Signal Transduction

On this page
🎯 Signal Transduction Mastery: Your Cellular Command Center
Cells receive thousands of signals every second, yet they respond with remarkable precision-converting a single hormone molecule at the surface into cascading intracellular events that alter metabolism, gene expression, and survival itself. You'll master how G-proteins act as molecular switches, how second messengers amplify signals a millionfold, and how these pathways explain diseases from cholera to cancer while revealing targeted treatment strategies. This is your blueprint for understanding the command-and-control systems that orchestrate human physiology and the clinical consequences when signaling goes wrong.
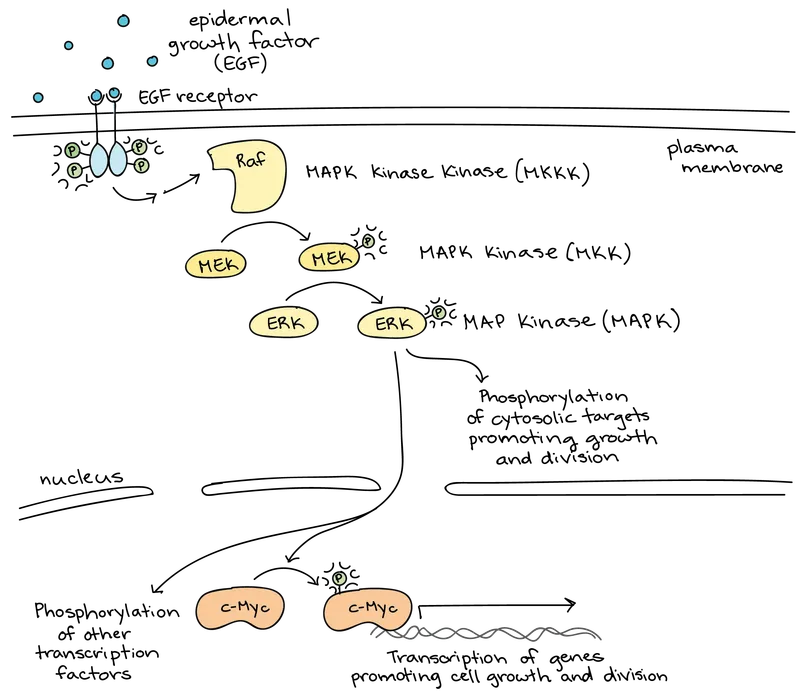
📌 Remember: SIGNAL - Stimulus detection, Intracellular cascade, Gprotein coupling, Nuclear response, Amplification occurs, Ligand specificity determines cellular fate with 10,000-fold amplification possible
Signal Transduction Architecture
-
Receptor Classes (4 major types)
- G-protein coupled receptors: >800 subtypes, 40% of drug targets
- Enzyme-linked receptors: 58 receptor tyrosine kinases identified
- Insulin receptor: 2 α and 2 β subunits
- Growth factor receptors: >20 families characterized
- Ion channel receptors: >400 subtypes, millisecond response times
- Nuclear receptors: 48 human subtypes, hours to response
-
Signal Amplification Cascade
- Single hormone molecule: 1 ligand binding event
- G-protein activation: 100 molecules activated per receptor
- Second messenger production: 10,000 cAMP molecules generated
- Protein kinase activation: 1,000,000 phosphorylation events
- Total amplification: 10^9-fold signal enhancement possible
| Receptor Type | Response Time | Amplification | Drug Targets | Clinical Examples |
|---|---|---|---|---|
| GPCRs | Seconds | 10^6-fold | 40% all drugs | β-blockers, antihistamines |
| RTKs | Minutes | 10^4-fold | 25% cancer drugs | Insulin, growth factors |
| Ion Channels | Milliseconds | 10^2-fold | 15% neurologic drugs | Anesthetics, anticonvulsants |
| Nuclear | Hours | 10^3-fold | 20% hormonal drugs | Steroids, thyroid hormones |
💡 Master This: Signal specificity depends on 3 critical factors: receptor distribution (tissue-specific), ligand concentration (nanomolar to micromolar), and temporal dynamics (seconds to hours) - understanding these relationships predicts every therapeutic response pattern
Understanding signal transduction fundamentals establishes the foundation for exploring how G-protein mechanisms orchestrate cellular responses with extraordinary precision and clinical relevance.
