasma in 50-70% of women.
📌 Remember: PREGNANCY PIGMENT - Progesterone surge, Rising estrogen, Enhanced MSH, Gestational changes, Ninea nigra, Areolar darkening, Nipple pigmentation, Chloasma development, Years to fade
- Hormonal Cascade During Pregnancy
-
First Trimester (Weeks 1-12)
- β-hCG stimulates MSH receptors directly
- Estradiol increases >20-fold from baseline
- Progesterone enhances tyrosinase sensitivity
- Early areolar darkening in >80% of women
-
Second Trimester (Weeks 13-26)
- Peak MSH activity with 5-fold elevation
- Melasma development in 50-70% of pregnancies
- Linea nigra appearance in >90% of cases
- Existing nevi darken in >60% of women
-
Third Trimester (Weeks 27-40)
- Maximum pigmentation intensity
- Estrogen peaks at 100x non-pregnant levels
- Progesterone reaches 10x baseline values
- Postpartum fading begins 6-12 months after delivery
-
| Pregnancy Stage | Estrogen Level | Progesterone Level | MSH Activity | Pigmentation Risk | Clinical Manifestations |
|---|---|---|---|---|---|
| Pre-pregnancy | Baseline (100 pg/mL) | Baseline (1 ng/mL) | Normal | Low (10%) | Minimal changes |
| First Trimester | 5-20x (500-2000 pg/mL) | 3-5x (3-5 ng/mL) | 2x elevation | Moderate (30%) | Areolar darkening |
| Second Trimester | 20-50x (2000-5000 pg/mL) | 5-8x (5-8 ng/mL) | 5x elevation | High (70%) | Melasma onset |
| Third Trimester | 50-100x (5000-10000 pg/mL) | 8-10x (8-10 ng/mL) | Peak activity | Maximum (90%) | Full pigmentation |
| Postpartum | Return to baseline | Return to baseline | Gradual decline | Fading (60%) | Slow resolution |
Thyroid-Pigmentation Interactions: The Metabolic Connection
Thyroid dysfunction significantly impacts pigmentation through metabolic rate changes affecting melanocyte turnover and hormone receptor sensitivity. Hyperthyroidism increases melanin production by 40%, while hypothyroidism reduces tyrosinase activity by 30%.
💡 Master This: Hormonal pigmentation changes follow predictable patterns - understanding the timing and magnitude of hormonal fluctuations enables accurate prognosis and optimal treatment planning.
Oral contraceptive use creates melasma in 25-30% of users through synthetic estrogen effects, with higher-dose formulations (>35 mcg ethinyl estradiol) showing 3-fold increased risk compared to low-dose preparations (<20 mcg).
Connect hormonal orchestration through environmental trigger recognition to understand how UV exposure, chemical contacts, and physical trauma create the clinical patterns seen in pigmentary disorders.
🌙 Hormonal Orchestration: The Endocrine Pigment Symphony
☀️ Environmental Triggers: The External Pigment Modulators
Ultraviolet Radiation: The Primary Pigment Driver
UV radiation serves as the most potent environmental trigger for pigmentation changes, with UVB (280-320 nm) causing direct DNA damage and UVA (320-400 nm) generating reactive oxygen species that activate melanogenesis within 24-48 hours. Minimal erythema dose (MED) varies >100-fold between skin phototypes.
📌 Remember: UV EFFECTS - UVB direct damage, Vitamin D synthesis, Erythema response, Free radical formation, Fibroblast activation, Elastosis development, Carcinogenesis risk, Tanning response, Sensitivity variation
- Phototype-Specific UV Responses
-
Type I (Always burns, never tans)
- MED: 15-30 mJ/cm²
- Melanoma risk: >10-fold increased
- Tanning capacity: Minimal (<10% increase)
- PIH risk: Low (20% following inflammation)
-
Type IV (Minimal burning, always tans)
- MED: 45-60 mJ/cm²
- Melanoma risk: Baseline population risk
- Tanning capacity: Excellent (>200% increase)
- PIH risk: High (85% following inflammation)
-
Type VI (Never burns, deeply pigmented)
- MED: >60 mJ/cm²
- Melanoma risk: Reduced (0.1x population)
- Tanning capacity: Constitutive pigmentation
- PIH risk: Maximum (>95% following inflammation)
-
| UV Type | Wavelength | Penetration | Primary Effect | Onset Time | Clinical Significance |
|---|---|---|---|---|---|
| UVC | 200-280 nm | Blocked by ozone | Germicidal | N/A | Artificial sources only |
| UVB | 280-320 nm | Epidermis | DNA damage, erythema | 2-6 hours | Sunburn, vitamin D |
| UVA2 | 320-340 nm | Upper dermis | Immediate tanning | Minutes | Photoaging acceleration |
| UVA1 | 340-400 nm | Deep dermis | Delayed tanning | 24-72 hours | Melasma, drug photosensitivity |
| Visible Light | 400-700 nm | Full dermis | Melasma trigger | Hours-days | Blue light, melasma persistence |
Chemical and Physical Triggers: The Contact Modulators
Chemical exposures create pigmentary changes through direct melanocyte toxicity or inflammatory cascade activation, with hydroquinone causing exogenous ochronosis in >35% of users after >6 months of continuous application at >4% concentrations.
