Immunization and Vaccine-Preventable Diseases

On this page
🛡️ Immunization and Vaccine-Preventable Diseases: The Biological Defense Arsenal
Vaccines represent one of medicine's most elegant interventions-training your immune system to recognize threats before they strike, preventing diseases that once devastated populations. You'll master how different vaccine types work at the molecular level, why timing schedules maximize protection across the lifespan, and how individual vaccination decisions create population-wide immunity networks. This lesson equips you with the biological principles, epidemiological patterns, and clinical algorithms to confidently recommend immunizations and understand their profound impact on public health.
The Immunological Foundation: Memory and Protection
Immunization harnesses your body's adaptive immune system to create lasting protection against specific pathogens. When vaccines introduce antigens-whether live attenuated organisms, inactivated pathogens, or purified components-they trigger a controlled immune response that generates immunological memory without causing disease.
📌 Remember: LAMP for vaccine-induced immunity - Lasting memory cells, Antibody production, Memory B cells, Protective T cells
The immune response follows a predictable timeline: primary response peaks at 10-14 days post-vaccination, while secondary response (booster effect) reaches peak levels within 3-5 days. This accelerated response provides the foundation for vaccine efficacy, with memory B cells persisting for decades and some T cell populations maintaining protection for lifetime.
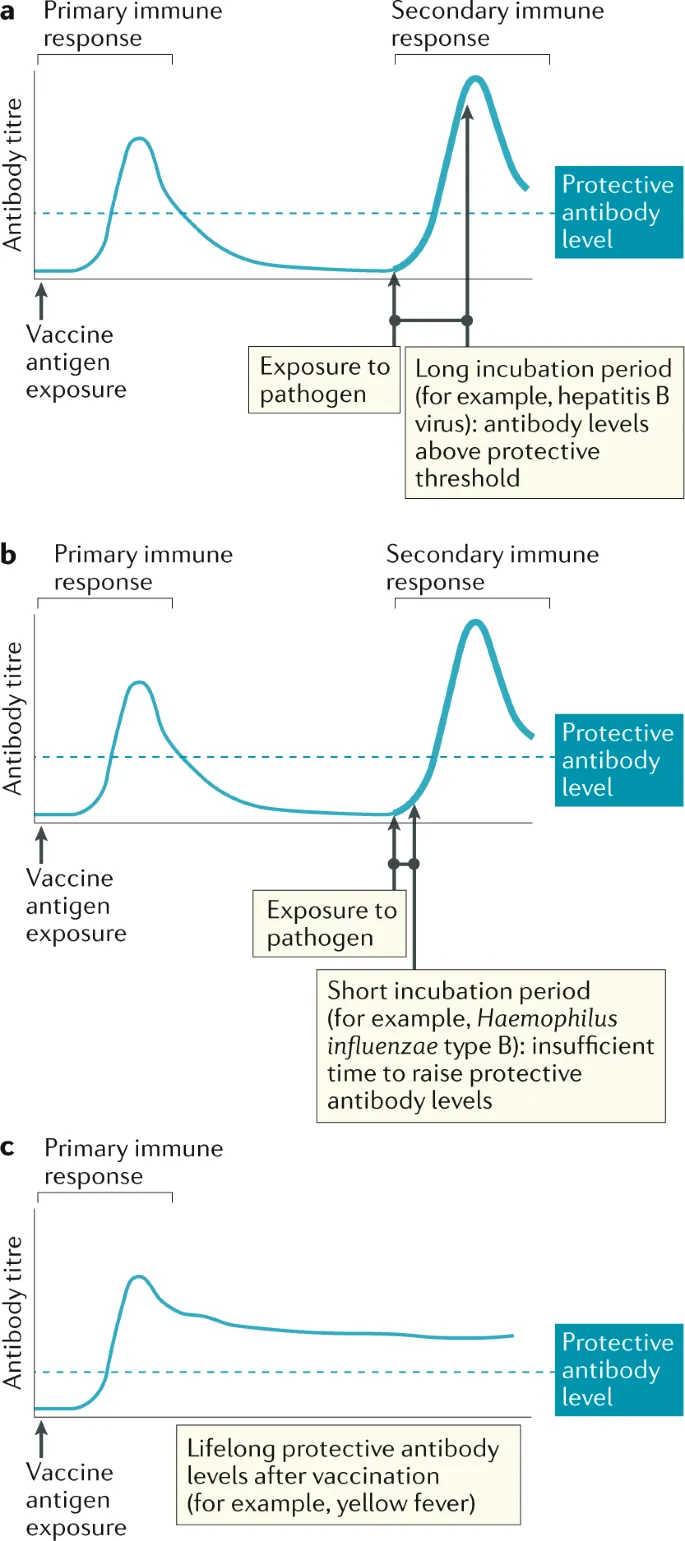
-
Primary Vaccination Response
- Initial antigen recognition: 24-48 hours
- B cell activation and proliferation: 3-7 days
- Peak antibody production: 10-14 days
- Memory cell formation: 14-21 days
- Memory B cells: 10^4-10^5 cells per antigen
- Memory T cells: 10^3-10^4 cells per antigen
-
Secondary Response (Booster)
- Rapid memory cell activation: 6-12 hours
- Peak antibody levels: 3-5 days
- Antibody concentration: 10-100x higher than primary
- Duration of protection: years to decades
⭐ Clinical Pearl: Vaccine efficacy correlates directly with antibody persistence-measles vaccine maintains >95% efficacy for 20+ years, while influenza vaccine requires annual updates due to 6-month antibody decline and viral antigenic drift.
