Overview of Analytical Epi - Hypothesis Hunters
- Purpose: To test pre-defined hypotheses about relationships between exposures and outcomes.
- Key Feature: Utilizes comparison groups (e.g., exposed vs. unexposed, cases vs. controls).
- Goal: Quantify the association and infer causality.
- Contrasts with descriptive epidemiology, which focuses on generating hypotheses by describing disease distribution.
⭐ Analytical studies are designed to test hypotheses, unlike descriptive studies which generate them.
- Answers "Why?" and "How?" disease occurs.
Case-Control Studies - Past Probers
- Retrospective: Start with outcome (cases vs. controls), look back for exposure. "Past Probers."
- Selection:
- Cases: Have the disease.
- Controls: Don't have disease; from same source population.
- Measure of Association: Odds Ratio (OR).
- Calculated as $ad/bc$. (a=exposed cases, b=exposed controls, c=unexposed cases, d=unexposed controls)
- OR > 1: ↑ risk; OR < 1: ↓ risk; OR = 1: No association.
- Advantages: Good for rare diseases, quick, less expensive, studies multiple exposures.
- Biases: 📌 People Selectively Remember (Selection, Recall), Berksonian bias.
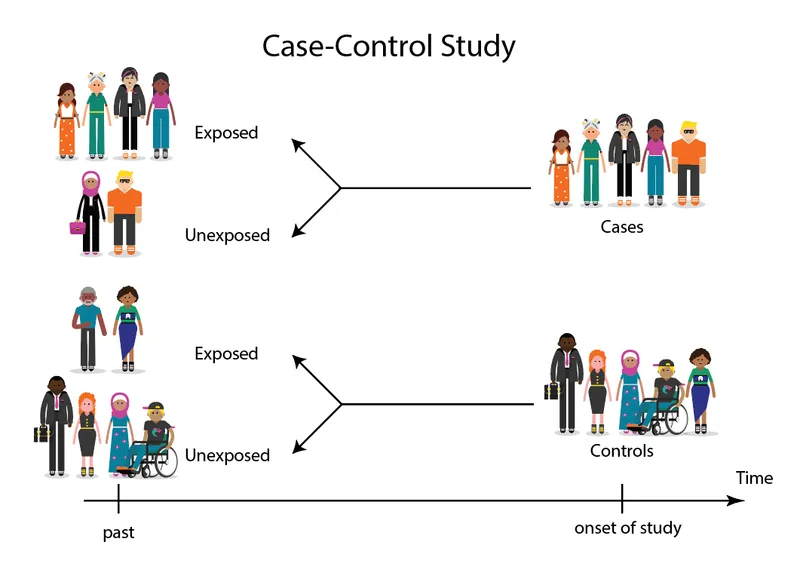
⭐ Odds Ratio (OR) is the key measure of association in case-control studies. It's suitable for rare diseases.
Cohort Studies - Future Followers
- Observational; subjects grouped by exposure, followed to assess outcome incidence.
- Types: 📌 PAR
- Prospective: Exposure → Future Outcome.
- Retrospective (Historical): Past Exposure (records) → Outcome.
- Ambidirectional: Combines past records & future follow-up.
- Key measure: Relative Risk (RR).
- Formula: $RR = [a/(a+b)] / [c/(c+d)]$.
- Strengths: Establishes temporality, studies multiple outcomes, good for rare exposures, calculates incidence.
- Weaknesses: Loss to follow-up bias, costly & lengthy (especially prospective).
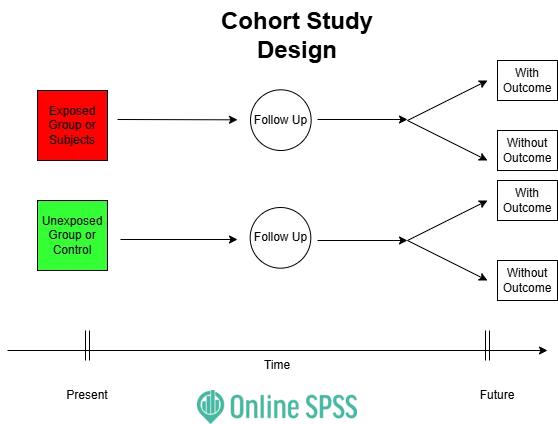
⭐ Relative Risk (RR) is the key measure of association. A major strength is establishing temporality (exposure precedes outcome).
