HRP Intro & Screening - Spotting Trouble Early
- High-Risk Pregnancy (HRP): Condition where mother, fetus, or neonate faces ↑ risk of morbidity/mortality pre, intra, or postpartum.
- **Key Risk Factors (Indian Context):
- Age: <18 yrs or >35 yrs
- Parity: Nullipara, Grand multipara (≥5 births)
- Bad Obstetric Hx: Recurrent loss, stillbirth, preterm, IUGR, previous LSCS, PIH
- Medical Illness: Anemia (Hb <10 g/dl), HTN, DM, Heart/Kidney disease, HIV, TB
- Socio-demographic: Low SES, malnutrition, <4 ANC visits
- Screening & Early Detection:
- Thorough history & physical exam at each ANC visit.
- Essential Labs: Hb, Blood group & Rh, Urine (albumin, sugar), VDRL, HIV, HBsAg, RBS.
- GDM screen: DIPSI (75g OGTT, plasma glucose ≥140 mg/dl at 2 hrs).

⭐ Booking visit (1st ANC) is crucial for risk assessment; ideally within 1st trimester (by 12 weeks).
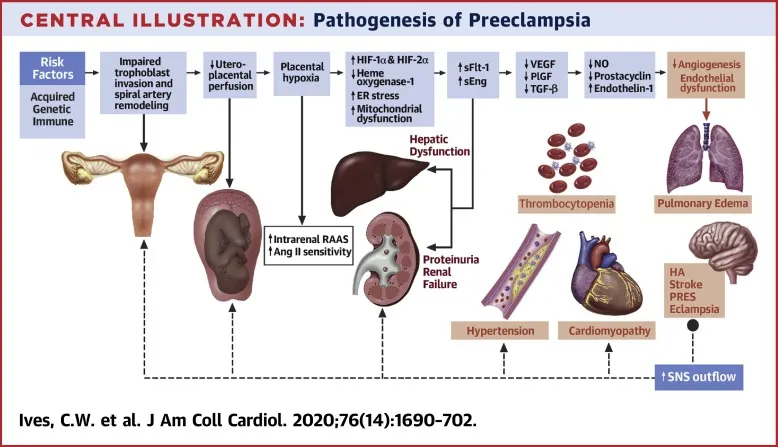
HRP: Hypertensive Disorders - Pressure Cooker Situations
- Classification:
- Chronic HTN: BP ≥140/90 mmHg <20 wks.
- Gestational HTN (GHTN): BP ≥140/90 mmHg >20 wks, no proteinuria.
- Preeclampsia (PE): GHTN + Proteinuria (≥300mg/24h / PCR ≥0.3).
- Severe Features: BP ≥160/110 mmHg OR end-organ dysfunction (platelets <100k/µL, LFTs ↑↑, Creat >1.1mg/dL, pulm edema, CNS sx).
- Eclampsia: PE + Seizures.
- HELLP: Hemolysis, Elevated Liver enzymes, Low Platelets.
- Management:
- Antihypertensives: Hydralazine, Methyldopa, Labetalol, Nifedipine (📌 HMLN). Avoid ACEi/ARBs.
- MgSO₄: Seizure prophylaxis/Rx (Sev PE/Eclampsia). Load 4-6g IV, Maint 1-2g/hr. Antidote: Ca Gluconate 1g.
- Delivery: Definitive Rx.
⭐ MgSO₄ is DOC for seizure prophylaxis in severe PE & seizure control in eclampsia.
HRP: GDM & Anemia - Sweet & Low Battles
Gestational Diabetes Mellitus (GDM):
- Screening (India): DIPSI - 75g OGTT (anytime, non-fasting).
- Diagnosis: 2-hr plasma glucose ≥ 140 mg/dL.
- Targets: FBS < 95; 1-hr PP < 140; 2-hr PP < 120 mg/dL.
- Management: MNT first; then Insulin. Metformin is an option.
Anemia in Pregnancy (India):
- Cut-off: Hb < 11 g/dL. Severe: Hb < 7 g/dL.
- Prophylaxis (National): 100 mg elemental Fe + 500 µg Folic Acid daily.
- Treatment: 100-200 mg elemental Fe daily. Parenteral Fe for severe/intolerant cases.
⭐ In India, GDM screening uses DIPSI: 75g OGTT (non-fasting), with a single 2-hr plasma glucose ≥ 140 mg/dL being diagnostic.
HRP: Obstetric Alarms - Early Birds & Small Wonders
- Preterm Labor (PTL): Labor < 37 weeks.
- Tocolysis (e.g., Nifedipine): If < 34 weeks, to delay for Antenatal Corticosteroids (ACS).
- ACS (Betamethasone 12mg IM x 2 doses, 24h apart): 24-34 weeks (up to 36+6 if high risk) for fetal lung maturity. 📌 BAM for Lungs.
- $MgSO_4$: Neuroprotection if < 32 weeks.
- Intrauterine Growth Restriction (IUGR)/SGA: Estimated Fetal Weight (EFW) < 10th percentile.
- Monitor: Serial USG, Doppler (Umbilical Artery - UA, Middle Cerebral Artery - MCA).
- Delivery: Timed by Doppler severity (e.g., UA absent/reversed end-diastolic flow).

⭐ $MgSO_4$ for fetal neuroprotection in preterm birth < 32 weeks significantly reduces cerebral palsy risk.
High‑Yield Points - ⚡ Biggest Takeaways
- Antenatal corticosteroids (e.g., Betamethasone) for fetal lung maturity (24-34 weeks) in threatened preterm labor.
- Magnesium sulfate (MgSO4) for fetal neuroprotection (<32 weeks) and eclampsia seizure control/prophylaxis.
- DIPSI criteria (single 75g OGTT) for GDM screening is widely used in India.
- Safe pregnancy antihypertensives: Labetalol, Nifedipine, Methyldopa.
- AMTSL with Oxytocin prevents PPH; manage active PPH with multiple uterotonics.
- Anti-D immunoglobulin at 28 weeks & post-delivery for Rh-negative mothers with Rh-positive infants.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