| Vaccine Type | Peak Response Time | Memory Duration | Booster Requirement | Efficacy Rate |
|---|---|---|---|---|
| Live Attenuated | 10-14 days | Lifelong | Minimal | 90-95% |
| Inactivated | 14-21 days | 3-10 years | Regular | 70-90% |
| Subunit/Conjugate | 21-28 days | 5-15 years | Periodic | 80-95% |
| mRNA | 7-14 days | 6-12 months | Frequent | 85-95% |
| Toxoid | 14-21 days | 10+ years | Decennial | 95-99% |
The concept of herd immunity emerges when vaccination coverage reaches critical thresholds, typically 80-95% depending on disease transmissibility. This population-level protection shields unvaccinated individuals through reduced pathogen circulation, creating community-wide disease elimination.
Understanding these immunological principles predicts vaccine scheduling, explains booster requirements, and guides public health vaccination strategies that have eliminated smallpox globally and reduced polio cases by >99.9% since 1988.
🛡️ Immunization and Vaccine-Preventable Diseases: The Biological Defense Arsenal
🎯 Vaccine Classification Mastery: The Strategic Arsenal
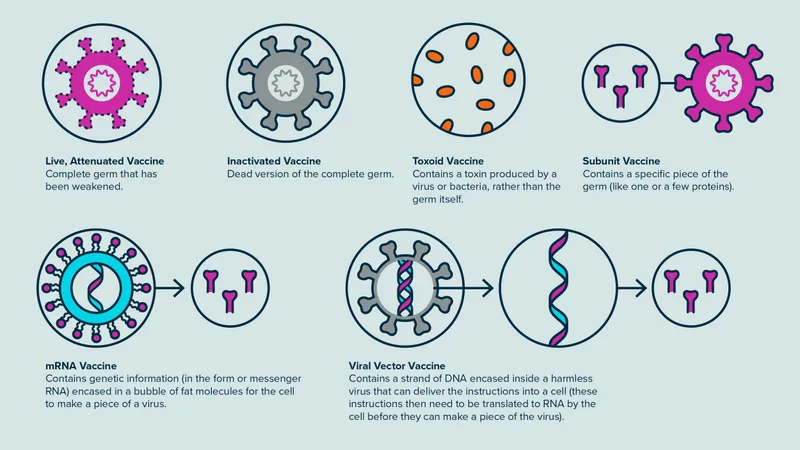
Live Attenuated Vaccines: Controlled Infection Strategy
Live attenuated vaccines contain weakened but viable pathogens that replicate in the host without causing disease. These vaccines undergo serial passage through 20-200 generations in laboratory conditions, accumulating mutations that reduce virulence while maintaining immunogenicity.
📌 Remember: MMRV-VZV for major live vaccines - Measles, Mumps, Rubella, Varicella, Varicella-Zoster, Viral (plus BCG, OPV, rotavirus)
-
Live Attenuated Characteristics
- Replication capacity: limited but sufficient for immune stimulation
- Immune response: cellular + humoral immunity
- Duration of protection: lifelong in most cases
- Administration: single dose often sufficient
- MMR: 2 doses for 99% efficacy
- Varicella: 2 doses for 98% protection
- BCG: single dose with variable efficacy (0-80%)
-
Storage and Handling Requirements
- Temperature sensitivity: extreme cold chain dependence
- Storage temperature: -15°C to +8°C (varies by vaccine)
- Reconstitution time limit: 6 hours maximum
- Light sensitivity: immediate protection required
⭐ Clinical Pearl: Live vaccines provide superior cellular immunity compared to inactivated vaccines, explaining why single-dose measles vaccination achieves 95% efficacy while inactivated polio requires multiple doses for comparable protection.
Inactivated and Subunit Vaccines: Precision Targeting
Inactivated vaccines contain killed pathogens or purified components that cannot replicate but retain immunogenic properties. These vaccines require adjuvants to enhance immune response and typically need multiple doses to achieve protective immunity.