🎯 Signal Transduction Mastery: Your Cellular Command Center
⚙️ G-Protein Mechanisms: The Molecular Switch Network
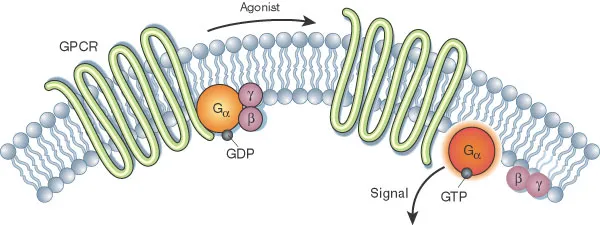
📌 Remember: GPCR CYCLE - GDP bound (inactive), Phosphorylation triggers, Conformational change, Release of Gα subunit, CAMP/IP3 production, Yield cellular response, Cleaved by GTPase, Ligand dissociation, Ends signaling cycle
G-Protein Subunit Classification
-
Gαs Subfamily (stimulatory pathway)
- Adenylyl cyclase activation: 10-fold cAMP increase
- PKA phosphorylation: >100 target proteins
- CREB phosphorylation: Ser133 residue critical
- Glycogen phosphorylase: Ser14 activation site
- Clinical targets: β2-agonists (albuterol), glucagon therapy
-
Gαq/11 Subfamily (phospholipase pathway)
- PLC-β activation: 5-second response time
- IP3 generation: 50-fold increase within 10 seconds
- Ca2+ release: 100-1000 nM cytoplasmic levels
- DAG production: PKC activation within 30 seconds
- Clinical relevance: α1-adrenergic receptors, angiotensin II signaling
| G-Protein Type | Primary Effector | Second Messenger | Response Time | Clinical Examples |
|---|---|---|---|---|
| Gαs | Adenylyl cyclase | ↑ cAMP | 5-10 sec | β-agonists, glucagon |
| Gαq/11 | Phospholipase C | ↑ IP3/DAG | 2-5 sec | α1-agonists, vasopressin |
| Gαi/o | Inhibits AC | ↓ cAMP | 3-8 sec | α2-agonists, opioids |
| Gα12/13 | RhoGEF | Rho activation | 10-30 sec | Thrombin, LPA |
💡 Master This: G-protein selectivity depends on receptor conformation changes that expose specific G-protein binding domains - this explains why single receptors can couple to multiple G-proteins depending on ligand concentration and tissue context
Understanding G-protein mechanisms reveals how cells achieve signal specificity, setting the stage for exploring second messenger amplification systems that transform these molecular switches into powerful cellular responses.
⚙️ G-Protein Mechanisms: The Molecular Switch Network
🔄 Second Messenger Amplification: The Signal Multiplication Matrix
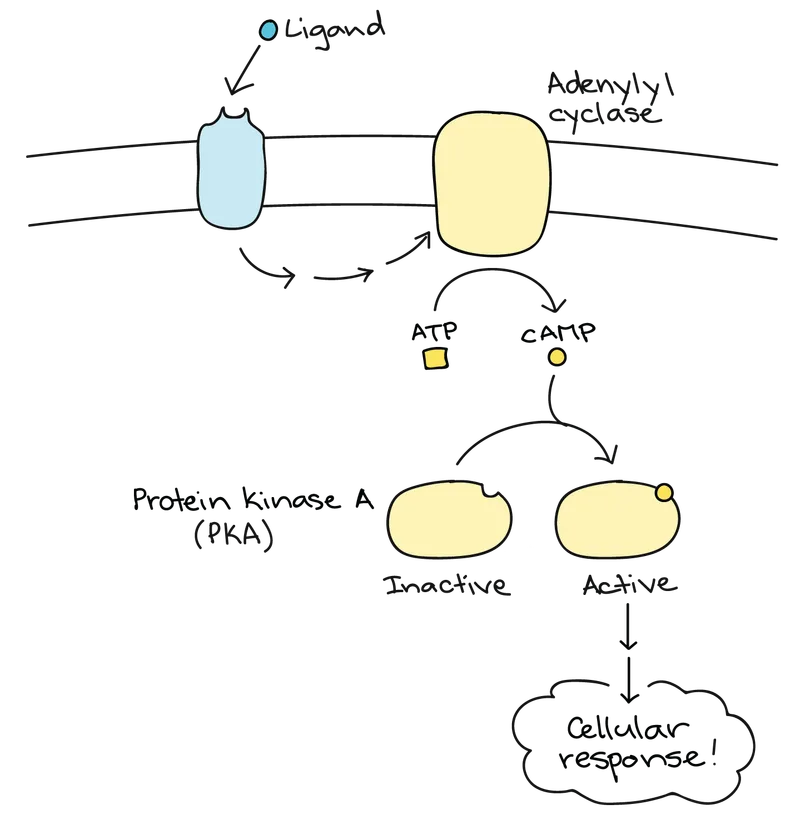
📌 Remember: cAMP CASCADE - cAMP synthesis, Adenylyl cyclase activation, Multiple PKA subunits, Phosphorylation targets, CREB activation, Amplification occurs, Signal specificity, Cellular response, ATP consumption, Degradation by PDE, Ends response cycle
cAMP Signaling Architecture
-
cAMP Production Kinetics
- Basal adenylyl cyclase: 10-50 pmol/min/mg protein
- Stimulated activity: 500-2000 pmol/min/mg (40-fold increase)
- Peak cAMP levels: 10-100 μM intracellular
- Response duration: 5-30 minutes depending on PDE activity
- PKA activation: EC50 = 150 nM cAMP concentration
-
Protein Kinase A Cascade
- Regulatory subunit dissociation: 4 cAMP molecules required