- High-Risk Chemical Exposures
-
Hydroquinone (>4% concentration)
- Ochronosis risk: 35% after 6 months
- Reversibility: <20% complete resolution
- Prevention: Limit to 3-month cycles
-
Phenolic compounds (industrial exposure)
- Occupational vitiligo: 15% of exposed workers
- Latency period: 6 months to 5 years
- Pattern: Contact site and distant spread
-
Heavy metals (mercury, silver)
- Argyria risk: Permanent blue-gray discoloration
- Dose threshold: >4 grams cumulative exposure
- Distribution: Sun-exposed areas predominantly
-
💡 Master This: Environmental trigger identification enables targeted prevention strategies - removing or modifying exposures prevents >80% of progression in environmentally-induced pigmentary disorders.
Physical trauma through friction, pressure, or thermal injury activates inflammatory cascades leading to PIH development in >90% of darker skin types, with healing time directly correlating with pigmentation intensity and duration.
Connect environmental trigger patterns through diagnostic framework development to understand how clinical presentation patterns enable accurate diagnosis and appropriate treatment selection.
☀️ Environmental Triggers: The External Pigment Modulators
🔍 Diagnostic Mastery: The Pattern Recognition Arsenal
Clinical Pattern Recognition: The Morphology Matrix
Lesion morphology provides the primary diagnostic clue, with well-demarcated borders suggesting autoimmune destruction (vitiligo), while ill-defined borders indicate inflammatory processes (PIH) or genetic mosaicism (nevus depigmentosus). Symmetrical distribution occurs in >80% of hormonal disorders (melasma) versus asymmetrical patterns in >90% of autoimmune conditions.
📌 Remember: PATTERN ANALYSIS - Pigment change type, Age of onset, Topographic distribution, Temporal evolution, Edge characteristics, Response to treatment, Nassociated symptoms
- Hypopigmentation Pattern Recognition
-
Vitiligo: Chalk-white, well-demarcated, symmetrical
- Wood's lamp: Bright white fluorescence
- Distribution: Acrofacial pattern in >70%
- Progression: Centrifugal spread over months-years
- Associated: Thyroid disease in 25%, diabetes in 15%
-
Nevus Depigmentosus: Off-white, stable since birth
- Wood's lamp: Dull white appearance
- Distribution: Unilateral in >90% of cases
- Progression: Proportional growth only
- Associated: No systemic associations
-
Pityriasis Alba: Hypopigmented, scaly, facial
- Wood's lamp: Minimal enhancement
- Distribution: Facial in >95% of children
- Progression: Self-limiting over 1-3 years
- Associated: Atopic dermatitis in >60%
-
| Condition | Color | Border | Distribution | Wood's Lamp | Onset Age | Progression |
|---|---|---|---|---|---|---|
| Vitiligo | Chalk-white | Sharp, convex | Symmetrical | Bright enhancement | Any age | Progressive |
| Nevus Depigmentosus | Off-white | Irregular | Unilateral | Dull enhancement | Birth | Stable |
| Pityriasis Alba | Light tan | Ill-defined | Facial | Minimal enhancement | Childhood | Self-limiting |
| PIH | Brown-black | Variable | Trauma sites | No enhancement | Post-inflammation | Slowly fading |
| Melasma | Brown | Geographic | Centrofacial | Variable enhancement | Reproductive age | Hormone-dependent |
Advanced Diagnostic Tools: The Technology Enhancement
Dermoscopy reveals microscopic features invisible to naked eye examination, with perifollicular pigmentation in vitiligo indicating >80% repigmentation potential, while absence of follicular pigment suggests <20% treatment response probability.
- Dermoscopic Diagnostic Features
-
Vitiligo Active Edge
- Trichrome pattern: Tan intermediate zone
- Inflammatory border: Pink-red coloration
- Confetti-like depigmentation spots
- Leukotrichia: White hair within lesions
-
Melasma Patterns
- Epidermal type: Brown color, sharp borders
- Dermal type: Blue-gray, diffuse borders
- Mixed type: Combination patterns (>60% of cases)
- Vascular component: Telangiectasias in >40%
-
💡 Master This: Combining clinical morphology with advanced diagnostic tools achieves >95% diagnostic accuracy in pigmentary disorders, enabling precise treatment selection and accurate prognostic counseling.
Reflectance confocal microscopy provides real-time histological imaging, identifying melanocyte density and inflammatory infiltrates without biopsy requirement, particularly valuable for monitoring treatment response in research settings.