- Inactivated Vaccine Categories
- Whole inactivated: complete organisms (IPV, hepatitis A)
- Split vaccines: disrupted organisms (influenza split)
- Subunit vaccines: purified antigens (hepatitis B, HPV)
- Toxoids: inactivated toxins (tetanus, diphtheria)
- Toxoid preparation: formaldehyde treatment for 2-4 weeks
- Adjuvant requirement: aluminum salts for enhanced response
💡 Master This: Inactivated vaccines sacrifice replication capacity for safety profile, requiring adjuvants and multiple doses to compensate for reduced immunogenicity, but offering predictable responses in immunocompromised patients.
| Vaccine Category | Replication | Doses Required | Immunity Type | Duration | Contraindications |
|---|---|---|---|---|---|
| Live Attenuated | Yes | 1-2 | Cellular + Humoral | Lifelong | Immunocompromised |
| Whole Inactivated | No | 2-3 | Primarily Humoral | 5-10 years | Minimal |
| Subunit | No | 2-4 | Targeted Humoral | 3-15 years | Minimal |
| Conjugate | No | 2-4 | Enhanced Humoral | 10+ years | Minimal |
| mRNA | No | 2-3 | Cellular + Humoral | 6-12 months | Severe allergy |
Modern vaccine development incorporates recombinant DNA technology, viral vectors, and mRNA platforms to create highly specific immune responses. These technologies enable rapid vaccine development and precise antigen presentation.
-
Conjugate Vaccines: Enhanced Immunogenicity
- Polysaccharide antigens linked to protein carriers
- Converts T-independent to T-dependent responses
- Examples: Hib, pneumococcal, meningococcal vaccines
- Carrier proteins: CRM197, tetanus toxoid, diphtheria toxoid
- Efficacy improvement: 70-95% vs. <50% for plain polysaccharides
-
Recombinant Vaccines: Precision Engineering
- Hepatitis B: HBsAg produced in yeast cells
- HPV: L1 capsid proteins creating virus-like particles
- Production time: 6-12 months vs. egg-based 6+ months
⭐ Clinical Pearl: Conjugate vaccines revolutionized pediatric immunization by converting poorly immunogenic polysaccharide antigens into highly effective vaccines, reducing Hib disease by >99% and pneumococcal disease by >80% in children.
Understanding vaccine classification guides clinical decision-making for special populations, predicts adverse event profiles, and explains the scientific rationale behind immunization schedules that protect individuals and communities from vaccine-preventable diseases.
🎯 Vaccine Classification Mastery: The Strategic Arsenal
🗓️ Immunization Scheduling Architecture: Timing Precision for Maximum Protection
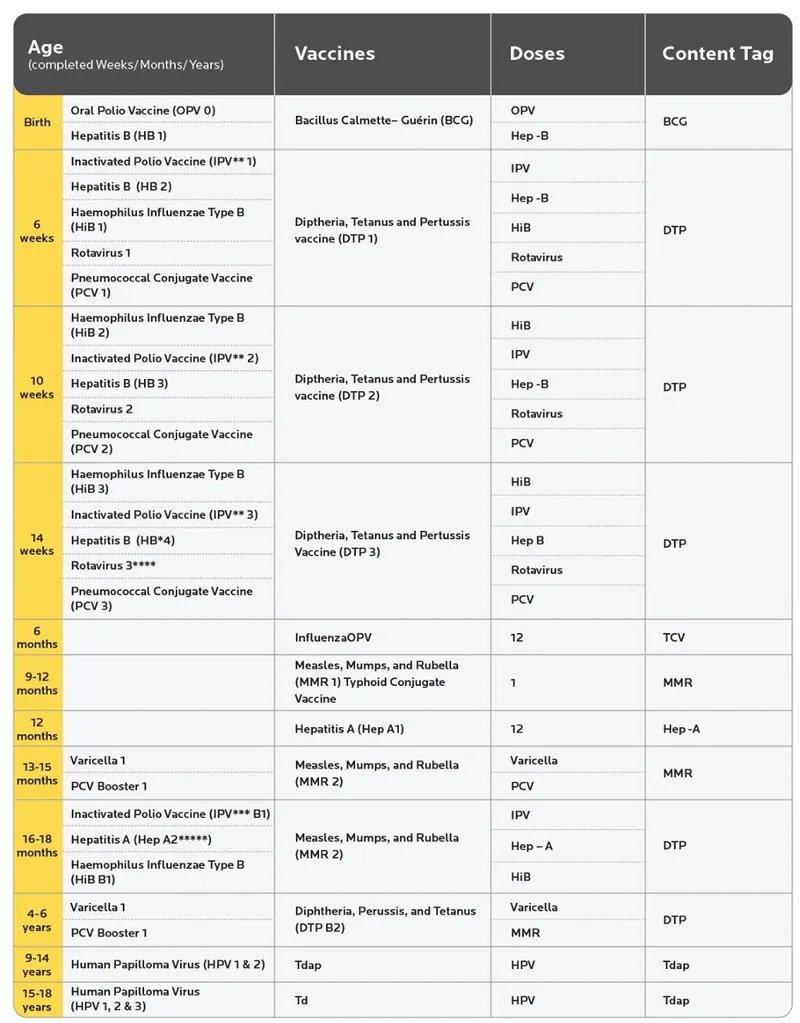
Infant Immunization: Building Early Defense
Newborns possess passive immunity from maternal antibodies that provides 3-6 months protection but interferes with vaccine responses. The immunization schedule navigates this immunological window to establish active immunity as maternal protection wanes.
