Vitamins and Coenzymes

On this page
🧬 The Molecular Machinery: Vitamins as Metabolic Catalysts
Vitamins function as precision molecular tools that transform dietary nutrients into usable energy and essential biomolecules, yet their deficiency creates predictable clinical patterns that span multiple organ systems. You'll discover how these organic cofactors enable enzymatic reactions, recognize the distinctive syndromes that emerge when they're absent, and master the laboratory and therapeutic strategies that restore metabolic balance. By integrating molecular mechanisms with clinical assessment, you'll build a systematic approach to diagnosing and treating vitamin-related disorders across diverse patient populations.
📌 Remember: Fat-ADEK (A, D, E, K) are stored in liver and adipose tissue with months-to-years reserves, while Water-B&C require daily replenishment due to rapid urinary excretion
The vitamin-coenzyme transformation follows predictable patterns that unlock metabolic mastery:
-
Water-Soluble Vitamins (B-complex + C)
- Rapid absorption via specific transporters
- Conversion to active coenzyme forms in liver
- Daily requirements: 1-100mg range
- B1 (Thiamine): 1.2mg/day → TPP coenzyme
- B2 (Riboflavin): 1.3mg/day → FAD/FMN coenzymes
- B3 (Niacin): 16mg/day → NAD+/NADP+ coenzymes
- B6 (Pyridoxine): 1.7mg/day → PLP coenzyme
-
Fat-Soluble Vitamins (A, D, E, K)
- Require bile salts for absorption
- Hepatic storage: A (1-2 years), D (2-4 months), E (8-12 months), K (1-2 weeks)
- Toxicity potential: Accumulation exceeds 10x normal levels
| Vitamin | Coenzyme Form | Primary Function | Deficiency Time | Storage Duration | Daily Requirement |
|---|---|---|---|---|---|
| B1 (Thiamine) | TPP | Decarboxylation | 2-3 weeks | Minimal | 1.2mg |
| B2 (Riboflavin) | FAD/FMN | Electron transport | 1-2 months | 2-6 weeks | 1.3mg |
| B3 (Niacin) | NAD+/NADP+ | Redox reactions | 2-4 weeks | Minimal | 16mg |
| B6 (Pyridoxine) | PLP | Amino acid metabolism | 2-6 weeks | 2-3 weeks | 1.7mg |
| B12 (Cobalamin) | Methylcobalamin | Methylation | 3-5 years | 3-5 years | 2.4μg |
💡 Master This: Water-soluble vitamin deficiencies follow the "weeks-to-months" rule, while fat-soluble deficiencies require months-to-years of inadequate intake, except vitamin K which depletes in 1-2 weeks
The coenzyme activation process reveals why certain populations develop specific deficiencies. Alcoholics develop thiamine deficiency because ethanol inhibits TPP synthesis by >70%, while vegans risk B12 deficiency due to exclusive animal source requirements.
Connect these foundational vitamin-coenzyme principles through enzymatic mechanisms to understand how molecular structure determines metabolic function.
🧬 The Molecular Machinery: Vitamins as Metabolic Catalysts
⚙️ The Catalytic Engine: How Coenzymes Drive Metabolism
📌 Remember: "REDOX-TRANSFER-CARBOXYL" - the three coenzyme mechanisms: Redox (NAD, FAD), Transfer (CoA, TPP), Carboxylation (Biotin, Folate)
Redox Coenzymes: Electron Transport Specialists
-
NAD+/NADH: +0.32V reduction potential
- Dehydrogenase reactions: Removes 2H+ + 2e-
- Cellular ratio: NAD+:NADH = 700:1 (cytosol), 8:1 (mitochondria)
- Daily turnover: 2-4 times total body NAD+ pool
-
FAD/FADH2: +0.22V reduction potential
- Flavoprotein binding: Covalent attachment to histidine residues
- Oxygen reactivity: Direct O2 reduction capability
- Substrate specificity: Acyl-CoA dehydrogenases, succinate dehydrogenase
| Coenzyme | Mechanism | Enzyme Examples | Products | Clinical Significance |
|---|---|---|---|---|
| TPP | Aldehyde transfer | Pyruvate DH, α-KGDH | Acetyl-CoA | Beriberi, Wernicke's |
| PLP | Amino group transfer | ALT, AST | Amino acids | >100 reactions |
| Biotin | CO2 carboxylation | Acetyl-CoA carboxylase | Malonyl-CoA | Fatty acid synthesis |
| Folate | One-carbon transfer | Thymidylate synthase | dTMP | DNA synthesis |
| CoA | Acyl group transfer | Fatty acid oxidation | Acetyl units | Energy metabolism |
Transfer Coenzymes: Molecular Shuttles
- Coenzyme A: Thiol group (-SH) forms high-energy thioester bonds
- Thiamine pyrophosphate (TPP): Carbanion stabilization enables aldehyde transfers
- Pyridoxal phosphate (PLP): Schiff base formation with amino groups
💡 Master This: Coenzyme binding affinity determines Km values - tight binding (nM range) indicates prosthetic groups (FAD), while loose binding (μM range) indicates cosubstrates (NAD+)
The induced-fit mechanism explains coenzyme specificity. Apoenzyme binding causes conformational changes that position reactive groups within 2-3 Angstroms of substrate bonds, reducing activation energy by 50-100 kJ/mol.