Key Epidemiological Measures - Link Logic
| Measure | When Used (Study Type) | Calculation (2x2 table: a,b,c,d) | Interpretation |
|---|---|---|---|
| Odds Ratio (OR) | Case-Control, Cross-sectional | $ad/bc$ | Odds of exposure: cases vs. controls. |
| Relative Risk (RR) | Cohort | $(a/(a+b)) / (c/(c+d))$ | Risk of disease: exposed vs. unexposed. |
- Attributable Risk (AR) / Risk Difference (RD): $I_e - I_u$
- Attributable Fraction (AF) for exposed ($AF_e$): $(AR / I_e) \times \textbf{100}\%$ or $((RR-1)/RR) \times \textbf{100}\%$
- Population Attributable Risk (PAR): $I_p - I_u$ or $AR \times P_e$ ($P_e$: exposure prevalence in pop.)
- Population Attributable Fraction (PAF) / PAR Percent ($PAF_p$): $(PAR / I_p) \times \textbf{100}\%$ or $(P_e(RR-1) / (P_e(RR-1)+1)) \times \textbf{100}\%$
⭐ Attributable Risk (AR) or Risk Difference ($I_e - I_u$) quantifies the excess risk in the exposed group directly attributable to the exposure.
Bias, Confounding & Causality - Truth Seekers
- Bias: Systematic error in design/conduct leading to incorrect association.
- Selection Bias: Distorted due to how subjects are selected (e.g., Berksonian, Neyman).
- Information Bias (Misclassification): Errors in measuring exposure/outcome (e.g., Recall, Interviewer, Observer). 📌 RIO (Recall, Interviewer, Observer).
- Confounding: Extraneous variable associated with exposure & outcome, distorts true relationship.
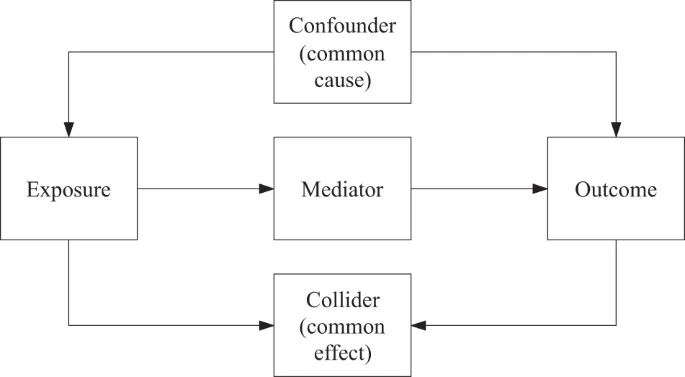
⭐ Confounding can be controlled at design (randomization, restriction, matching) or analysis stage (stratification, multivariate analysis).
- Causality (Bradford Hill): Key criteria: Temporality (E before O - essential!), Strength, Consistency, Biological gradient, Plausibility, Coherence.
High‑Yield Points - ⚡ Biggest Takeaways
- Case-control studies: Effect to cause (disease → exposure). Metric: Odds Ratio (OR). For rare diseases.
- Cohort studies: Cause to effect (exposure → disease). Metrics: Relative Risk (RR), Attributable Risk (AR). For rare exposures.
- Odds Ratio (OR) = (ad/bc). Approximates RR if disease prevalence is low.
- Relative Risk (RR) indicates strength of association. >1 means ↑ risk.
- Attributable Risk (AR) represents excess risk due to exposure.
- Key Biases: Selection bias, Recall bias (in case-control), Interviewer bias.
- Control confounding with matching, stratification, or multivariate analysis.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