📌 Remember: BIRTH-6-10-14 for India's primary infant schedule - Birth (BCG, OPV, Hepatitis B), 6 weeks (DPT, OPV, Hepatitis B), 10 weeks (DPT, OPV), 14 weeks (DPT, OPV)
-
Birth Dose Vaccines: Immediate Protection
- BCG: Single dose provides variable protection (0-80% efficacy)
- Hepatitis B: >95% efficacy in preventing perinatal transmission
- OPV: Zero dose for immediate gut immunity
- Timing rationale: immediate exposure risk
- Maternal antibody interference: minimal for these vaccines
-
Primary Series (6-14 weeks): Foundation Building
- DPT: 3 doses achieve >95% protection against diphtheria/tetanus
- OPV: 3 doses provide >99% protection against paralytic polio
- Hepatitis B: 3 doses achieve >95% seroconversion
- Interval requirement: minimum 4 weeks between doses
- Maximum interval: no upper limit (series continues)
⭐ Clinical Pearl: The 6-week starting point for most vaccines represents the optimal balance between maternal antibody decline (allowing vaccine response) and disease susceptibility (before peak infection risk at 2-6 months).
Booster Strategy: Reinforcing Protection
Booster doses exploit immunological memory to achieve rapid, robust secondary immune responses. The timing of boosters depends on antibody kinetics, disease epidemiology, and memory cell persistence.
-
Childhood Boosters (16-24 months)
- DPT Booster 1: Reinforces waning antibody levels
- OPV Booster: Enhances intestinal immunity
- MMR: First dose at 9-12 months (maternal antibody clearance)
- Measles antibody persistence: >20 years after vaccination
- Mumps/Rubella: >15 years protection duration
-
School Entry Boosters (5-6 years)
- DPT Booster 2: Maintains protection through school exposure
- OPV: Final dose for lifelong immunity
- MMR Booster: Ensures >99% population immunity
- Rationale: increased transmission in school settings
- Coverage target: >95% for herd immunity
💡 Master This: Booster timing exploits the immunological memory principle-secondary responses occur 10x faster and achieve 10-100x higher antibody levels than primary responses, providing superior protection with minimal antigen exposure.
| Age Group | Vaccines | Primary Goal | Protection Duration | Coverage Target |
|---|---|---|---|---|
| Birth | BCG, HepB, OPV | Immediate protection | Variable-Lifelong | >90% |
| 6-14 weeks | DPT, OPV, HepB | Primary immunity | 3-5 years | >95% |
| 9-12 months | MMR, JE | Maternal antibody gap | 15-20 years | >95% |
| 16-24 months | DPT, OPV boosters | Reinforce immunity | 10+ years | >90% |
| 5-6 years | DPT, OPV, MMR | School entry protection | Lifelong | >95% |
| 10-16 years | Td, TT | Adolescent/adult immunity | 10 years | >85% |
Immunocompromised individuals, pregnant women, and travelers require modified schedules that account for altered immune responses, safety considerations, and specific risk exposures.
-
Immunocompromised Patients
- Live vaccines: Generally contraindicated
- Inactivated vaccines: additional doses may be required
- Timing: Vaccinate before immunosuppression when possible
- Response rates: 50-80% of normal population
- Monitoring: serological testing to confirm response
-
Pregnancy Immunization
- Tetanus toxoid: 2 doses during pregnancy
- Influenza: inactivated vaccine any trimester
- Contraindications: all live vaccines
- Maternal protection: direct antibody response
- Neonatal protection: passive antibody transfer

⭐ Clinical Pearl: Catch-up immunization follows the principle that interrupted schedules need not restart-continue from where stopped, as the immune system remembers previous doses regardless of interval length.
Understanding immunization scheduling enables clinicians to optimize protection for individuals and populations, modify schedules for special circumstances, and contribute to disease elimination through strategic timing of vaccine administration.
