Epidemiological Study Designs - Study Blueprints
- Foundation: Plans to study disease distribution & determinants.
- Two Main Types:
- Observational: Researcher observes existing patterns; no intervention.
- Descriptive: Who, What, Where, When? (Hypothesis generating)
- Case Report/Series
- Ecological (population-level data)
- Cross-sectional (prevalence at a point in time; snapshot)
- Analytical: Why? How? (Hypothesis testing)
- Case-Control (Odds Ratio; retrospective: outcome → exposure)
- Cohort (Relative Risk, Incidence; prospective/retrospective: exposure → outcome)
- Descriptive: Who, What, Where, When? (Hypothesis generating)
- Experimental (Interventional): Researcher actively manipulates exposure to assess effect.
- Randomized Controlled Trial (RCT): Gold standard for causality; random allocation.
- Non-Randomized Trials: Intervention, but no random allocation.
- Observational: Researcher observes existing patterns; no intervention.
⭐ Cohort studies are best for determining incidence and the natural history of a disease.
Epidemiological Study Designs - Snapshot & Rewind
- Observational Designs:
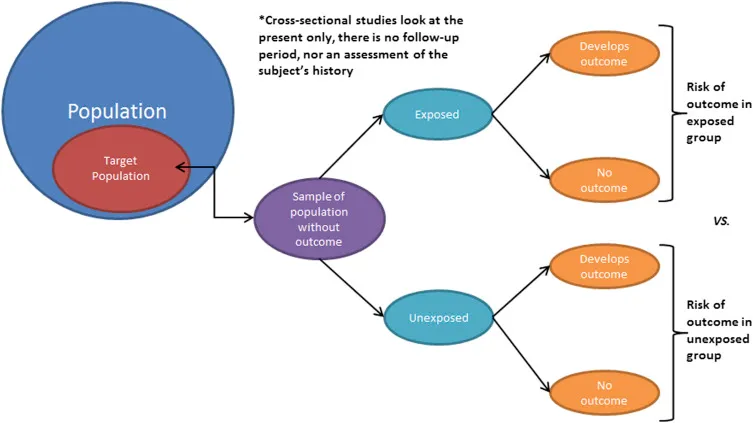
- Cross-sectional (Prevalence Study):
- "Snapshot": Assesses exposure & outcome simultaneously at one point in time.
- Measures prevalence.
- Uses: Health surveys, hypothesis generation.
- Adv: Quick, inexpensive.
- Disadv: No temporality (cannot infer causation), not for rare diseases.
- Measure: Prevalence Ratio, Odds Ratio.
- Case-Control (Retrospective/Trohoc Study):
- "Rewind": Starts with outcome (Cases with disease vs. Controls without disease).
- Looks back (retrospective) for past exposure.
- Uses: Rare diseases, outbreak investigation. 📌 Mnemonic for selection of controls: Population-based, Hospital-based, Special group (PHS).
- Adv: Quick, inexpensive, good for rare diseases, multiple exposures.
- Disadv: Recall bias, selection bias, interviewer bias. Cannot calculate incidence/prevalence directly.
- Measure: Odds Ratio ($OR = (ad) / (bc)$).
⭐ Odds Ratio from a case-control study can approximate Relative Risk if the disease is rare (prevalence < 10%).
- Cross-sectional (Prevalence Study):
Epidemiological Study Designs - Forward & Future
- Cohort Studies (Observational): Follows groups from exposure to outcome.
- Prospective: Present exposure → Future outcome.
- Strengths: Establishes temporality, measures incidence, $RR = [a/(a+b)] / [c/(c+d)]$, $AR$. Good for rare exposures.
- Weaknesses: Costly, long duration, attrition.
- Retrospective (Historical): Past exposure (records) → Outcome.
- Strengths: Quicker, cheaper.
- Weaknesses: Record dependency, bias.
- 📌 COhort = COming Outcomes.
- Prospective: Present exposure → Future outcome.
- Randomized Controlled Trials (RCTs) (Experimental): Intervention → Outcome.
- Gold standard for causality. Investigator assigns exposure.
- Key Features: Randomization, Blinding, Control.
- Measures: Efficacy, $RR$, $AR$.
- Strengths: ↑Internal validity, ↓bias, causal inference.
- Weaknesses: Costly, ethical limits, ↓external validity.
⭐ RCTs provide the strongest evidence for causal relationships.
 oka
oka
Epidemiological Study Designs - Bias & Blunders
- Bias: Systematic error in design, conduct, or analysis leading to erroneous association.
- Selection Bias: Non-random subject selection.
- Berksonian bias: Hospital-based controls differ from general population.
- Neyman bias (Incidence-Prevalence bias): Selective survival in prevalent cases.
- Information Bias (Measurement/Misclassification Bias): Errors in data collection/measurement.
- Recall bias: Cases recall exposure more/less than controls (📌 common in case-control).
- Interviewer bias: Interviewer influences responses.
- Selection Bias: Non-random subject selection.
- Confounding: Distortion of exposure-disease relationship by a third variable (confounder).
- Control: Randomization, Restriction, Matching, Stratification, Multivariate analysis.
- Effect Modification: True difference in effect across strata of a third variable. Not a bias.
⭐ Randomization is the best method to control for unknown confounders in RCTs.
- Bradford Hill Criteria: Guidelines for inferring causality (e.g., Strength, Consistency, Temporality, Dose-response).
High‑Yield Points - ⚡ Biggest Takeaways
- Case-control studies: Retrospective, start with outcome, calculate Odds Ratio (OR); efficient for rare diseases.
- Cohort studies: Prospective or retrospective, start with exposure, calculate Relative Risk (RR), Incidence; establish temporality.
- Randomized Controlled Trials (RCTs): Gold standard for causality; use randomization, control group, blinding.
- Cross-sectional studies: Measure prevalence (disease & exposure) at a single point in time; "snapshot".
- Ecological studies: Use group-level data (not individual); prone to ecological fallacy.
- Bias control: Randomization & blinding in RCTs; matching in case-control for confounders.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