Coenzyme Regeneration Cycles:
- NAD+ regeneration: Lactate dehydrogenase (anaerobic), electron transport chain (aerobic)
- FAD regeneration: Complex II direct electron transfer to ubiquinone
- CoA regeneration: Thioesterase activity releases free CoA from acyl derivatives
Connect these catalytic mechanisms through metabolic integration to understand how coenzyme deficiencies disrupt entire metabolic networks.
⚙️ The Catalytic Engine: How Coenzymes Drive Metabolism
🎯 The Recognition Matrix: Vitamin Deficiency Patterns
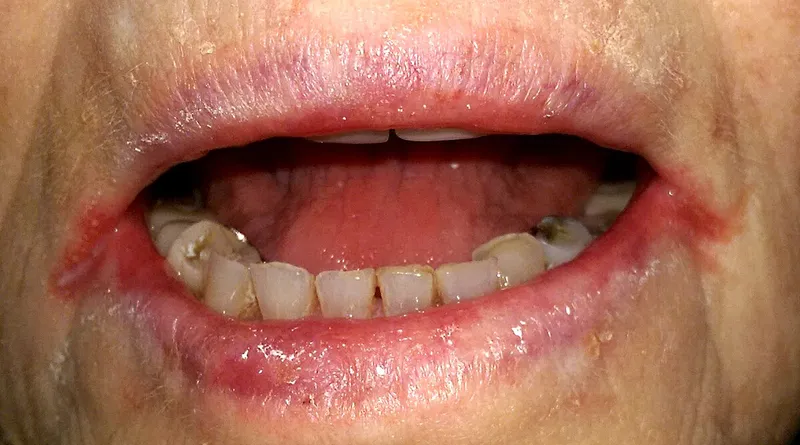
📌 Remember: "NIGHT-BLEEDS-CRACKS-TIRED" - Night blindness (A), Bleeding (K), Cracks/cheilosis (B2), Tired/beriberi (B1) for rapid vitamin deficiency screening
Neurological Pattern Recognition:
-
Peripheral neuropathy + confusion = Thiamine (B1) deficiency
- Dry beriberi: Symmetric distal neuropathy, muscle weakness
- Wet beriberi: High-output heart failure, edema
- Wernicke-Korsakoff: Ataxia, ophthalmoplegia, memory loss
-
Subacute combined degeneration = B12 deficiency
- Posterior column: Vibration/position sense loss
- Lateral corticospinal: Spastic weakness, hyperreflexia
- Peripheral nerves: Distal sensory loss
-
Seizures in infants = Pyridoxine (B6) deficiency
- GABA synthesis impairment: Glutamate decarboxylase requires PLP
- Isoniazid-induced: Competitive PLP inhibition
Hematological Fingerprints:
-
Megaloblastic anemia patterns:
- B12 deficiency: MCV >100 fL, hypersegmented neutrophils (>5 lobes)
- Folate deficiency: MCV >110 fL, normal B12 levels
- Combined deficiency: MCV >120 fL, pancytopenia
-
Microcytic anemia = B6 deficiency
- Sideroblastic pattern: Ring sideroblasts in >15% of erythroblasts
- Iron overload: Transferrin saturation >50%
| Vitamin | Neurological | Hematological | Dermatological | Metabolic |
|---|---|---|---|---|
| B1 | Beriberi, Wernicke's | Normal | Edema (wet) | Lactic acidosis |
| B2 | Rare | Normocytic anemia | Cheilosis, glossitis | Normal |
| B6 | Seizures, neuropathy | Microcytic anemia | Seborrheic dermatitis | Oxalate stones |
| B12 | SCD, dementia | Megaloblastic | Hyperpigmentation | Methylmalonic aciduria |
| Folate | Rare | Megaloblastic | Glossitis | Homocysteinemia |
Dermatological Recognition Patterns:
-
Angular cheilitis + glossitis = Riboflavin (B2) deficiency
-
Pellagrous dermatitis (4 D's) = Niacin (B3) deficiency
- Dermatitis: Photosensitive, symmetric
- Diarrhea: Malabsorption
- Dementia: Cognitive decline
- Death: Untreated progression
-
Seborrheic dermatitis = Biotin or B6 deficiency

💡 Master This: "See the pattern, test specifically" - Night blindness → Serum retinol, Megaloblastic anemia → B12/Folate/MMA, Bleeding tendency → PT/INR for vitamin K status
Metabolic Biomarker Patterns:
- Elevated pyruvate/lactate = Thiamine deficiency
- Elevated methylmalonic acid = B12 deficiency
- Elevated homocysteine = B12, Folate, or B6 deficiency
- Prolonged PT/INR = Vitamin K deficiency
High-Risk Population Screening:
- Alcoholics: B1, B6, Folate deficiency risk >80%
- Vegans: B12 deficiency risk >60% after 3-5 years