🗓️ Immunization Scheduling Architecture: Timing Precision for Maximum Protection
🔍 Vaccine-Preventable Disease Patterns: Epidemiological Intelligence
High-Transmission Diseases: Airborne Threats
Respiratory-transmitted diseases achieve basic reproduction numbers (R₀) of 12-18 for measles, 4-7 for pertussis, and 5-7 for varicella, making them highly contagious and requiring >90% vaccination coverage for elimination.
📌 Remember: MEASLES-MUMPS-PERTUSSIS for high R₀ diseases - Measles (R₀=12-18), Epidemic potential, Airborne transmission, Seasonal peaks, Lifelong immunity, Elimination possible, School outbreaks
-
Measles: The Transmission Champion
- R₀ = 12-18: Most contagious vaccine-preventable disease
- Attack rate: >90% in susceptible populations
- Infectious period: 4 days before to 4 days after rash onset
- Airborne persistence: 2 hours in closed spaces
- Herd immunity threshold: >95% vaccination coverage
- Case fatality rate: 0.1-0.2% (developed countries), >10% (developing)
- Complications: 30% of cases (pneumonia, encephalitis, blindness)
-
Pertussis: The Resurgent Threat
- R₀ = 4-7: High transmission in close contacts
- Infectious period: 3 weeks without treatment, 5 days with antibiotics
- Age vulnerability: <6 months highest mortality risk
- Infant mortality: >1% in first 6 months
- Adult reservoir: waning immunity after 5-10 years
- Seasonal pattern: late summer/early fall peaks
⭐ Clinical Pearl: Measles serves as an indicator disease for immunization program quality-measles outbreaks signal <95% coverage and predict vulnerability to other vaccine-preventable diseases with lower transmission rates.
Moderate-Transmission Diseases: Strategic Targets
Diseases with R₀ = 2-6 require 80-85% coverage for control and present opportunities for regional elimination through sustained vaccination programs.
-
Polio: Elimination Success Story
- R₀ = 2-5: Moderate transmission enables elimination
- Paralytic rate: 1 in 200 infections
- Infectious shedding: weeks to months in stool
- Global reduction: >99.9% since 1988 (350,000 → <100 cases)
- Remaining endemic countries: Afghanistan, Pakistan only
- Eradication strategy: >95% coverage with surveillance
-
Rubella: Congenital Risk Focus
- R₀ = 2-3: Lower transmission than measles
- Congenital rubella syndrome: >90% risk in first trimester
- Elimination target: <1 case per 100,000 births
- Vaccination strategy: MMR achieving >95% coverage
- Surveillance focus: pregnant women and newborns
| Disease | R₀ Value | Herd Immunity Threshold | Elimination Status | Key Strategy |
|---|---|---|---|---|
| Measles | 12-18 | >95% | Eliminated (many regions) | High coverage MMR |
| Pertussis | 4-7 | >85% | Controlled | Maternal vaccination |
| Polio | 2-5 | >80% | Near eradication | OPV + surveillance |
| Rubella | 2-3 | >80% | Eliminated (many regions) | MMR + CRS prevention |
| Mumps | 4-7 | >85% | Controlled | Two-dose MMR |
| Varicella | 5-7 | >85% | Controlled | Routine childhood vaccination |
Diseases transmitted through specific routes (contaminated food/water, vectors, direct contact) or affecting specific populations require targeted vaccination strategies rather than universal coverage.
-
Hepatitis A: Sanitation-Linked Disease
- Transmission: Fecal-oral route, contaminated food/water
- Epidemiology: Endemic in poor sanitation areas
- Vaccination strategy: travelers, high-risk groups
- Seroprevalence: >90% in endemic areas by age 10
- Vaccine efficacy: >95% with 2-dose schedule
- Duration: >20 years protection
-
Japanese Encephalitis: Vector-Borne Threat
- Transmission: Culex mosquitoes, rural/agricultural areas
- Seasonality: monsoon/post-monsoon peaks
- Case fatality: 20-30%, neurological sequelae 30-50%
- Vaccination target: endemic districts with >1 case/100,000
- Vaccine efficacy: >90% with single dose
💡 Master This: Disease transmission patterns determine vaccination strategies-high R₀ diseases require universal high coverage, moderate R₀ diseases enable elimination programs, and low R₀ diseases need targeted approaches based on risk factors.
Understanding vaccine-preventable disease patterns enables clinicians to predict outbreak risks, implement appropriate prevention strategies, and contribute to disease elimination efforts through evidence-based vaccination programs.