- Catalytic subunit release: 2 active kinases per holoenzyme
- Phosphorylation rate: >1000 substrates/minute per kinase
- Consensus sequence: R-R-X-S/T (Arg-Arg-any-Ser/Thr)
- Nuclear translocation: 50% of catalytic subunits within 10 minutes
| Second Messenger | Synthesis Rate | Peak Concentration | Half-life | Primary Targets |
|---|---|---|---|---|
| cAMP | 40-fold ↑ | 10-100 μM | 30 sec | PKA, EPAC |
| IP3 | 50-fold ↑ | 1-10 μM | 5 sec | IP3 receptors |
| DAG | 20-fold ↑ | 10-50 μM | 60 sec | PKC isoforms |
| Ca2+ | 1000-fold ↑ | 1-10 μM | 2 sec | Calmodulin, troponin |
| cGMP | 100-fold ↑ | 1-50 μM | 10 sec | PKG, ion channels |
-
Intracellular Calcium Mobilization
- Resting [Ca2+]: 50-100 nM cytoplasmic
- Stimulated levels: 500-2000 nM (20-fold increase)
- ER calcium stores: 500 μM concentration
- Release kinetics: Peak in 2-5 seconds
- Calmodulin binding: 4 Ca2+ ions required for activation
-
Calcium-Dependent Processes
- Muscle contraction: Troponin C binding (4 Ca2+ sites)
- Enzyme activation: >50 calcium-dependent kinases
- CaMKII autophosphorylation: Thr286 critical residue
- Calcineurin activation: Phosphatase activity increases 10-fold
⭐ Clinical Pearl: Phosphodiesterase inhibitors (theophylline, sildenafil) block cAMP/cGMP degradation, extending second messenger half-life from 30 seconds to >5 minutes, explaining their therapeutic efficacy
💡 Master This: Second messenger specificity depends on subcellular compartmentalization - cAMP levels can vary 100-fold between membrane and nuclear regions, creating spatial gradients that determine which proteins get activated
Second messenger amplification systems provide the foundation for understanding how cells translate molecular signals into coordinated responses, leading us to explore the pattern recognition frameworks essential for clinical diagnosis.
🔄 Second Messenger Amplification: The Signal Multiplication Matrix
🎯 Signal Recognition Patterns: The Clinical Correlation Matrix
📌 Remember: PATHWAY DIAGNOSIS - Pattern recognition, Assay correlation, Timing of symptoms, Hormone levels, When receptors fail, Amplification defects, Yield clinical signs, Deficiency patterns, Inhibitor effects, Abnormal responses, Genetic mutations, Normal vs pathologic, Outcome prediction, Specific treatments, Intervention timing, System integration
Receptor Dysfunction Patterns
-
GPCR Pathway Failures
- Gs protein defects: McCune-Albright syndrome
- Clinical pattern: Precocious puberty + café-au-lait spots + fibrous dysplasia
- Biochemical signature: Elevated cAMP in affected tissues
- Mutation frequency: <1% of population, somatic mosaicism
- Gi protein dysfunction: Pseudohypoparathyroidism
- Resistance pattern: ↑ PTH with ↓ cAMP response