- Malabsorption: Fat-soluble vitamins (A, D, E, K) deficiency >70%
Connect these recognition patterns through systematic differential diagnosis to understand how multiple vitamin deficiencies interact and compound clinical presentations.
🎯 The Recognition Matrix: Vitamin Deficiency Patterns
🔬 The Diagnostic Arsenal: Laboratory Precision Tools
📌 Remember: "MEASURE-METABOLITE-FUNCTION" - Serum vitamin (direct), Metabolite accumulation (indirect), Enzyme activity (functional) for comprehensive vitamin status assessment
Direct Vitamin Measurements:
-
Serum B12: Normal >300 pg/mL, Deficient <200 pg/mL
- Gray zone: 200-300 pg/mL requires MMA/homocysteine testing
- Holotranscobalamin: Active B12, >25 pmol/L indicates adequacy
-
Serum Folate: Normal >3 ng/mL, Deficient <2 ng/mL
- RBC folate: Better tissue stores indicator, >140 ng/mL
- Falsely elevated: Recent dietary intake within 24-48 hours
-
25-hydroxyvitamin D: Optimal >30 ng/mL, Deficient <20 ng/mL
- Seasonal variation: ±40% difference winter vs summer
- 1,25-dihydroxy D: Normal in deficiency due to PTH stimulation
Indirect Metabolite Assessment:
-
Methylmalonic acid (MMA): B12 deficiency marker
- Normal: <280 nmol/L
- B12 deficiency: >1000 nmol/L
- Renal function: Falsely elevated in CKD
-
Homocysteine: B12, Folate, B6 deficiency
- Normal: <15 μmol/L
- Elevated: >20 μmol/L indicates deficiency
- Cardiovascular risk: >10 μmol/L increases CVD risk by 20%
| Test | Normal Range | Deficiency Threshold | Specificity | Sensitivity | Clinical Notes |
|---|---|---|---|---|---|
| Serum B12 | >300 pg/mL | <200 pg/mL | 95% | 80% | False normal in 30% |
| MMA | <280 nmol/L | >1000 nmol/L | 99% | 95% | Gold standard for B12 |
| Homocysteine | <15 μmol/L | >20 μmol/L | 85% | 90% | Multiple vitamins |
| RBC Folate | >140 ng/mL | <100 ng/mL | 90% | 85% | Tissue stores |
| 25(OH)D | >30 ng/mL | <20 ng/mL | 95% | 90% | Seasonal variation |
Functional Enzyme Assays:
-
Transketolase activity: Thiamine (B1) status
- Thiamine pyrophosphate effect: >25% indicates deficiency
- Erythrocyte transketolase: Functional B1 assessment
-
Erythrocyte glutathione reductase: Riboflavin (B2) status
- FAD stimulation test: >40% indicates deficiency
- Activity coefficient: >1.4 suggests inadequate B2
-
Pyridoxal phosphate: B6 functional status
- Plasma PLP: <20 nmol/L indicates deficiency
- Tryptophan load test: Xanthurenic acid excretion >50 mg/day
Specialized Testing Protocols:
- Schilling test: B12 absorption assessment (historical)
- D-xylose test: Small bowel absorption function
- Fat-soluble vitamin panel: A, D, E, K simultaneous measurement
💡 Master This: "Normal serum ≠ Normal tissue" - 30% of B12-deficient patients have normal serum B12 but elevated MMA, requiring functional assessment for accurate diagnosis
Therapeutic Monitoring:
- B12 replacement: MMA normalization in 2-4 weeks
- Folate replacement: Homocysteine normalization in 1-2 weeks
- Vitamin D: 25(OH)D target >30 ng/mL in 6-8 weeks
Connect these diagnostic precision tools through evidence-based treatment algorithms to understand how laboratory results guide therapeutic interventions and monitoring strategies.