🔍 Vaccine-Preventable Disease Patterns: Epidemiological Intelligence
⚖️ Vaccination Decision Algorithms: Evidence-Based Clinical Protocols
Contraindication Assessment: Safety First Protocol
Absolute contraindications represent situations where vaccination poses unacceptable risk and must be avoided. Relative contraindications require risk-benefit analysis and may allow vaccination under specific circumstances.
📌 Remember: LIVE-IMMUNE-ALLERGY for major contraindications - Live vaccines in immunocompromised, Illness (moderate/severe), Vaccine allergy, Encephalopathy (pertussis), Immune deficiency, Malignancy, Medications (immunosuppressive), Unstable conditions, Neurologic disorders, Egg allergy (some vaccines)
-
Live Vaccine Contraindications
- Primary immunodeficiency: Severe combined immunodeficiency, agammaglobulinemia
- Secondary immunodeficiency: HIV with CD4 <200, malignancy, immunosuppressive therapy
- Pregnancy: All live vaccines contraindicated
- Recent blood products: 3-11 months delay depending on product
- Immunoglobulin interference: 3 months for IVIG, 11 months for high-dose
- Mechanism: passive antibodies neutralize vaccine virus
-
General Contraindications
- Severe illness: Moderate to severe acute illness with/without fever
- Previous severe reaction: Anaphylaxis within 4 hours of vaccination
- Component allergy: Known allergy to vaccine components
- Egg allergy: Influenza, yellow fever vaccines (case-by-case)
- Gelatin allergy: MMR, varicella vaccines
- Neomycin allergy: MMR, IPV, varicella vaccines
⭐ Clinical Pearl: Minor illness (low-grade fever <38°C, upper respiratory symptoms) is NOT a contraindication to vaccination-delaying vaccination for minor illness reduces coverage and increases disease risk without safety benefit.
Special Population Protocols: Tailored Approaches
Specific populations require modified vaccination approaches based on altered immune responses, increased disease risk, or safety considerations.
-
Immunocompromised Patients: Enhanced Protection Strategy
- Inactivated vaccines: Additional doses often required
- Live vaccines: Generally contraindicated
- Timing: Vaccinate before planned immunosuppression
- Monitoring: Serological testing to confirm response
- Response rates: 50-80% of immunocompetent individuals
- Booster frequency: More frequent than standard schedule
- Family vaccination: Close contacts should receive live vaccines (except OPV)
-
Pregnancy Vaccination: Maternal-Fetal Protection
- Recommended: Tdap (27-36 weeks), influenza (any trimester)
- Contraindicated: All live vaccines
- Timing: Tdap optimally at 32 weeks for maximum antibody transfer
- Maternal antibody transfer: Peak at 32-36 weeks gestation
- Neonatal protection: 2-3 months passive immunity
- Pertussis prevention: >90% efficacy in infants <2 months
| Patient Population | Live Vaccines | Inactivated Vaccines | Special Considerations | Monitoring Required |
|---|---|---|---|---|
| Immunocompromised | Contraindicated | Additional doses | Pre-immunosuppression timing | Serological testing |
| Pregnancy | Contraindicated | Selected vaccines | Tdap at 32 weeks | Standard prenatal |
| Elderly (>65) | Standard | High-dose options | Pneumococcal, zoster | Annual influenza |
| Healthcare Workers | Required | Enhanced schedule | Hepatitis B, MMR | Serological confirmation |
| Travelers | Risk-based | Geographic-specific | Yellow fever, typhoid | Pre-travel consultation |
| Chronic Disease | Standard | Additional vaccines | Pneumococcal, influenza | Disease-specific |
Adverse Events Following Immunization (AEFI) require systematic assessment, management, and reporting to maintain vaccine safety and public confidence.
-
Immediate Reactions (0-4 hours)
- Anaphylaxis: 1 in 1,000,000 doses, requires epinephrine
- Vasovagal syncope: Common in adolescents, position and observation
- Local reactions: Pain, swelling at injection site
- Management: Cold compress, analgesics, observation
- Duration: 24-48 hours for most reactions
-
Delayed Reactions (1-4 weeks)
- Febrile seizures: 1 in 3,000-4,000 MMR doses
- Intussusception: 1-2 per 100,000 rotavirus doses
- Shoulder injury: SIRVA from improper injection technique
- Risk factors: High injection, excessive force, wrong needle length
- Prevention: Proper technique, anatomical landmarks
💡 Master This: AEFI causality assessment follows Bradford Hill criteria-temporal relationship, biological plausibility, consistency, and dose-response relationship determine whether events are vaccine-related, coincidental, or indeterminate.
Understanding vaccination decision algorithms enables clinicians to safely administer vaccines, manage adverse events appropriately, and maintain public confidence in immunization programs through evidence-based practice.