- Clinical triad: Short stature + brachydactyly + subcutaneous ossification
- Gs protein defects: McCune-Albright syndrome
-
Receptor Tyrosine Kinase Disorders
- Insulin receptor mutations: Type A insulin resistance
- Biochemical pattern: Severe hyperinsulinemia (>100 μU/mL)
- Clinical presentation: Acanthosis nigricans + hirsutism + ovarian dysfunction
- Prevalence: 1 in 60,000 births
- Growth hormone receptor defects: Laron syndrome
- Hormone pattern: ↑ GH (>40 ng/mL) with ↓ IGF-1 (<50 ng/mL)
- Clinical features: Severe dwarfism + normal intelligence
- Insulin receptor mutations: Type A insulin resistance
| Pathway Defect | Hormone Pattern | Response Time | Clinical Clues | Diagnostic Test |
|---|---|---|---|---|
| GPCR dysfunction | ↑ Hormone, ↓ cAMP | Minutes | Resistance syndromes | Urinary cAMP |
| RTK mutations | ↑ Ligand, ↓ Response | Hours | Growth/metabolic issues | Receptor sequencing |
| Nuclear receptor | ↑ Hormone, ↓ Gene expression | Days | Developmental defects | Transcription assays |
| Second messenger | Normal hormone, ↓ Amplification | Variable | Partial resistance | Enzyme activity |
-
Pathway-Specific Drug Actions
- β-adrenergic system: >20 cardiovascular medications
- β1-selective: Metoprolol (Ki = 1 nM for β1 vs 100 nM for β2)
- β2-selective: Albuterol (EC50 = 0.1 μM for bronchodilation)
- Response onset: 5-15 minutes for β2-agonists
- Phosphodiesterase targeting: Tissue-specific PDE isoforms
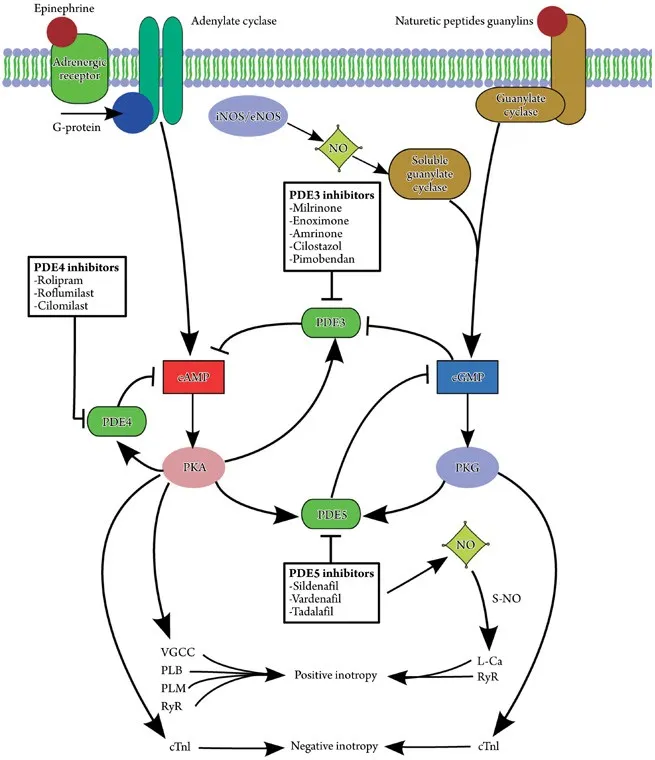
- PDE3 inhibition: Milrinone for heart failure (↑ cardiac cAMP)
- PDE5 inhibition: Sildenafil for pulmonary hypertension (↑ vascular cGMP)
- β-adrenergic system: >20 cardiovascular medications
-
Signal Amplification Therapeutics
- Insulin sensitizers: Metformin enhances AMPK activation
- Mechanism: ↑ AMP:ATP ratio triggers metabolic switching
- Clinical effect: 20-30% reduction in hepatic glucose production
- Growth factor modulators: mTOR inhibitors in cancer
- Target: Rapamycin-sensitive growth signaling
- Response rate: 30-60% in specific tumor types
- Insulin sensitizers: Metformin enhances AMPK activation
⭐ Clinical Pearl: Hormone resistance syndromes show inverse correlation between hormone levels and clinical response - 10-fold ↑ hormone with 90% ↓ biological effect indicates receptor or post-receptor defects
💡 Master This: Pattern recognition depends on temporal relationships - acute responses (minutes) suggest membrane receptor issues, delayed responses (hours-days) indicate nuclear receptor or transcriptional defects
Signal recognition patterns provide the diagnostic framework for understanding pathway dysfunction, preparing us to explore the systematic approaches used in differential diagnosis of signal transduction disorders.