🔬 The Diagnostic Arsenal: Laboratory Precision Tools
💊 The Therapeutic Command Center: Evidence-Based Interventions
📌 Remember: "DOSE-ROUTE-DURATION-MONITOR" - High-dose replacement, Appropriate route (oral vs parenteral), Adequate duration, Biochemical monitoring for successful vitamin deficiency treatment
B12 Deficiency Treatment Protocols:
-
Severe deficiency (neurological symptoms):
- Cyanocobalamin: 1000 μg IM daily × 7 days
- Maintenance: 1000 μg IM monthly or 2000 μg oral daily
- Monitoring: MMA normalization in 2-4 weeks
-
Pernicious anemia: Lifelong B12 replacement
- Loading dose: 1000 μg IM × 6 doses over 2-3 weeks
- Maintenance: 1000 μg IM every 3 months
- Oral alternative: 2000 μg daily (absorption 1-2% via passive diffusion)
Folate Deficiency Management:
- Acute replacement: 5 mg oral daily × 4 months
- Pregnancy: 400-800 μg daily (prevention), 5 mg daily (treatment)
- Malabsorption: 15 mg daily or parenteral administration
- Drug interactions: Methotrexate, phenytoin require higher doses
| Vitamin | Deficiency Dose | Duration | Maintenance | Route | Monitoring |
|---|---|---|---|---|---|
| B1 | 100 mg daily | 2-4 weeks | 10 mg daily | Oral/IV | Clinical response |
| B2 | 10-20 mg daily | 4-6 weeks | 2 mg daily | Oral | Enzyme activity |
| B6 | 50-100 mg daily | 3-4 weeks | 2 mg daily | Oral | PLP levels |
| B12 | 1000 μg IM | Weekly × 4 | Monthly IM | IM/Oral | MMA, B12 |
| Folate | 5 mg daily | 4 months | 400 μg daily | Oral | RBC folate |
Fat-Soluble Vitamin Replacement:
-
Vitamin A deficiency:
- Severe: 200,000 IU oral × 2 days, then weekly × 4 weeks
- Xerophthalmia: Emergency treatment to prevent blindness
- Maintenance: 10,000-25,000 IU daily
-
Vitamin D deficiency:
- Severe: 50,000 IU weekly × 8 weeks or 6000 IU daily
- Maintenance: 1000-2000 IU daily
- Target: 25(OH)D >30 ng/mL
-
Vitamin K deficiency:
- Bleeding: 10 mg IV (emergency), 5 mg oral daily (maintenance)
- Warfarin reversal: 2.5-10 mg oral based on INR
- Newborn prophylaxis: 1 mg IM at birth
Special Population Considerations:
- Alcoholics: Thiamine 100 mg IV before glucose administration
- Malabsorption: Parenteral routes for fat-soluble vitamins
- Pregnancy: Folate 400-800 μg daily, B12 monitoring
- Elderly: B12 absorption decreases >60 years due to achlorhydria
💡 Master This: "Thiamine before glucose" - Glucose administration without thiamine replacement can precipitate Wernicke encephalopathy in thiamine-deficient patients
Treatment Response Monitoring:
- Hematological response: Reticulocytosis in 3-5 days (B12/folate)
- Neurological improvement: Weeks to months (may be incomplete)
- Biochemical normalization: MMA (2-4 weeks), Homocysteine (1-2 weeks)
Toxicity Prevention:
- Fat-soluble vitamins: Monitor for accumulation - Vitamin A >25,000 IU/day, Vitamin D >4000 IU/day
- Water-soluble vitamins: Generally safe except B6 >100 mg/day (neuropathy)
Connect these evidence-based treatment protocols through advanced integration strategies to understand how vitamin replacement therapy interacts with other medications and medical conditions.
