Critical Appraisal - Study Sleuthing 101
- What: Systematic evaluation of research for validity, results, and relevance.
- Why: Essential for evidence-based medicine (EBM); informs clinical decisions.
- Key Appraisal Questions:
- Validity: Were study methods sound? Biases (selection, information, confounding) minimized?
- Results: What are the findings? Statistically significant (p-value)? Clinically important (effect size)? Precise (Confidence Intervals)?
- Applicability: Are results relevant to my patient population? Can I use them in practice?
- 📌 Checklist Approach: Use standardized critical appraisal tools (e.g., CASP checklists).

⭐ Internal validity (i.e., the extent to which the study's conclusions are true for the study population) is a prerequisite for external validity (i.e., generalizability).
Critical Appraisal - Flaw Finder's Guide
Assess study validity by identifying potential flaws:
- Bias (Systematic Error): Flaw in design/conduct; distorts results. Not reduced by ↑ sample size.
- Selection Bias: Non-comparable groups (e.g., Berksonian, Neyman's, volunteer).
- Information/Observation Bias: Incorrect data collection/measurement (e.g., Recall, Interviewer, Hawthorne). Misclassification (Differential/Non-differential).
- Confounding: Distortion by a third variable linked to exposure & outcome. Not on causal pathway.
- Control: Randomization, Restriction, Matching, Stratification, Multivariate analysis.
- Chance (Random Error): Due to sampling variability. Reduced by ↑ sample size.
- Assessed by: p-value, Confidence Intervals (CI).
- Type I error ($\alpha$): False positive.
- Type II error ($\beta$): False negative. Power = $1 - \beta$.
⭐ Randomization is the best method to control for unknown confounders.
Critical Appraisal - Design Dissection
- Randomized Controlled Trials (RCTs):
- Guideline: CONSORT (Consolidated Standards of Reporting Trials).
- Key checks: Randomization, allocation concealment, blinding, Intention-To-Treat (ITT).
- Observational Studies (Cohort, Case-Control, Cross-sectional):
- Guideline: STROBE (Strengthening Reporting of Observational studies in Epidemiology).
- Key checks: Selection bias, information bias, confounding.
- Systematic Reviews & Meta-Analyses:
- Guideline: PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses).
- Key checks: Search strategy, risk of bias, heterogeneity ($I^2$), publication bias.
- Diagnostic Accuracy Studies:
- Guideline: STARD (Standards for Reporting Diagnostic Accuracy).
- Key checks: Patient spectrum, reference standard, blinding of assessors.

⭐ For meta-analyses, $I^2$ statistic quantifies heterogeneity: < 25% (low), 25-75% (moderate), > 75% (high).
Critical Appraisal - Stats Sense Check
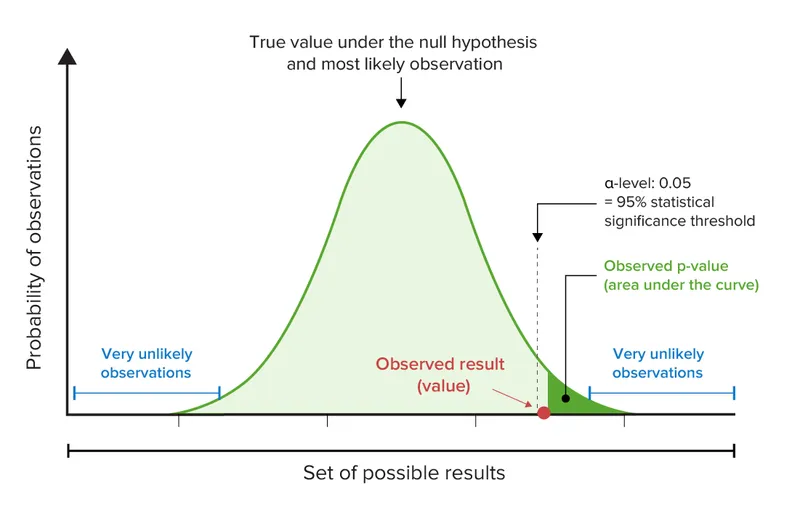
- P-value: Probability of observing the data (or more extreme) if no true effect exists. Common threshold: < 0.05.
- Small p-value ≠ clinical importance.
- Confidence Interval (CI): Range of plausible true values for an effect (e.g., RR, OR).
- 95% CI for RR/OR: If it includes 1, the result is NOT statistically significant.
- Effect Measures:
- RR (Relative Risk) / OR (Odds Ratio): Strength of association. >1 indicates ↑ risk/odds; <1 indicates ↓ risk/odds.
- ARR (Absolute Risk Reduction): $CER - EER$ (Control Event Rate - Experimental Event Rate).
- NNT (Number Needed to Treat): $1/ARR$. Lower is better.
- Significance: Distinguish statistical (p-value, CI) from clinical significance (magnitude of effect, NNT, patient relevance).
⭐ If the 95% Confidence Interval for a Relative Risk (RR) or Odds Ratio (OR) includes the value 1, the association is NOT statistically significant at the p < 0.05 level.
High‑Yield Points - ⚡ Biggest Takeaways
- Identify and assess potential Bias: selection bias, information bias (recall, interviewer), and confounding.
- Confounding distorts associations; control with randomization, matching, stratification, or multivariate analysis.
- Internal validity (study's truth) is crucial before considering external validity (generalizability).
- Examine strength of association (e.g., RR, OR) and precision using confidence intervals.
- Temporality (exposure precedes outcome) is a critical criterion for causality.
- Look for dose-response relationship and consistency of findings across different studies_._
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