🎯 Signal Recognition Patterns: The Clinical Correlation Matrix
🔬 Differential Diagnosis Framework: The Pathway Detective System
📌 Remember: DIFFERENTIAL SIGNALS - Determine hormone levels, Identify receptor status, Function tests performed, Family history checked, Enzyme activity measured, Response to stimulation, Elimination of alternatives, Nuclear studies done, Timing of symptoms, Imaging correlations, Amplification assessed, Laboratory patterns, Specific mutations, Inhibitor responses, Genetic counseling, Normal variants ruled out, Age-related changes, Lifestyle factors, Systemic effects
Hormone-Receptor Mismatch Analysis
-
Primary Resistance Patterns
- Androgen insensitivity syndrome: Complete vs partial
- Complete AIS: 46,XY with female phenotype, testosterone >300 ng/dL
- Partial AIS: Ambiguous genitalia, variable virilization
- Receptor binding: <10% of normal DHT binding capacity
- Prevalence: 1 in 20,000 to 1 in 64,000 births
- Thyroid hormone resistance: RTHα vs RTHβ
- RTHβ mutations: ↑ TSH (>10 mIU/L) with ↑ T4 (>12 μg/dL)
- Clinical pattern: Tachycardia + attention deficit + normal growth
- Mutation frequency: >3000 families identified worldwide
- Androgen insensitivity syndrome: Complete vs partial
-
Secondary Signaling Defects
- Pseudohypoparathyroidism type 1a: Gs protein deficiency
- Biochemical: ↑ PTH (>100 pg/mL) + ↓ urinary cAMP (<1 nmol/L)
- Physical features: Albright hereditary osteodystrophy phenotype
- Hormone resistance: Multiple (PTH, TSH, LH, FSH)
- McCune-Albright syndrome: Gs protein hyperactivity
- Somatic mutations: Gαs Arg201 or Gln227 residues
- Clinical triad: Polyostotic fibrous dysplasia + café-au-lait spots + precocious puberty
- Pseudohypoparathyroidism type 1a: Gs protein deficiency
| Disorder Category | Hormone Pattern | Receptor Function | Second Messenger | Genetic Basis |
|---|---|---|---|---|
| Primary resistance | ↑↑ Hormone | <10% binding | Variable | Receptor mutations |
| Post-receptor defects | ↑ Hormone | Normal binding | <50% response | Signaling protein mutations |
| Amplification defects | Normal hormone | Normal binding | Reduced magnitude | Enzyme deficiencies |
| Feedback disorders | Variable | Normal function | Normal response | Regulatory mutations |
| Hypersensitivity | ↓ Hormone | ↑ Sensitivity | Excessive response | Gain-of-function mutations |
-
Step 1: Hormone Level Assessment
- Basal measurements: Morning cortisol, fasting insulin, TSH/T4
- Dynamic testing: Stimulation (ACTH, TRH) and suppression (dexamethasone)
- Normal cortisol response: >18 μg/dL post-ACTH stimulation
- Insulin suppression: <2 μU/mL during 75g OGTT
- Temporal patterns: Circadian rhythms, pulsatile secretion
-
Step 2: Receptor Function Analysis
- Binding studies: Radioligand binding assays
- Normal insulin binding: 8-12% at 37°C for 2 hours
- Receptor number: 10,000-40,000 sites per cell
- Functional assays: Second messenger generation
- cAMP response: 5-20 fold increase over basal
- Calcium mobilization: Peak within 30 seconds
- Binding studies: Radioligand binding assays
-
Step 3: Downstream Pathway Assessment
- Enzyme activity: Adenylyl cyclase, phospholipase C
- Protein phosphorylation: Western blot analysis
- PKA substrates: CREB Ser133, ACC Ser79
- PKC substrates: Multiple serine/threonine residues
- Gene expression: qRT-PCR for target genes
⭐ Clinical Pearl: Discordant hormone-response patterns indicate specific pathway defects - normal receptor binding with <50% second messenger response suggests G-protein or effector enzyme mutations
💡 Master This: Temporal response analysis distinguishes pathway levels - immediate responses (<1 minute) indicate membrane events, intermediate responses (1-30 minutes) suggest second messenger cascades, delayed responses (>1 hour) point to transcriptional mechanisms
Differential diagnosis frameworks provide systematic approaches to pathway analysis, setting the foundation for exploring evidence-based treatment algorithms that target specific signal transduction defects.