💊 The Therapeutic Command Center: Evidence-Based Interventions
🌐 The Integration Network: Multi-System Vitamin Interactions
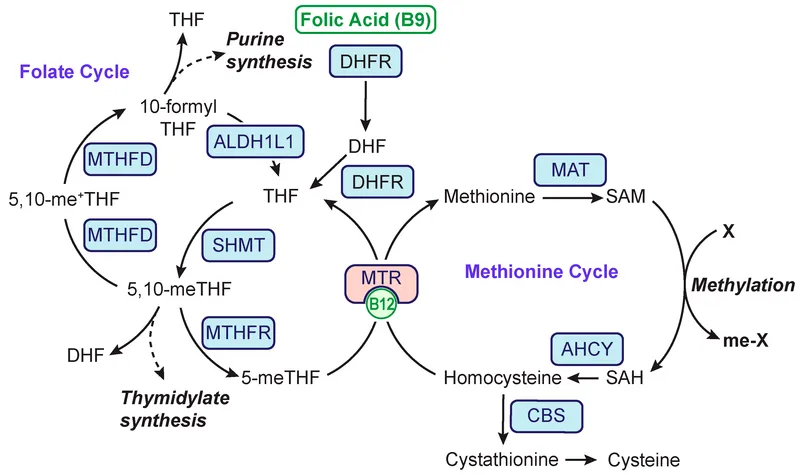
📌 Remember: "METHYL-REDOX-ENERGY" - Methylation (B12/Folate), Redox balance (C/E), Energy production (B1/B2/B3) represent the three major vitamin interaction networks
One-Carbon Metabolism Integration:
-
Folate-B12-B6 triad controls methylation reactions
- Methionine synthase: Requires methylcobalamin (B12) + 5-methylTHF (folate)
- Homocysteine metabolism: B6 (cystathionine synthase) provides alternative pathway
- SAM:SAH ratio: >3:1 indicates adequate methylation capacity
-
Clinical integration patterns:
- Combined B12/Folate deficiency: Homocysteine >50 μmol/L
- B6 deficiency: Homocysteine elevation despite normal B12/folate
- Methylation dysfunction: DNA hypomethylation, increased cancer risk
Antioxidant Network Synergy:
-
Vitamin C-E recycling system:
- α-tocopherol radical + Ascorbate → α-tocopherol + Ascorbyl radical
- Glutathione regenerates ascorbate from ascorbyl radical
- Network efficiency: 10-fold increase in antioxidant capacity
-
Selenium-Vitamin E interaction:
- Glutathione peroxidase (selenium) + α-tocopherol = synergistic protection
- Deficiency combination: Keshan disease (cardiomyopathy)
| Vitamin Pair | Interaction Type | Clinical Significance | Synergy Factor | Deficiency Risk |
|---|---|---|---|---|
| B12 + Folate | Methylation | DNA synthesis | 5-fold | Megaloblastic anemia |
| C + E | Antioxidant | Membrane protection | 10-fold | Oxidative stress |
| D + K | Bone metabolism | Calcium regulation | 3-fold | Osteoporosis |
| B1 + B2 + B3 | Energy | ATP production | 8-fold | Metabolic dysfunction |
| A + E | Membrane | Visual/antioxidant | 4-fold | Night blindness |
Energy Metabolism Integration:
-
B-vitamin complex in ATP production:
- B1 (TPP): Pyruvate dehydrogenase, α-ketoglutarate dehydrogenase
- B2 (FAD): Complex I, Complex II electron transport
- B3 (NAD+): Glycolysis, TCA cycle, β-oxidation
- B5 (CoA): Acetyl-CoA formation, fatty acid metabolism
-
Metabolic efficiency: Complete B-complex increases ATP yield by >200% compared to individual vitamins
Cutting-Edge Research Insights:
- Epigenetic vitamin interactions: Folate, B12, choline control DNA methylation patterns
- Microbiome-vitamin synthesis: Gut bacteria produce vitamin K, biotin, folate
- Pharmacogenomics: MTHFR polymorphisms affect folate requirements by 50-100%
Advanced Clinical Applications:
- Cardiovascular protection: B6 + B12 + Folate reduces homocysteine and CVD risk by 15%
- Cognitive preservation: B-complex + antioxidants slow cognitive decline by 30%
- Cancer prevention: Adequate folate (not excess) reduces colorectal cancer risk by 20%