⚖️ Vaccination Decision Algorithms: Evidence-Based Clinical Protocols
🔗 Population Immunity Dynamics: The Network Effect Architecture
Herd Immunity Mathematics: The Protection Equation
Herd immunity threshold depends on basic reproduction number (R₀) and follows the formula: Herd Immunity Threshold = (R₀ - 1) / R₀. This mathematical relationship determines the minimum vaccination coverage required to prevent epidemic spread.
📌 Remember: R₀-THRESHOLD-COVERAGE calculation - R₀ determines threshold, threshold determines coverage, coverage determines protection
-
High R₀ Diseases: Demanding Thresholds
- Measles (R₀ = 15): Threshold = 93%, requires >95% coverage
- Pertussis (R₀ = 6): Threshold = 83%, requires >85% coverage
- Varicella (R₀ = 6): Threshold = 83%, requires >85% coverage
- Coverage buffer: 2-5% above threshold for demographic heterogeneity
- Vaccine efficacy factor: Coverage = Threshold / Efficacy
- Example: Measles requires 95% / 0.95 = 100% coverage for 95% efficacy
-
Moderate R₀ Diseases: Achievable Targets
- Polio (R₀ = 3): Threshold = 67%, requires >80% coverage
- Rubella (R₀ = 3): Threshold = 67%, requires >80% coverage
- Mumps (R₀ = 4): Threshold = 75%, requires >85% coverage
- Elimination feasibility: Higher due to lower thresholds
- Maintenance requirement: Sustained coverage over multiple years
⭐ Clinical Pearl: Demographic heterogeneity requires higher coverage than mathematical thresholds suggest-urban areas need 5-10% higher coverage than rural areas due to increased mixing patterns and transmission opportunities.
Vaccination Coverage Assessment: Measuring Protection
Coverage assessment uses multiple indicators to evaluate population immunity and identify vulnerability gaps that could lead to outbreaks.
-
Coverage Indicators: Multi-Dimensional Assessment
- Administrative coverage: Doses delivered / Target population
- Survey coverage: Household surveys with vaccination cards
- Serological coverage: Antibody prevalence in population samples
- Administrative bias: Over-reporting due to denominator errors
- Survey accuracy: ±5% with proper sampling
- Serological gold standard: Actual immunity measurement
-
Spatial Analysis: Geographic Vulnerability
- District-level coverage: Identifies low-coverage pockets
- Urban-rural disparities: Urban often 5-15% higher coverage
- Socioeconomic gradients: Wealth quintile differences of 20-40%
- High-risk areas: <80% coverage for any vaccine
- Outbreak prediction: Spatial clustering of susceptible individuals
- Intervention targeting: Micro-planning for high-risk areas
| Coverage Assessment Method | Accuracy | Cost | Timeliness | Population Level |
|---|---|---|---|---|
| Administrative Data | ±10-20% | Low | Real-time | District/State |
| Household Surveys | ±5% | High | Annual | National/State |
| School Surveys | ±3% | Medium | Annual | District |
| Serological Surveys | ±2% | Very High | 3-5 years | National |
| Rapid Monitoring | ±8% | Medium | Quarterly | District |
Disease outbreaks occur when effective reproduction number (Re) exceeds 1.0, indicating sustained transmission in the population. Understanding outbreak dynamics enables rapid response and containment strategies.
-
Outbreak Triggers: Critical Factors
- Coverage gaps: <80% coverage in susceptible populations
- Waning immunity: Antibody decline over 5-15 years
- Population mixing: Mass gatherings, migration, travel
- Vaccine failure: Primary (no response) or secondary (waning)
- Primary failure: 2-5% of vaccinated individuals
- Secondary failure: Time-dependent antibody decline
- Importation risk: Travel-related cases in susceptible populations
-
Outbreak Response: Rapid Containment
- Case investigation: Within 24 hours of notification
- Contact tracing: All contacts within infectious period
- Ring vaccination: Contacts and high-risk populations
- Mass vaccination: Outbreak areas achieving >95% coverage
- Response timeline: <72 hours for effective containment
- Coverage target: >95% in outbreak areas
- Age targeting: Susceptible age groups based on disease epidemiology
💡 Master This: Population immunity operates as a complex adaptive system where local coverage variations, demographic mixing patterns, and temporal immunity changes interact to determine disease transmission dynamics and outbreak risk.
Understanding population immunity dynamics enables public health professionals to design effective vaccination strategies, predict outbreak risks, and implement targeted interventions that protect entire communities through strategic immunization programs.
🔗 Population Immunity Dynamics: The Network Effect Architecture
🎯 Immunization Mastery Toolkit: Clinical Excellence Framework
Essential Clinical Assessment Matrix
Systematic patient evaluation integrates medical history, vaccination status, risk factors, and contraindications to create individualized immunization plans that optimize protection while ensuring safety.