🔬 Differential Diagnosis Framework: The Pathway Detective System
⚕️ Treatment Algorithm Mastery: The Therapeutic Precision Protocol
📌 Remember: TREATMENT PRECISION - Target identification, Receptor selectivity, Efficacy measurement, Adverse effects monitored, Timing optimization, Mechanism-based dosing, Endpoint assessment, Normal function preserved, Toxicity prevention, Patient selection, Response prediction, Evidence-based protocols, Combination strategies, Individualized therapy, Safety monitoring, Improvement tracking, Outcome optimization, New approaches considered
Pathway-Specific Therapeutic Strategies
-
GPCR-Targeted Interventions
- β-adrenergic modulation: Cardiovascular applications
- β1-selective blockade: Metoprolol (25-200 mg BID)
- Efficacy: 25-35% reduction in cardiovascular mortality
- Response time: Peak effect in 1-2 hours, steady state in 3-5 days
- Selectivity ratio: β1:β2 = 75:1 at therapeutic doses
- α-adrenergic targeting: Hypertension management
- α1-blockade: Doxazosin (1-16 mg daily)
- Blood pressure reduction: 10-15 mmHg systolic, 5-10 mmHg diastolic
- Response rate: >70% of patients achieve <140/90 mmHg
- β-adrenergic modulation: Cardiovascular applications
-
Enzyme-Linked Receptor Therapeutics
- Insulin sensitizer protocols: Type 2 diabetes management
- Metformin: 500-2000 mg daily, ↑ AMPK activation
- HbA1c reduction: 0.5-1.5% decrease from baseline
- Weight effect: 2-5 kg weight loss over 6 months
- Growth factor inhibition: Cancer therapeutics
- Trastuzumab: HER2-positive breast cancer
- Response rate: 35-50% in metastatic disease
- Survival benefit: 4-6 month median survival extension
- Insulin sensitizer protocols: Type 2 diabetes management
| Therapeutic Class | Target Pathway | Onset Time | Peak Effect | Response Rate | Monitoring Parameter |
|---|---|---|---|---|---|
| β-blockers | Gαs-cAMP | 1-2 hours | 3-5 days | 70-85% | Heart rate, BP |
| PDE inhibitors | cAMP/cGMP | 30-60 min | 2-4 hours | 60-80% | Functional capacity |
| Insulin sensitizers | RTK-PI3K | 2-4 weeks | 8-12 weeks | 65-75% | HbA1c, HOMA-IR |
| Steroid antagonists | Nuclear receptors | Days-weeks | 4-8 weeks | 50-70% | Hormone levels |
| Calcium blockers | Ca2+ signaling | 30-60 min | 2-6 hours | 75-90% | BP, arrhythmias |
-
Receptor Occupancy-Based Dosing
- β-blocker optimization: Target 60-80% receptor occupancy
- Dose titration: Start 25% target, increase weekly
- Monitoring: Resting HR 55-65 bpm, exercise HR <85% predicted
- Maximum benefit: >75% β1-receptor blockade required
- Insulin therapy: Physiologic replacement patterns
- Basal insulin: 0.2-0.4 units/kg/day (40-50% total daily dose)
- Bolus insulin: 1 unit per 10-15g carbohydrate
- Target glucose: 70-180 mg/dL (>70% time in range)
- β-blocker optimization: Target 60-80% receptor occupancy
-
Second Messenger Modulation
- PDE inhibitor dosing: Tissue-specific targeting
- Sildenafil: 25-100 mg for PDE5 inhibition
- Selectivity: PDE5:PDE6 = 10:1 (visual side effects)
- Duration: 4-6 hours effective cGMP elevation
- Theophylline protocols: PDE3/4 inhibition
- Therapeutic range: 10-20 μg/mL serum concentration
- Dosing: 10-15 mg/kg/day divided BID-TID
- Monitoring: Weekly levels until stable
- PDE inhibitor dosing: Tissue-specific targeting
Combination Therapy Strategies
- Synergistic Pathway Targeting
- Diabetes combination: Metformin + GLP-1 agonist
- Mechanism: AMPK activation + cAMP elevation
- HbA1c reduction: 1.5-2.0% combined effect
- Weight loss: 5-10 kg over 12 months
- Heart failure protocol: ACE inhibitor + β-blocker + diuretic
- Mortality reduction: 35-45% with triple therapy
- Hospitalization: 50-60% reduction in HF admissions
- Diabetes combination: Metformin + GLP-1 agonist
⭐ Clinical Pearl: Therapeutic response prediction requires pathway genotyping - CYP2D6 poor metabolizers need 50% dose reduction for β-blockers, while ADRB1 Arg389Gly polymorphism affects β-blocker efficacy by 20-30%
💡 Master This: Dose-response relationships follow receptor occupancy curves - 50% receptor occupancy typically produces 80% maximum response due to receptor reserve, explaining why moderate doses often achieve near-maximal clinical effects
Treatment algorithm mastery provides the therapeutic foundation for understanding pathway interventions, leading us to explore the advanced integration concepts that connect multiple signaling systems in complex physiological networks.