💡 Master This: "Vitamin networks amplify effects" - Combination therapy targeting metabolic networks achieves superior outcomes compared to isolated vitamin replacement, explaining why multivitamin formulations often outperform single nutrients
Therapeutic Network Strategies:
- Methylation support: B12 + Folate + B6 + Betaine
- Antioxidant protection: C + E + Selenium + Glutathione
- Energy optimization: Complete B-complex + CoQ10 + Magnesium
Connect these multi-system integration concepts through rapid mastery frameworks to understand how vitamin network optimization transforms clinical practice and patient outcomes.
🌐 The Integration Network: Multi-System Vitamin Interactions
🎯 The Clinical Mastery Toolkit: Rapid Assessment & Intervention
📌 Remember: "SPOT-DOSE-MONITOR-PREVENT" - Rapid recognition, Appropriate dosing, Response monitoring, Recurrence prevention for vitamin deficiency mastery
The 30-Second Vitamin Assessment:
- Neurological screen: B1 (confusion), B12 (neuropathy), B6 (seizures)
- Hematological clues: Megaloblastic (B12/Folate), Microcytic (B6), Bleeding (K)
- Dermatological signs: Cheilosis (B2), Pellagra (B3), Night blindness (A)
- Risk factors: Alcohol, malabsorption, dietary restriction, medications
Essential Clinical Arsenal:
- Critical thresholds: B12 <200 pg/mL, 25(OH)D <20 ng/mL, MMA >1000 nmol/L
- Emergency doses: Thiamine 100 mg IV, B12 1000 μg IM, Vitamin K 10 mg IV
- Monitoring targets: MMA normalization (2-4 weeks), Homocysteine <15 μmol/L
| Clinical Scenario | Suspect Vitamin | First Test | Emergency Dose | Monitoring |
|---|---|---|---|---|
| Confusion + Alcohol | B1 | Thiamine level | 100 mg IV | Clinical response |
| Megaloblastic anemia | B12/Folate | B12, Folate, MMA | 1000 μg IM | MMA, Homocysteine |
| Bleeding + Antibiotics | K | PT/INR | 10 mg IV | INR normalization |
| Night blindness | A | Serum retinol | 200,000 IU | Visual improvement |
| Bone pain + Low sun | D | 25(OH)D | 50,000 IU weekly | 25(OH)D >30 ng/mL |
Rapid Intervention Protocols:
- Alcoholic patient: Thiamine 100 mg IV before any glucose administration
- Megaloblastic anemia: B12 1000 μg IM + Folate 5 mg oral simultaneously
- Malabsorption syndrome: Fat-soluble vitamin panel + parenteral replacement
💡 Master This: Vitamin deficiency mastery transforms scattered symptoms into predictable patterns, complex biochemistry into actionable protocols, and uncertain diagnoses into confident interventions that optimize patient outcomes and prevent complications
🎯 The Clinical Mastery Toolkit: Rapid Assessment & Intervention
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more
Have doubts about this lesson?
Ask Rezzy, your AI Study Mate, to explain anything you didn't understand
Everything you need for NEET-PG prep
Get full Oncourse access with lessons, practice questions, flashcards and AI study tools.
Scan to download app
