📌 Remember: VACCINE-SAFE-EFFECTIVE assessment - Vaccination history, Age-appropriate schedule, Contraindications, Chronic conditions, Immune status, Needs assessment, Exposure risks, Safety profile, Adverse events, Follow-up, Efficacy monitoring
-
Rapid Assessment Protocol (5-Minute Evaluation)
- Vaccination card review: Complete vs. incomplete series
- Age-appropriate vaccines: Due today vs. future scheduling
- Contraindication screening: Absolute vs. relative vs. none
- Risk factor identification: High-risk conditions requiring additional vaccines
- Assessment time: <5 minutes for routine evaluation
- Documentation: Electronic health records with decision support
- Quality metrics: >95% appropriate vaccination decisions
-
High-Risk Population Identification
- Immunocompromised patients: Modified schedules and additional monitoring
- Chronic disease patients: Enhanced vaccination (pneumococcal, influenza)
- Healthcare workers: Occupational requirements and annual updates
- Travelers: Geographic-specific vaccines and timing considerations
- Risk stratification: Low, moderate, high based on multiple factors
- Intervention intensity: Proportional to risk level
| Risk Category | Assessment Time | Additional Vaccines | Monitoring Frequency | Special Considerations |
|---|---|---|---|---|
| Standard Risk | 5 minutes | Routine schedule | Annual | Standard protocols |
| Chronic Disease | 10 minutes | Pneumococcal, influenza | Bi-annual | Disease-specific |
| Immunocompromised | 15 minutes | Inactivated only | Quarterly | Serological testing |
| Healthcare Worker | 10 minutes | Hepatitis B, annual flu | Annual | Occupational health |
| International Traveler | 20 minutes | Geographic-specific | Pre-travel | Risk assessment |
| Pregnancy | 15 minutes | Tdap, influenza | Prenatal schedule | Timing optimization |
Continuous quality improvement uses performance indicators, outcome monitoring, and systematic feedback to optimize vaccination delivery and maintain high standards of care.
-
Coverage Quality Indicators
- Timely vaccination: >90% of vaccines on schedule
- Series completion: >95% completion rates for multi-dose vaccines
- Missed opportunities: <5% of eligible visits without appropriate vaccination
- Contraindication accuracy: >98% appropriate contraindication assessment
- Measurement frequency: Monthly for key indicators
- Benchmark comparison: National and regional standards
- Improvement targets: Annual goals with quarterly monitoring
-
Safety Monitoring Systems
- AEFI reporting: 100% of serious events reported within 24 hours
- Storage compliance: >99% cold chain maintenance
- Administration errors: <1% of total doses administered
- Error categories: Wrong vaccine, wrong dose, wrong patient, wrong timing
- Root cause analysis: Systematic investigation of all errors
- Prevention strategies: Process improvement and staff training
Advanced Clinical Integration: Systems Thinking
Immunization excellence requires systems-level thinking that integrates individual patient care, population health goals, and public health surveillance to achieve optimal outcomes across multiple levels of intervention.
-
Electronic Health Record Integration
- Decision support systems: Real-time recommendations based on patient data
- Reminder systems: Automated notifications for due vaccines
- Registry connectivity: Population-level tracking and coverage monitoring
- Alert accuracy: >95% appropriate vaccination recommendations
- False positive rate: <5% inappropriate alerts
- User satisfaction: >85% clinician acceptance of decision support
-
Population Health Integration
- Coverage monitoring: Real-time tracking of population immunity
- Outbreak response: Rapid identification of susceptible populations
- Health equity: Targeted interventions for underserved populations
- Data integration: Individual and population-level metrics
- Intervention targeting: Geographic and demographic precision
- Outcome measurement: Health equity and disease prevention indicators
⭐ Clinical Pearl: Immunization excellence requires triple integration-clinical expertise (individual patient optimization), population health knowledge (community protection), and systems thinking (quality improvement)-to achieve >95% coverage with <1% adverse events.
💡 Master This: The immunization mastery framework transforms routine vaccination into strategic population health intervention by integrating evidence-based protocols, quality improvement systems, and population health surveillance to achieve disease elimination through clinical excellence.
Understanding the immunization mastery toolkit enables healthcare professionals to deliver high-quality vaccination services, contribute to population health goals, and maintain the public trust essential for successful immunization programs that protect individuals and communities from vaccine-preventable diseases.
🎯 Immunization Mastery Toolkit: Clinical Excellence Framework
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more
Have doubts about this lesson?
Ask Rezzy, your AI Study Mate, to explain anything you didn't understand
Everything you need for NEET-PG prep
Get full Oncourse access with lessons, practice questions, flashcards and AI study tools.
Scan to download app
