⚕️ Treatment Algorithm Mastery: The Therapeutic Precision Protocol
🌐 Multi-System Integration Hub: The Signaling Convergence Network
📌 Remember: NETWORK INTEGRATION - Nodes of convergence, Effector sharing, Temporal coordination, Waveform patterns, Organ specificity, Regulatory feedback, Kinetic matching, Inhibitory crosstalk, Nuclear integration, Tissue responses, Energy coordination, Growth signals, Repair mechanisms, Adaptive responses, Toxicity prevention, Immune coordination, Oxidative balance, Nutrient sensing
Signaling Network Convergence Points
-
mTOR Integration Hub (Master growth coordinator)
- Input pathways: >15 signaling networks converge
- Insulin/IGF-1: PI3K-Akt activation (EC50 = 10 nM)
- Amino acids: Rag GTPase complex (leucine sensing)
- Energy status: AMPK inhibition (AMP:ATP >3:1)
- Oxygen levels: HIF-1α regulation (<5% O2)
- Output coordination: >200 downstream targets
- Protein synthesis: S6K1 and 4E-BP1 phosphorylation
- Lipid synthesis: SREBP-1 activation (>5-fold increase)
- Autophagy: ULK1 Ser757 phosphorylation (inhibitory)
- Input pathways: >15 signaling networks converge
-
AMPK Energy Sensor (Metabolic master switch)
- Activation triggers: AMP:ATP ratio >2:1
- Exercise: >10-fold AMPK activation in muscle
- Fasting: 3-5 fold activation in liver after 12 hours
- Hypoxia: >20-fold activation when O2 <2%
- Pathway coordination: >50 metabolic enzymes regulated
- Fatty acid oxidation: ACC Ser79 phosphorylation (↑ β-oxidation)
- Glucose uptake: GLUT4 translocation (insulin-independent)
- Protein synthesis: mTOR inhibition (energy conservation)
- Activation triggers: AMP:ATP ratio >2:1
| Integration Node | Input Signals | Response Time | Output Pathways | Clinical Relevance |
|---|---|---|---|---|
| mTOR | Growth factors, nutrients | Minutes | >200 targets | Cancer, aging, diabetes |
| AMPK | Energy, exercise, drugs | Seconds | >50 enzymes | Metabolism, longevity |
| p53 | DNA damage, stress | Hours | >500 genes | Cancer, cell death |
| NF-κB | Inflammation, infection | Minutes | >150 genes | Immunity, inflammation |
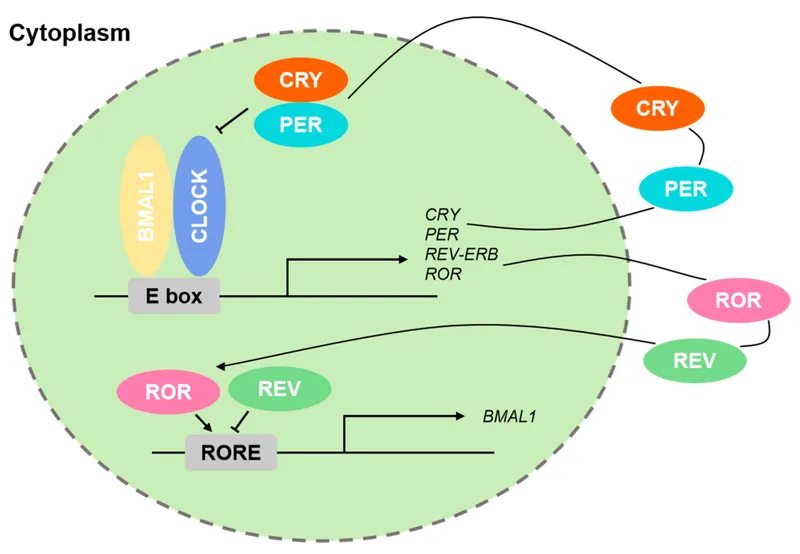
| CLOCK/BMAL1 | Light, feeding | Hours | >1000 genes | Circadian disorders |
-
Circadian Signaling Architecture
- Master clock: SCN neurons (~20,000 cells)
- Period: 24.2 ± 0.2 hours intrinsic rhythm
- Light entrainment: Melanopsin pathway (480 nm peak sensitivity)
- Temperature cycles: ±1-2°C core body temperature
- Peripheral clocks: >95% of tissues have autonomous oscillators
- Liver: >3000 genes show circadian expression
- Muscle: >1500 genes with metabolic rhythms
- Adipose: >800 genes controlling lipid metabolism
- Master clock: SCN neurons (~20,000 cells)
-
Stress Response Integration
- HPA axis coordination: Hypothalamic-pituitary-adrenal
- CRH release: Peak at 6-8 AM (circadian maximum)
- Cortisol rhythm: 10-25 μg/dL morning, <5 μg/dL evening
- Feedback loops: >5 negative feedback mechanisms
- Sympathetic activation: Fight-or-flight responses
- Norepinephrine: >10-fold increase during acute stress
- Duration: Minutes to hours depending on stressor intensity
- Recovery: 30-60 minutes to baseline levels
- HPA axis coordination: Hypothalamic-pituitary-adrenal
Cutting-Edge Integration Mechanisms
-
Exosome-Mediated Communication
- Intercellular signaling: 30-150 nm vesicles
- Content: >1000 different miRNAs and proteins
- Range: Local (μm) to systemic (circulation)
- Specificity: Tissue-specific surface receptor targeting
- Clinical applications: Biomarker discovery and therapeutic delivery
- Cancer diagnosis: >50 exosomal biomarkers identified
- Drug delivery: >90% targeting efficiency possible
- Intercellular signaling: 30-150 nm vesicles
-
Metabolite Signaling Networks
- Metabolic intermediates as signaling molecules
- α-ketoglutarate: Epigenetic regulation (histone demethylation)
- Acetyl-CoA: Histone acetylation (gene activation)
- NAD+/NADH: Sirtuin activation (longevity pathways)
- Tissue communication: >200 metabolites in circulation
- Muscle-derived: Myokines (>50 identified)
- Adipose-derived: Adipokines (>100 characterized)
- Gut-derived: Short-chain fatty acids (microbiome signals)
- Metabolic intermediates as signaling molecules
⭐ Clinical Pearl: Chronotherapy leverages circadian integration - statins are 30-40% more effective when dosed at bedtime due to peak HMG-CoA reductase activity during sleep, while blood pressure medications show optimal efficacy with evening dosing
💡 Master This: Network robustness depends on redundant pathways - >80% of essential cellular functions have ≥3 backup mechanisms, explaining why single pathway inhibitors often show limited clinical efficacy compared to combination approaches
Multi-system integration reveals the sophisticated coordination underlying physiological responses, preparing us to synthesize these concepts into practical clinical mastery tools for immediate application.