Connect diagnostic mastery through therapeutic intervention strategies to understand how accurate diagnosis guides treatment selection and monitoring protocols for optimal clinical outcomes.
🔍 Diagnostic Mastery: The Pattern Recognition Arsenal
🎯 Therapeutic Arsenal: The Precision Treatment Protocols
Evidence-Based Treatment Hierarchies: The Systematic Approach
First-line therapies achieve >60% improvement in >80% of patients when selected appropriately, with combination approaches demonstrating superior efficacy compared to monotherapy in >75% of comparative studies. Treatment response correlates directly with accurate diagnosis, appropriate patient selection, and adherence protocols.
📌 Remember: TREATMENT SUCCESS - Timing optimization, Response monitoring, Evidence-based selection, Adherence support, Toxicity awareness, Maintenance planning, Expectation management, New therapy integration, Team coordination
- Vitiligo Treatment Hierarchy
-
First-Line (Localized Disease <10% BSA)
- Topical corticosteroids: Class III-IV for body, Class VI-VII for face
- Response rate: 45-60% repigmentation in 6 months
- Calcineurin inhibitors: Tacrolimus 0.1%, pimecrolimus 1%
- Facial efficacy: >70% improvement in >80% of patients
-
Second-Line (Extensive Disease >10% BSA)
- Narrowband UVB: 3x weekly for 6-12 months
- Response rate: >75% achieve >25% repigmentation
- JAK inhibitors: Topical ruxolitinib (FDA approved 2022)
- Efficacy: >50% facial improvement in >60% of patients
-
Third-Line (Refractory Disease)
- Surgical interventions: Punch grafting, melanocyte transplant
- Candidate criteria: Stable disease >12 months
- Success rate: >80% in appropriate candidates
- Depigmentation: Monobenzone for >50% involvement
-
| Treatment | Mechanism | Response Rate | Time to Response | Maintenance Required | Side Effects |
|---|---|---|---|---|---|
| Topical Steroids | Anti-inflammatory | 45-60% | 3-6 months | No | Atrophy, telangiectasias |
| Calcineurin Inhibitors | T-cell suppression | 60-75% | 2-4 months | Yes | Burning, infection risk |
| Narrowband UVB | Melanocyte stimulation | 75-85% | 6-12 months | Yes | Erythema, photoaging |
| JAK Inhibitors | Cytokine blockade | 50-70% | 6-24 weeks | Yes | Local irritation |
| Surgical Options | Cell transplantation | 80-95% | 3-6 months | No | Scarring, infection |
Melasma Management Protocols: The Hormonal Challenge
Melasma treatment requires multi-modal approaches due to complex pathophysiology involving hormonal, UV, and genetic factors. Triple combination therapy (hydroquinone + tretinoin + corticosteroid) achieves >70% improvement in >80% of patients within 8-12 weeks.
- Melasma Treatment Algorithm
-
Step 1: Strict photoprotection + topical depigmenting agents
- Hydroquinone 4%: Gold standard with >70% efficacy
- Tretinoin 0.05%: Enhances penetration and cell turnover
- Mild corticosteroid: Reduces inflammation during initial phase
- Duration: 8-12 weeks initial treatment cycle
-
Step 2: Chemical peels for refractory cases
- Glycolic acid 20-70%: Monthly treatments for 3-6 sessions
- TCA 10-25%: Deeper penetration for dermal component
- Jessner's solution: Combination approach for mixed-type melasma
- Response rate: >60% additional improvement
-
Step 3: Laser therapy for resistant cases
- Q-switched lasers: 1064 nm Nd:YAG for dermal pigment
- Fractional lasers: 1550 nm erbium for epidermal remodeling
- Success rate: Variable (30-80% depending on type)
- Risk: PIH development in >40% of darker skin types
-
💡 Master This: Treatment success in pigmentary disorders depends on accurate phenotyping, appropriate therapy selection, realistic timeline expectations, and comprehensive maintenance protocols - rushing treatment or skipping maintenance leads to >60% recurrence rates.
Maintenance therapy prevents recurrence in >80% of successfully treated patients, typically involving reduced-strength topical agents, continued photoprotection, and periodic professional treatments every 3-6 months.
The therapeutic arsenal for pigmentary disorders continues expanding with novel targets including JAK inhibitors, prostaglandin analogs, and stem cell therapies, offering hope for previously refractory conditions while requiring careful risk-benefit analysis and long-term safety monitoring.
🎯 Therapeutic Arsenal: The Precision Treatment Protocols
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more
Have doubts about this lesson?
Ask Rezzy, your AI Study Mate, to explain anything you didn't understand
Everything you need for NEET-PG prep
Get full Oncourse access with lessons, practice questions, flashcards and AI study tools.
Scan to download app