🌐 Multi-System Integration Hub: The Signaling Convergence Network
🎖️ Clinical Mastery Arsenal: The Signal Transduction Command Center
📌 Remember: MASTERY ESSENTIALS - Mechanism understanding, Assessment protocols, Signal recognition, Therapeutic targeting, Evidence integration, Rapid diagnosis, Yield optimization, Error prevention, Safety monitoring, System thinking, Efficiency maximization, New developments, Timing precision, Individualization, Adaptive strategies, Lifelong learning, Synthetic approaches
Essential Clinical Arsenal
-
Rapid Pathway Assessment Protocol
- 5-Minute Diagnostic Framework
- Step 1: Hormone levels (high/normal/low) - 30 seconds
- Step 2: Clinical response pattern (present/absent/partial) - 60 seconds
- Step 3: Temporal characteristics (acute/chronic/episodic) - 30 seconds
- Step 4: Family history (genetic/sporadic) - 60 seconds
- Step 5: Treatment response (responsive/resistant) - 120 seconds
- 5-Minute Diagnostic Framework
-
Critical Numbers Arsenal
- GPCR Pathways: β-blocker heart rate target 55-65 bpm
- RTK Systems: Insulin resistance HOMA-IR >2.5
- Nuclear Receptors: Thyroid resistance TSH >10 mIU/L + T4 >12 μg/dL
- Second Messengers: Normal cAMP response >5-fold increase
- Calcium Signaling: Cytoplasmic Ca2+ 100-1000 nM range
| Clinical Scenario | Key Pathway | Diagnostic Test | Normal Response | Abnormal Pattern |
|---|---|---|---|---|
| Cardiac failure | β-adrenergic | Exercise tolerance | >5 METs | <3 METs |
| Diabetes | Insulin signaling | HOMA-IR | <2.5 | >4.0 |
| Hypertension | RAAS/sympathetic | 24h BP | <130/80 | >140/90 |
| Thyroid disease | TSH-receptor | TSH stimulation | T4 ↑ 2-3x | No response |
| Growth disorders | GH-IGF axis | IGF-1 levels | Age-appropriate | <5th percentile |
⭐ Clinical Pearl: Resistance syndrome pattern - ↑↑ hormone + ↓ clinical response + normal receptor binding = post-receptor defect requiring alternative pathway activation or signal amplification strategies
💡 Master This: Therapeutic window optimization - receptor occupancy of 60-80% typically provides maximum clinical benefit with minimal side effects, while >90% occupancy increases adverse events without proportional efficacy gains
⭐ Clinical Pearl: Temporal response signatures distinguish pathway levels - <1 minute responses indicate ion channels, 1-30 minutes suggest second messengers, >1 hour points to gene transcription
💡 Master This: Combination therapy synergy - additive effects occur with parallel pathways, synergistic effects (>2x) occur with convergent pathways, antagonistic effects occur with competing pathways
Understanding signal transduction mastery provides the foundation for advanced clinical practice, where molecular mechanisms translate directly into diagnostic precision and therapeutic success across all medical specialties.
🎖️ Clinical Mastery Arsenal: The Signal Transduction Command Center
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more
Have doubts about this lesson?
Ask Rezzy, your AI Study Mate, to explain anything you didn't understand
Everything you need for NEET-PG prep
Get full Oncourse access with lessons, practice questions, flashcards and AI study tools.
Scan to download app
























